What is the new volume of the gas, if the volume of an ideal gas enclosed in a thin, elastic membrane in a room at sea level where the air temperature is 18°C is 8×10-3 m³: The temperature of the room is increased by 10°C?
What is the new volume of the gas, if the volume of an ideal gas enclosed in a thin, elastic membrane in a room at sea level where the air temperature is 18°C is 8×10-3 m³: The temperature of the room is increased by 10°C?
Chapter4: The Second Law Of Thermodynamics
Section: Chapter Questions
Problem 71AP: A system consisting of 20.0 mol of a monoatomic ideal gas is cooled at constant pressure from a...
Related questions
Question
100%
how can this solution be correct when the question asks if the orginal temp (18*) was raised by 10*? Shouldn't that make the final Temp 28*?
if this is so i get 1.24x10^-2
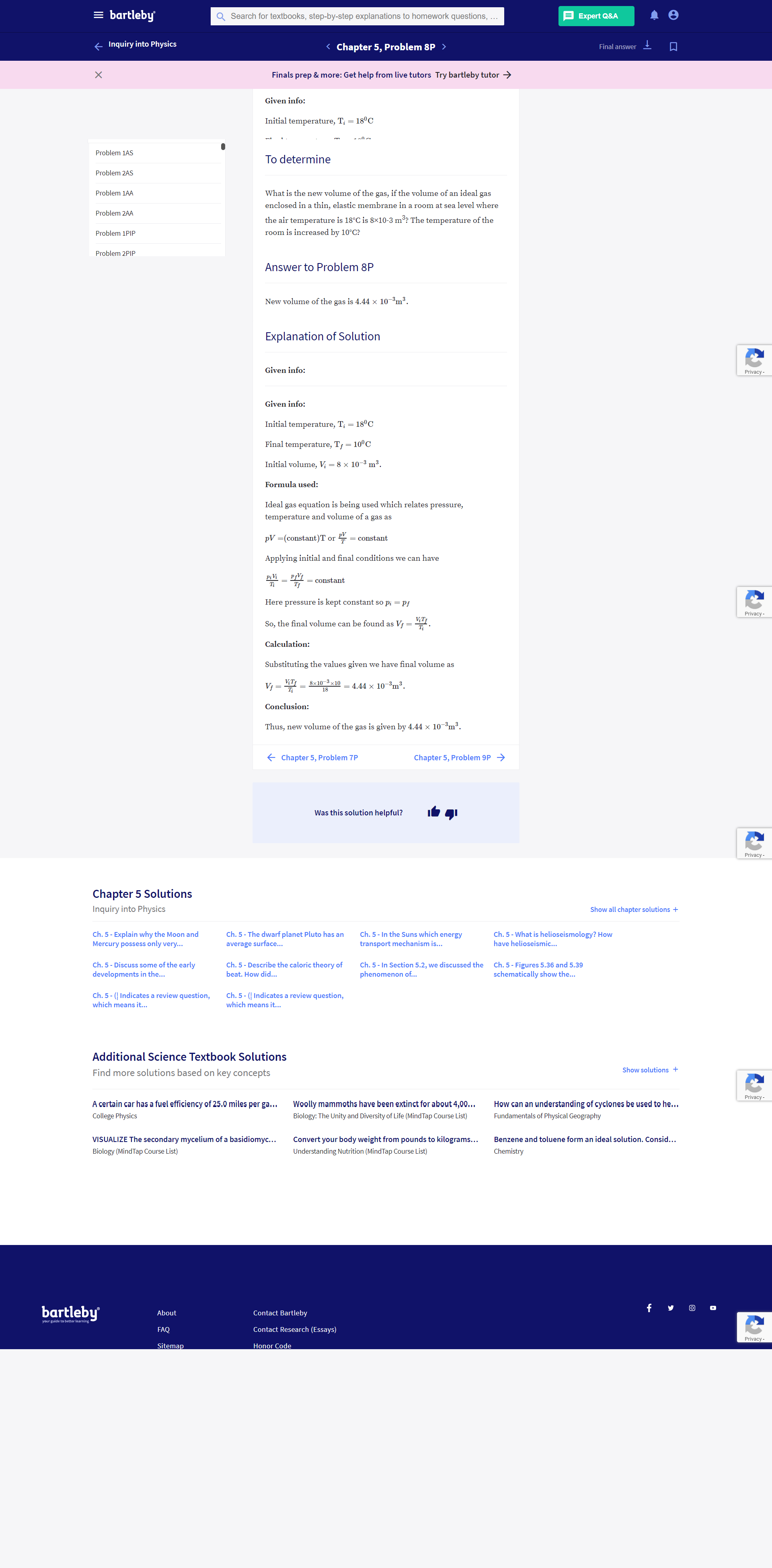
Transcribed Image Text:What is the new volume of the gas, if the volume of an ideal gas
enclosed in a thin, elastic membrane in a room at sea level where
the air temperature is 18°C is 8×10-3 m³: The temperature of the
room is increased by 10°C?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you

