Chapter33: High-performance Liquid Chromatography
Section: Chapter Questions
Problem 33.1QAP
Related questions
Question

Transcribed Image Text:What is the %composition of the Vitamin A peak in the sample?
O 78.43%
77.73%
7.867%
72.74%
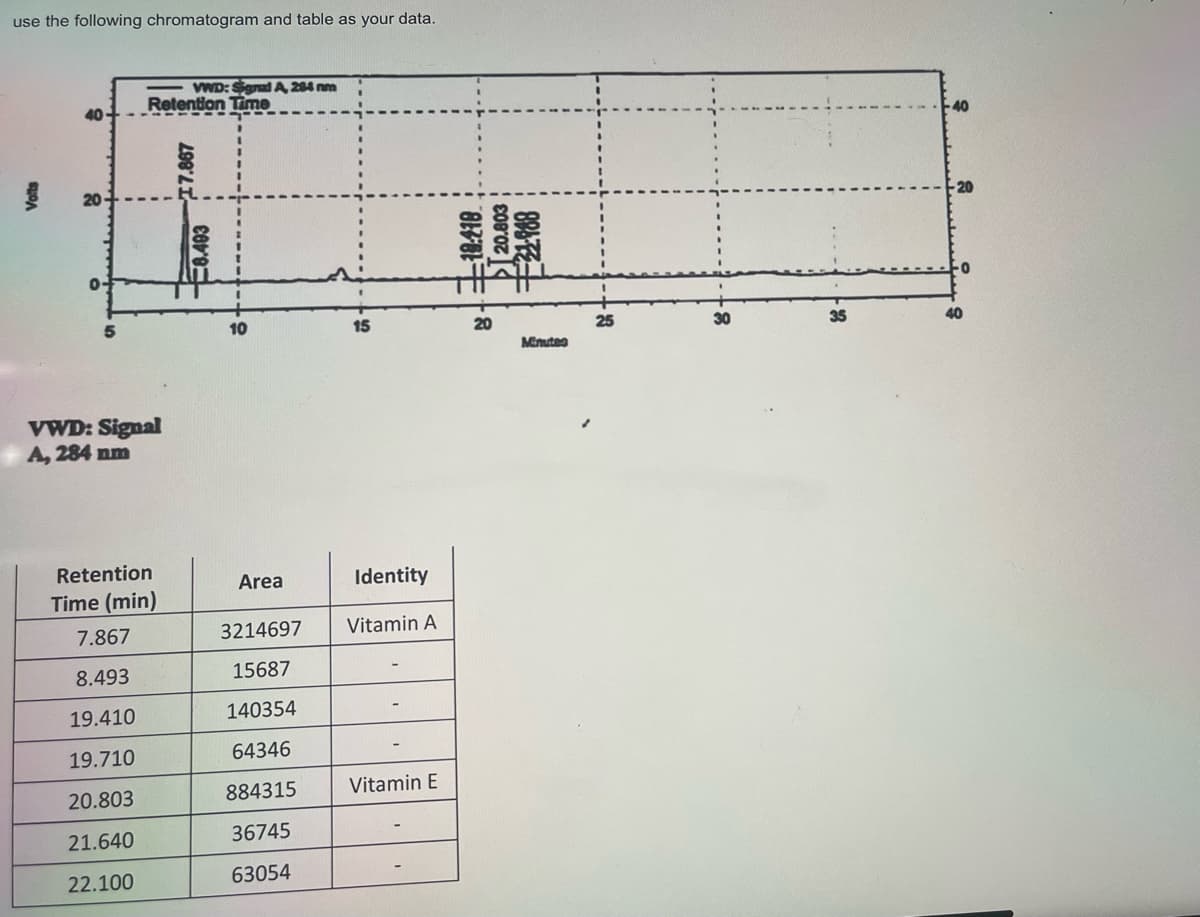
Transcribed Image Text:use the following chromatogram and table as your data.
VWD: Sgrad A 284 nm
Retention Time
40-
-40
20
20
5
10
15
20
25
30
35
Minuteo
VWD: Signal
A, 284 nm
Retention
Area
Identity
Time (min)
7.867
3214697
Vitamin A
8.493
15687
19.410
140354
19.710
64346
20.803
884315
Vitamin E
21.640
36745
22.100
63054
8.403
L98
ÞI 20.803 ;
84年
81
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole