Q: The pH of a 500.00 mL buffer solution containing sodium mandelate and mandelic acid (ka= 3.89×10-4)…
A: Given, Volume of buffer solution (V) = 500 mL Ka = 3.89 x 10-4 αo = 0.90 To find : pH
Q: Which species is responsible for the formation of red color of the solution using red cabbage…
A:
Q: A weak acid has Ka of 4.47 x 10-4. What is pKa for this weak acid?
A:
Q: 5) The oxides and hydroxides of gallium exhibit both acidic and basic behaviors B) What is pCa for a…
A: Gallium metal is stable in dry air. It gradually oxidizes in moist air to form a protective film…
Q: Concentration Buffer solution pka Desired ph 1 0.010 M Phosphate 7.21 8.0 2 0.50 M…
A: Buffer capacity can be calculated as follows, As desired pH is greater than pKa value, than…
Q: 1.Calculate the change in ph when 5ml of 1M HCl solution is added to 1L of 100mM Tris solution at ph…
A:
Q: Starting with solid glycine-HCl and 0.75 M NaOH, how would you prepare 500 mL of 0.050 M glycine…
A: The question is based on the concept of chemical equilibrium. a buffer is a solution which resist…
Q: 15.0 mL of 0.475M CH3COOH is titrated with 0.450 M RbOH. What is the pH after 18.0 mL of RbOH has…
A: Given: Concentration of CH3COOH= 0.475M Volume of CH3COOH= 15.0mL Concentration of RbOH = 0.450M…
Q: 2.A solution prepared by dissolving 2.25 g HA (MW = 51.0) and 4.00 g of NaA in 500 mL of solution…
A: Weight of HA=2.25gm MW of HA =51.0 Weight of NaH=4.00gm Volume=500ml pH=6.02 Mw of NaA = MW of HA -…
Q: Describe the preparation of 100 mL 0.050 M and 100 mL 0.0050 M phosphate buffer (both having pH =…
A: Stock solution of phosphate buffer of pH 6.5. Molarity of stock solution = 0.50M Ka= 6.32 ×10-8 We…
Q: What volumes of 0.100 M acetic acid and 0.100 M sodium acetate would be required to produce 1.00 L…
A:
Q: 2. The weak acid hypochlorous acid (HClO) is formed in bleach solutions. If the pH of a 0.12 M…
A: In multiplart questions we solve only three subquestion according to Bartleby Guidelines. pH of any…
Q: A beaker contains a total concentration of 0.33 molar of weak acetic acid with pKa=4.27 and pH=2.02.…
A:
Q: If 24.8 mL of 0.107 M acid with a pKa of 5.76 is titrated with 0.102 M NaOH solution, what is the pH…
A: pH = pKa + log [salt]/[acid] Number of moles = molarity x volume (L)
Q: The following observations are made about a diprotic acidH2A: (i) A 0.10 M solution of H2A has pH =…
A:
Q: Calculate the pH of buffer system with 0.25 M C6H5NH3 and 0.31 M C6H5NH3+.Ka of anilinium ion = 2.5…
A:
Q: Which acid-conjugate base system should you use to prepare a buffer with a pH of 9.0?…
A: pH = 9.0 According to Henderson hasselbach equation pH of buffer solution can be calculated as- pH…
Q: What is the final pH of a buffer prepared by mixing 450 mL of 0.25 M Tris base with 130 mL of 0.50 M…
A:
Q: 8. An indicator with Ka = 6.45 X 10-5 is yellow in acid and blue in base. Showing a calculation for…
A: In indicator is a substance which is used to detect the equivalence point of an acid base titration…
Q: 120 mg of codeine hydrochloride (CodHCl) is dissolved in 10.0 mL of water. Calculate the expected…
A: Since you have posted multiple completely unrelated questions, we are entitled to answer the first…
Q: The base ephedrine has a pKa value of 9.6. Calculate the theoretical end point pH when a 0.1 M…
A: Ephedrine, the organic molecule used in the prevention of low blood pressure and asthma. The…
Q: Which of the following is the best buffer at neutral pH (pH 7.0)? O Water, pKa = 14 Phosphoric acid,…
A: The question is based on the concept of pH of the solution. it is defined as a negative logarithm…
Q: Which of the following statements is true concerning a 0.50 M hydrozine N2H4 solution of 30.0 ml…
A: When there is no HCl solution, then the solution contain only N2H4 solution. By addition of HCl,…
Q: A hypochlorous acid buffer is prepared with 0.10 moles HCIO and 0.10 moles CIO- and pH = pKa = 7.40.…
A: pH = pKa + log[salt]/[acid] ClO- + HCl -----> HClO + Cl- Number of moles of HClO formed =…
Q: The Ka of bromoacetic acid is 2.00 ×10–3. What masses of bromoacetic acid (CH2BrCOOH) and sodium…
A:
Q: Consider a buffer prepared by mixing 0.100 M propionic acid (CH3CH2COOH, pKa = 4.87) and 0.200 M…
A: Given: Buffer solution of weak acid CH3CH2COOH and its conjugate base salt CH3CH2COONa. And HCl is…
Q: Why is the concentration ratio the same for the calculation of pKa2? "The pKa values of a compound…
A: Electrolytes are the species that dissociate into their ionic components in water and conduct…
Q: A 100 mL sample contains H3PO4 and NaH2PO4. A 10 mL aliquot was taken and methyl orange was added as…
A: The sample contains a mixture of H3PO4 and NaH2PO4.
Q: A physician wishes to prepare a buffer solution at pH = 3.82 that efficiently resists changes in pH…
A: Given, that a physician wishes to prepare a buffer solution at pH=3.82 that efficiently resists…
Q: 1. In Acidic Buffer solution (HNO;J/INO,] when the value of pH - pka +1, the ratio of (HNO,/[NO,] is…
A: You have posted multipart of question as per the guidelines i have solved first three kindly asked…
Q: CH3COOH
A: A) mmoles of Acid = 25mL • 0.2molL-1 = 5.0mmol B) mmoles of Base = 35mL • 0.1molL-1 = 3.5mmol…
Q: Which of the following weak acids when paired with its conjugate base would be the best choice for a…
A: Given group of answer choices : a). HClO: pKa = 7.54 b). C5H4NH+: pKa = 5.25 c). HNO2: pKa =…
Q: Histidine has ionizable groups with pKa values of 1.8, 6.0, and 9.2, as shown below (His = imidazole…
A: The three pKa values shows that there are 3 replaceable hydrogens in the molecule(represented as…
Q: NOH2PO4 and Na2HPO4 For what ratio between the concentrations of the two substances above would the…
A: The given buffer solution is a mixture of weak acid NaH2PO4 and conjugate base Na2HPO4 . The pH of…
Q: Calculate the pH of a buffer prepared by dissolving 0.80 moles acetic acid (CH3COOH) and 0.40 moles…
A: pH of acidic Buffer solution is calculated Handerson equation that is pH = pKa +…
Q: 0.0158 M LiClO and 0.099 M HClO in 2.5 L. The ka of HClO = 2.9e-8.
A: Given data are as follows: LiClO concentration=0.0158 M HClO concentration=0.099 M ka of HClO =…
Q: Harriet Rowki prepared 100.0 mL of 2.50 M H3BO3-NaH2BO3 buffer solution (pH = 8.50). The pKa value…
A: A buffer solution is an aqueous solution consisting of a mixture of a weak acid and its conjugate…
Q: What volumes of 0.50 M HNO2 and 0.50 M NaNO2 must be mixed to prepare 1.00 L of a solution buffered…
A:
Q: pH
A: As you have asked multiple questions, we will solve the first question for you. If you want any…
Q: What is the pH of a buffer solution that contains 0.245 M weak acid and 0.450 M of the conjugate…
A:
Q: In titrating 10 ml of 0.1 N HA (pKa-D4),the pH after the addition of 12 ml C NAOH is O 3.69 O 11.2 O…
A: In this question HA is limiting reagent so NaOH and salt remains in the final solution.
Q: Phosphoric acid is a triprotic acid (?a1=6.9×10^3Ka1=6.9×10^3, ?a2=6.2×10^8 Ka2=6.2×10−8, and…
A: Buffer solutions are those solutions which resist the change in pH upon addition of small amount of…
Q: Suppose a biochemist has 10 mL of a 1.0 M solution of a compound with two ionizable groups at a pH…
A: Given in following question
Q: How many moles of sodium acetate must be added to 500 mL of 0.25 M acetic acid solution to produce a…
A:
Q: Given the titration curve of the hypothetical polyprotic acid X at 0.100 M concentration (pKa1=4.0,…
A: A polyprotic acid donates a proton as an Arrhenius acid donates a proton. However, a polyprotic acid…
Q: 14. Acetic acid has a Ka of 1.8 x 10-5. What is the pH of a buffer solution containing 0.15M HC2H3O2…
A: This an acidic buffer of the acetic acid and acetate ion. The pH of the acidic buffer can be…
Q: A primary amine R-NH2 has pKb = 5.65 at 25°C. A buffer solution is made by adding 66.0 mL of 1.55…
A:
Q: What weight of Na2HPO4 and H2PO4 would be required to prepare 200mL of a buffer solution of pH 7.40…
A: Given data, Na2HPO4 and H2PO4 prepared 200mL of a buffer solution of pH 7.40 Ionic strength =…
Q: A 10.00 mL sample of 1.20 M HNO2 (analyte) (pKa = 3.34 and Ka = 4.57x10-4) is titrated with 1.20 M…
A: Given, A 10.00 mL sample of 1.20 M HNO2 (analyte) (pKa = 3.34 and Ka = 4.57x10-4) is titrated with…
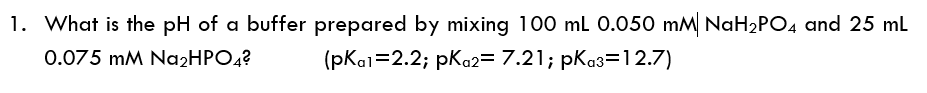
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

- What will be the appropriate pH of a buffer prepared by mixing 100 ml of 0.1 M Na2HPO4 and 100 ml of 0.1 M NaH2PO4? (For H3PO4, pKa1 = 2.1; pKa2 = 6.8; pKa3 = 12.5)How many mL of 0.600 M NaOH should be added to 5.38 g of carbonic acid (H2CO3, FW = 60.02) (pKa1 = 6.351, pKa1 =10.329) to give a buffer with pH = 9.50 when diluted to 250 mL?Calculate the pH of a buffer in which [HC4H7O2] = 0.36 M and [C4H7O2 –] = 0.31 M. How do you calculate pKa acid dissociation constant??
- A student needs to prepare a buffer solution with a pH of 4.80. Assuming a pKa of 4.73, how many mL of 0.1 M N a C2H3O2 would need to be added to 21.0 mL of 0.1 M HC2H3O2 to prepare this buffer?the 1.00 L buffer solution is 0.150 M in HC7H5O2 and .250 M in LiC7H5O2. calculate the pH of the solution after the addition of 100.0 mL of 1.00 M HCl the Ka for HC7H5O2 is 6.5 x 10^-5What will be the appropriate pH of a buffer prepared by mixing 100 ml of 0.1 M Na2HPO4 and 100 ml of 0.1 M NaH2PO4? (For H3PO4, pKa1 = 2.1; pKa2 = 6.8; pKa3 = 12.5) Ans in 2 significant figures
- What is the pH of a buffer that results when 5.46 mole of H3PO4 is mixed with 2.97 mole of NaOH and diluted with water to 1.00 L? (The acid dissociation constants of phosphoric acid are Ka1 = 7.5 × 10–3, Ka2 = 6.2 × 10–8, and Ka3 = 3.6 × 10–13)A 0.5131-g sample that contains KBr (MM: 119.0023) is dissolved in 50 mL of distilled water. Titrating with 0.04614 M AgNO3 requires 25.13 mL to reach the Mohr end point. A blank titration requires 0.65 mL to reach the same end point. Report the %w/w KBr in the sample.Can someone please explain why and how the step 1 was done? What was its purpose? Why was only the pKa2 value was used in the second step 2? I don't understand shortcuts and I would appreciate it if it's explained step by step. This was the solution to my previous question in which I asked: What is the pH of a buffer prepared by mixing 100 mL 0.050 mM NaH2PO4 and 25 mL 0.075 mM Na2HPO4? (pKa1=2.2; pKa2= 7.21; pKa3=12.7)
- Consider a buffer prepared by mixing 0.100 M propionic acid (CH3CH2COOH, pKa = 4.87) and 0.200 M sodium propionate (NaCH3CH2COO). Which species will most likely react upon addition of aqueous HCl solution? CH3CH2COOH CH3CH2COO– H2O Na+A buffer is created by adding 0.321 moles of bromoacetic acid (pKa = 2.902) and 0.555 moles of sodium bromoacetate to 1.00 L of solution.a) What is the pH of this solution?Iminodiacetic acid (IDA) is often used in ion exchange resins as a way to remove heavy metals from industrial waste waters. It forms a 2:1 complex with many heavy metals, with a high selectivity for divalent over monovalent ions. A 14.50 mL sample of 0.0315 M Ni2+ was added to 14.50 mL of 0.187 M iminodiacetic acid (the completely protonated form is abbreviated as H3A+) buffered at pH 7.00. Given that ?A2− is 4.6×10−3 at pH 7.00, what is the final concentration of Ni2+ remaining in solution? Ni2+(aq)+2A2−(aq)↽−−⇀NiA2−2(aq)?f=2.00×1014 [Ni2+]= __________ M

