Q: AICI3 Zn(Hg) Compound A Compound Me Me. Me HCI, A Me
A: Benzene derivatives can give aromatic electrophilic substitution reaction.
Q: What is the correct Ko expression for the following reaction? N2 (g) + 3 H2 (g) = 2 NH3 (g)
A: Given :- N2(g) + 3H2(g) <----> 2NH3(g) To write :- Its correct Kc expression
Q: CHEM 302 Name HO.
A: 1HNMR spectrum identified number of chemically non-equivalent protons present in molecule. Splitting...
Q: Balance this redox reaction in a basic solution, showing the reduction and the oxidation half reacti...
A: Oxidation is the loss of electrons. Reduction is the gain of electrons. In the oxidation, the oxid...
Q: 3. Circle the alkene that is most electron rich. A В Ме Ме Me Me Me -Me Me Ме
A: The alkene having more electron density is electron rich alkene.
Q: A chemistry graduate student is studying the rate of this reaction! 2HI (g) -H, () + 1, (g) He fills...
A: The given reaction is 2HI (g) ---> H2(g) + I2(g) Rate law expression states that rate of a react...
Q: What other methods can be used to further extract lipids from coconut aside from hot extraction? Wha...
A: Answer - According to the question - Coconut oil is traditionally extracted by boiling coconut milk ...
Q: t temperature doe
A:
Q: A semiconductor has wide energy gap and requires less energy to move an electron from the valence ba...
A:
Q: What is the molecular formula of the compound?
A: To solve this problem we have to first write the Condensed Structural Formula of the given compound ...
Q: Write the concentration equilibrium constant expression 2+ 2 Cu (aq)+41 (aq)-2 Cul(s)+I,(aq)
A: To solve this problem we have to write the equilibrium constant expression .
Q: What is the Main Content uffer that is 0.452 M HF and 0.404 M LiF? The K, for HF is 3.5 x 10-4. Your...
A: Buffer solution :- A solution which resists the change in its pH value by the addition of small amou...
Q: Which of the following structures is NOT a contributing resonance structure A that explains the regi...
A:
Q: A 3.40 gram mixture of potassium chlorate KCLO3, and potassium chloride, KCL, was heated until all o...
A: Given, KClO3(s) ➝ KCl(s) + O2(g) Balancing the above equation, 2KClO3(s) ➝ 2KCl(s) + 3O2(g) Pressure...
Q: Solutions that are electrolytes have ions in solution. These ions are surrounded by Tayers of water ...
A: In this question, we have to identify the ions from the compounds that will occur in the solution.
Q: What is the ΔrG° for the following reaction (in kJ mol-1)? N2 (g) + O2 (g) + Cl2 (g) ⇌ 2 NOCl (g) To...
A:
Q: Calculate the thermal conductivity of Argon at 300 K and 15 Mbar. Gas is confined in a cubic vessel ...
A:
Q: 1. Circle the substrate that will not result in a tertiary alkyl chloride when treated with HCI. A В...
A: In the given all three reactions, H —Cl is added.
Q: 10.1 Draw the major product(s) each reaction below. ROOR HBr hv or A ROOR hv or A b. HBr Li. NH,
A:
Q: Which arrow-pushing mechanism best explains activation of acetyl chloride for Friedel-Crafts acylati...
A: The arrow pushing mechanism that best explains the activation of acetyl chloride for friedal crafts ...
Q: 2. Balance the equation below for the combustion of propane in a camping stove. Then answer question...
A:
Q: How much heat is gained by the water? B. How much heat heat is lost by the zinc metal? C. From the d...
A: To solve this problem we will use the formula q = m • C •∆T Where , q = heat released or absorbed...
Q: Part A What is the OH concentration in an aqueous solution at 25°C in which [H30*]= 3.8 x 10 M? O 3....
A:
Q: Part A The surface area of an object to be gold plated is 49.9 cm? and the density of gold is 19.3 g...
A: Given: The surface area of the area to be gold plated = 49.9 cm3 The thickness of the gold layer = 1...
Q: 云。 LL CHEM 1510, MWF 12:00 - 12:50 p.m., Spring 2022 mistry Assignments <Homework: Ch. 15 Chapter 15...
A: Given:-
Q: ns ΔΗ and ΔS.
A:
Q: What is the pH of a 0.419 M C6H5CO2H M solution if the Ka of C6H5CO2H is 6.5 *10^-
A: pH of a solution is related to the concentration of H+ ions in the solution and is given by the foll...
Q: HO
A:
Q: If 250.grams of sodium azide undergoes decomposition, how many grams of sodium are produced?
A:
Q: consider the following exothermic reaction at 0.00 °C and 1.00 atm Se04 (g) Se(g) + O2 (g) , Kc = 2....
A:
Q: If the filter paper is weighed before instead of after being placed in the 105°C oven for an hour, w...
A: Limiting reagents are those reactant which is present in lesser amount and is consumed first, This ...
Q: Calculate the pH of a mixture that contains 0.21 M of HCOOH and 0.12 M of HCIO. The K, of HCOOH is 1...
A:
Q: * 00 Ili Σ | A 0.00100 mol sample of Ca(OH) requires 20.00 mL of aqueous HCI for 2. neutralization a...
A:
Q: MISSED THIS? Watch KCV 16.3, IWE 16.1; Read Section 16.3. You can click on the Review link to access...
A: Concept: Conjugate acid base: According to the Brønsted–Lowry definition of acids and bases, the co...
Q: 4. Calculate the change in pH that results from adding 25 mL of 0.010F HCI to 500ml buffer solution ...
A: Given, Volume of the buffer in mL = 500 mL [NH3] = 0.25 M [NH4Cl] = 0.15 M [HCl] = 0.010 F or 0....
Q: Identify the triprotic acid. H2CO3 O HFO4 O HNO3 O H2SO4 O H3PO4
A:
Q: In a redox reaction, electrons are transferred O from the substance being oxidized to the reducing a...
A:
Q: Which reaction schemes is best to give the product? HO. NO 2 O The following reaction scheme: CH3 KM...
A:
Q: Substitution with a(n) to an aromatic ring would result in reaction? Deactivator group, fast Activat...
A: Activator - electron donating group and para and ortho directing Example - OH, OCH3 Deactivator - ...
Q: What would be the ideal buffer range for an acid if its Ka = 1.6 x 10-5?
A:
Q: 1.Which acid is strongest? a. H3AsO3, Ka=5.1x10-10 b. HCN, Ka=4.9x10-10 c. H3BO3, Ka=5.8x10-10 d. C...
A:
Q: How long must constant current of 50.0 A be passed through an electrolytic cell containing aqueous C...
A: According to the first law of Faraday, the mass of a metal deposited on the anode during electrolysi...
Q: What minimum mass of copper (II) nitrate must be added to 20.0 mL of a 0.0387 M phosphate solution i...
A: Volume of phosphate solution = 20.0 ml Molarity of phosphate solution = 0.0387 M Molar mass of coppe...
Q: What is the change in Gibb's Free Energy for a reaction that has an enthalpy change of -270.8 kJ/mol...
A: The change in enthalpy is equal to the energy supplied as heat at constant pressure. The equation ΔG...
Q: Part A Identify the Brønsted-Lowry acid, the Brønsted-Lowry base, the conjugate acid, and the conjug...
A: We need to identify the Bronsted Lowry acid, base and the conjugate acid and base for the given reac...
Q: 9. The evaporation of water is endothermic: H2O (1) → H2O (g) AH°rxn = 44.01 kJ/mol What minimum mas...
A: H°rxn = 44.01 kJ / mol
Q: Explain the compounds/ions present in solution when: 0.050M HNO3 is mixed with 250mL of water 0.050...
A: HNO3(aq) is a strong acid, whereas NH3(aq) is a weak base.
Q: (Q12) Hydrazoic acid (HN3) is a weak acid with a Ką of 2.5 x 10³. What will be the equilibrium pH of...
A: We need to calculate the pH of hydrazoic acid at equilibrium
Q: O number of molecules.
A: Answer: equal volume propane and carbon monoxide at the same temperature and pressure have the numbe...
Q: Mg + Fe + Mgo Type of reaction: + 02- O'H Type of reaction: <--- 'Osqd PBSO3 + O2 Type of reaction:
A: Note : As per our company guidelines we are supposed to answer only first 3 sub-parts. Kindly repost...
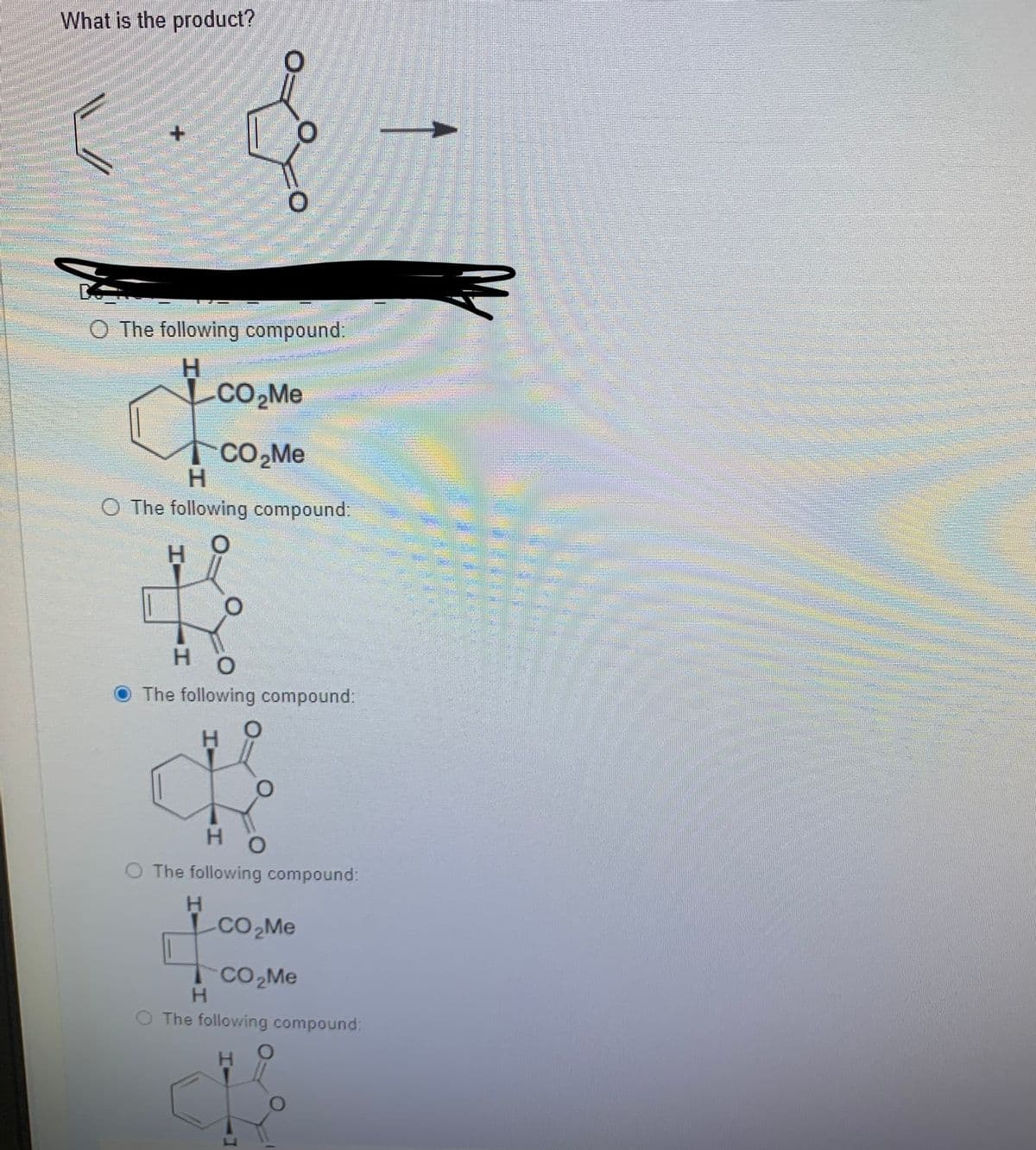
Step by step
Solved in 2 steps with 2 images

- For 20.7 the solutions for part c and d dont seem right. Shouldn't reacting with 1 eq of H2 just take away the C=C bond and not the C=O bond. (H2 reduces C=C selectively to form a ketone according to page 738. Also the both the C=C and C=O bond should be reduced if excess H2 is usedA mixture of 0.10 mol benzene and 0.10 mol p-xylene was allowed to react with 0.10 mol nitronium ion until all the nitronium ion was gone. Two products were obtained: 0.002 mol of one and 0.098 mol of the other. a. What was the major product? b. Why was more of one product obtained than of the other?How to get the wt. sample, g.
- Tunicates are marine animals that are called "sea squirts" because when they are taken out of water, they tend to contract and expel seawater. Lepadiformine is a cytotoxic agent (toxic to cells) isolated from a marine tunicate. During a recent synthesis of lepadiformine, the investigators observed the formation of an interesting by-product (3) while treating diol 1 with a reagent similar in function to PBr3 (J. Org. Chem. 2012, 77, 3390–3400):Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(CzHs)3). 3SnCl4 + 4Al(C2H5)3 → 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d= 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?What are the pKa’sof HCN (hydrocyanic acid), and HF (hydrofluoric acid)? Based only on those pKavalues, what do you expect to be the products of the reaction below? Briefly explain. ???+??→?
- The molecular formula of unknown compound B is C10H16.Quantitative brominationof a 0.473-g sample of compound Brequired 48.22mL of a 0.216MBr2/CCl4to produce a color change. How many rings and pi bonds does compound Bcontain?Include any explanations/calculations to justify your answers.when H2O2 is added into acidified K2Cr2O7 which colour appears?Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?
- The following IR and NMR were obtained for Alicia’s final product, Bicyclo[2.2.1]hept-5-ene-2,3-dicarboxylic acid. Label all hydrogens with the data from 1H NMR. Label all carbons with the provided data from 13C NMR.give the reagents for parts a-pChemistry Give the products of the reaction of 1 mole of 2-methy1-1,3-pentadiene with 1 mole of HBr. Whichproduct(s) will predominate if the reaction is under kinetic control? Which products) will predominateif the reaction is under thermodynamic control?





