Q: Using enthalpies of formation, calculate the standard change in enthalpy for the thermite reaction:…
A:
Q: You will make Nylon-6,10. What does the 6 and 10 refer to? PROCEDURE Synthesis of Nylon In a small…
A: Given that, the preparation of Nylon-6,10. We have to tell what the numbers 6 and 10 refer.
Q: Calculate the pH at 25 °C of a 0.64M solution of pyridinium chloride (C;H;NHCI). Note that pyridine…
A: The value of pKb for the given weak base is = 8.77 The concentration of the base solution is = 0.64…
Q: Yeast is used in baking the morning bread of Filipinos known as "pandesal". Why?
A: Yeast is used for the leaving of bread. Yeast uses the sugars and oxygen dough to produce more yeast…
Q: What is the energy (in kJ) of one mole of photons associated with radiation that has a frequency of…
A:
Q: C. %23 The wavelength and frequency of an electromagnetic wave are related to each other through the…
A: In a given question , we have to find out the frequency of given wavelength.
Q: Incorrect Is the final answer in percent imultiplied by 100 N Did you and molar mass? Calculate the…
A: Molar mass is sum of atomic weight of all the atom in molecules. We have to calculate percentage of…
Q: For which of the mixtures will Ag2SO4(s) precipitate? 150.0 mL of 0.10 M NazSO4(aq) and 5.0 mL of…
A:
Q: Which one is larger? K or K+
A: K is the symbol of potassium Atomic no. of K = 19
Q: Which of the following statements is false? O The absolute entropy of a perfect crystal at absolute…
A: Here, we have to find the statement from the given statements that is false.
Q: Ignore inorganic byproducts and the phthalhydrazide side-product. :N: K Br of +
A: ->phthalimide used for the formation of amine.
Q: HCN is a weak acid (K, = 6.20 × 10-10), so the salt, KCN, acts as a weak base. What is the pH of a…
A: Given-> Concentration of KCN = 0.0795 M Ka(HCN) = 6.20 × 10-10
Q: When 1 mole of methane (CH4) is burned at constant pressure, 890 kJ of energy is released as heat.…
A: Given, When 1 mole of methane (CH4) is burned at constant pressure, 890 kJ of energy is released as…
Q: Br 2. a) Name this compound according to the IUPAC system: CH3-C-C=C-CH-CH3 CH3 b) Write in the…
A:
Q: 9. Human lungs can absorb 8 gm 02 per hour by respiration. If all oxygen atoms are converted to…
A: 1 mole = 6.022×1023 [Avogadro's number] The mass of 1 mole of atoms or molecules is known as molar…
Q: The boiling point of ethanol is 173.1 F. In an experiment the entropy change for the reaction…
A:
Q: Problems How many Ma moleaules 1. .are in 62.5g of magnesium? 2. Calculate the mass of 1.52 moles of…
A:
Q: A Br FeBr3 2 -Li then H30* D LIAIH4 then H3O* Propose reaction conditions (A) and draw the products…
A: The reaction schema given is,
Q: For each compound: i. Calculate the mass of the molecular ion. ii. Draw the most likely…
A: As per rule i should have to answer only three subparts so please ask remaining questions in another…
Q: An 1855 grams sample of water, initially at 28.6°C, was heated under a pressure of 760 Torr to…
A: Answer: It is a three step process in which during 1. water will be heated to it boiling point 2.…
Q: Name: s,
A: There are 2 ways of naming any organic compound- 1- Common Name 2- IUPAC name For multipart…
Q: Suppose a 500. mL flask is filled with 1.2 mol of CO, 2.0 mol of H,O and 0.90 mol of CO,. The…
A: Given: The volume of the flask is 500 mL The number of moles of CO is 1.2 mol The number of moles of…
Q: Muscles produce lactic acid, CH, CH(OH)COOH(aq), during exercise. Calculate the percent ionization…
A: Given that, the concentration of lactic acid is C = 0.1046 M. The dissociation constant of lactic…
Q: What volume of 0.333 M Al(NO3)3 is needed to obtain 26.7 g of Al(NO3)3?
A: Given:: Molarity of Al(NO3)3=0.333 M mass of Al(NO3)3= 26.7 g molar mass of Al(NO3)3= 213 g mol-1…
Q: In the following reaction, which only goes from left to right, which species is being oxidized?…
A: In the following reaction, which only goes from left to right, which species is being oxidized? 2K…
Q: Complete the following table, which lists information about the measured acid dissociation constants…
A: The acidic stregth of an acid is determined by its acid dissocation constant. The acid dissociation…
Q: In nitrous acid (HNO2) the oxidation state of N is.. 1. +3 2. -3 3. +5 4. +6 а. 1 O b. 2 С. 3 O d. 4
A: The oxidation state of N in nitrous acid HNO2 = ?
Q: Decide what kind of crystalline solid each element or compound in the table forms, and what kind of…
A:
Q: is lactaid real or placebo? love you thanks
A: Lactaid does not work ,it is a placebo.
Q: 6. What are the possible values of the angular momentum quantum number (€) when n= 3 ? 7. What are…
A: There are four types of quantum numbers :- Principle quantum number Angular momentum quantum…
Q: In a reaction Al become Al+3 , it shows that.. 1. Al gained 3 electrons 2. Al lost 3 electrons 3. Al…
A: Electrochemistry is branch of chemistry in which we deal with redox reaction. Oxidation means loss…
Q: 5. Another unit that is used quite frequently in microwave spectroscopy is GHz (Gigahertz, 1x 10°…
A:
Q: Which one is an example of a standard solution? 1. Sugar solution 2. Basic solution 3. Acidic…
A: A homogeneous mixture of two or more substances is known as a solution. The species which present in…
Q: 1.Explain what is anomeric effect and why it happens?
A: As there are two questions ,I am solving the first question.
Q: At what substrate concentration would an enzyme with a kcat of 30.0 s-1 and a Km of 3. (а) 0.0050 M…
A:
Q: Draw the major product of this reaction. Ignore inorganic byproducts. Br2 CH3SH Drawing
A:
Q: Calculate the pH at 25 °C of a 0.44M solution of potassium butanoate (KC,H,CO,). Note that butanoic…
A: Given- Concentration of potassium butanoate= 0.44M pKa of weak acid=4.82 Temperature =…
Q: Ammonium perchlorate (NH Clo,) is the solid rocket fuel used by the U.S. Space Shuttle. It reacts…
A: Given -> Weight of ammonium perchlorate = 5.2 gm
Q: Consider a 25.0 mL buffer solution containing 0.100 M sodium alderate and 0.126 M alderic acid.…
A:
Q: Which of the following is not an allowed quantum state (n,l,mi,ms) a)5,4,-3,-1/2 B)3,2,2,1/2…
A: Which of the following is not an allowed quantum state (n,l,mi,ms)----->
Q: How many ml of water do you add to 10ml of glycerol stock if you want to dilute it by a factor of a)…
A: We have to calculate volume of water need to be added to 10 mL of glycerol stock, for the given…
Q: Consider a 0.15M solution of ascorbic acid (vitamin C). It has a K1 of 8.0 x 10 and a K2 of 1.6 x…
A:
Q: PPh3, DIAD CH2OH THF (A) mitsunobu H2N seaction NH2 excess AgOH (B) (C13H20 INS) me I me me
A: Since you have asked multiple questions we will answer the first one for you. To get the remaining…
Q: 5. If temperature is constant, the relationship between pressure and volume is C. equal D. unequal…
A: According to Boyle's law the relation between pressure and volume can be explained .
Q: Which of the compounds is more soluble in an acidic solution than in pure water? AIPO4 O CSCIO4 Cas…
A:
Q: Q2. This question is about the periodicity of the Period 3 elements. (a) State and explain the…
A: Answer: Energy required to release the most loosely binded electron in an isolated gaseous atom is…
Q: 1. Which one of the following is an example of week electrolyte? 1. NaOH 2. Naci 3. CH3COOH 4.…
A: here we are required to classify the given compounds as weak and strong electrolyte
Q: How can you use 1H NMR to determine if you synthesized a cis or trans alkene? O Look at the chemical…
A: NMR is branch of chemistry in which we deal with shielding and deshielding effect on protons by…
Q: 3. A solution containing 10 g of NH3 at 90°C can be described as- ib e tuloa insoluble…
A:
Q: The molecule shown is a lipid. How would you classified it? H3 C-(CH2 ) 28 CH2 -Ô-C-(CH2 )16 CH3
A: Lipids are macromolecule made up of fatty acids. Lipid is any organic compounds that are insoluble…
4
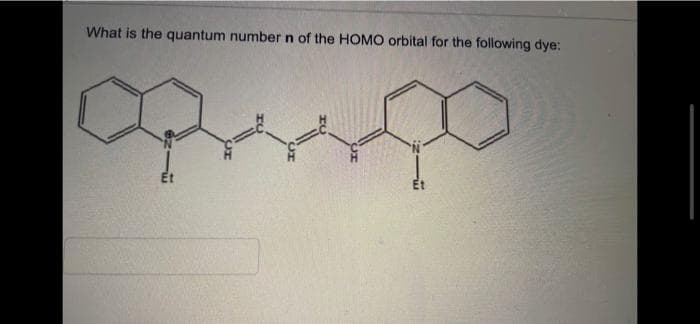
Step by step
Solved in 2 steps

- The photoelectron spectrum of HBr has two main groups of peaks. The first has ionization energy 11.88 eV. The next peak has ionization energy 15.2 eV, and it is followed by a long progression of peaks with higher ionization energies. Identify the molecular orbitals corresponding to these two groups of peaks.Which of the following represents the HOMO in carbon monoxide (CO)? Why is the answer sigma star (antibonding)? I thought the HOMO was the highest occupied electron orbital, so why would the 2p pi level be wrong?The molecule PtCl 2 - is an important square planar starting material in synthetic inorganic chemistry. Using the p orbitals of Cl as the basis find the irreducible representations of the p orbitals and indicate which orbitals could possibly overlap with any of the orbitals on Pt . Assume that the pz orbitals on Cl are orientated in the same direction as the principle rotation axis, px orbitals are pointed at the Pt , and the py orbitals are orthogonal to the px orbitals. Show the characters of each orbital and identify which orbitals on Pt would match from a pure symmetry standpoint.
- How do you write the Huckel determinant for the pi electrons of butadiene? How would you find the energies of the molecular orbitals?What is the ground-state configuration of the valence electrons of brominemolecules (Br2)? Is bromine paramagnetic or diamagnetic?Which of the following molecules is an example of a C∞v point group? A. HCN B. CH2Cl2 C. CCl4 D. BCl3 E. NH3
- Draw the structure of PCL5 calculate their hybridization and also determine the shapes and geometriesThe FEMO theory of conjugated molecules is crude and marginally better results are obtained with simple Huckel theory. (a) For a linear conjugated polyene with each of N carbon atoms contributing an electron in a 2porbital, the energies Ek of the resulting π molecular orbitals are given by: Ek = α + 2β cos kπ/N+1 k = 1,2,3, ... N Use this expression to estimate the resonance integral β for the series consisting of ethene, butadiene, hexatriene, and octatetraene given that ult raviolet absorptions from the HOMO, which is a bonding π orbital, to the LUMO, which is an antibonding π* orbital, occur at 61 500, 46 080, 39 750,and 32 900 cm- 1, respectively. (b) Calculate the π-electron delocalization energy, Edelcc = Eπ -n(α + β). of octatetraene, where Eπ, is the total π-electron binding energy and n is the total number of π-electrons.Explain the concept of bonding and antibonding molecular orbitals and draw examples of s and p MOs?

