Q: 20.5 Provide reasonable mechanisms for the following transformations. b. NH₂ BrMg MgBr HS -NH H*,…
A:
Q: Macmillan Learning Use the van der Waals equation of state to calculate the pressure of 2.80 mol of…
A: The van der Waals equation of state for real gases is given by, P+an2V2V-nb=nRTwhere,P=Pressure…
Q: a. Manganese(II) nitrate and sodium carbonate. 02Mn²+ (aq) + CO32 (aq) → Mn2CO3 (8) 2+ Mn²+ (aq) +…
A: The question is based on the concept of chemical reactions. we need to write net ionic equation for…
Q: H2SO4(aq) + 2 KOH(aq) → K2SO4(Blank 1) + 2 H2O(Blank 2) Fill in the blanks above with the…
A: The given balanced chemical equation for the neutralization of acid and base is H2SO4(aq) + 2…
Q: 9. What is the starting material in the following reaction? A. I B. II || C. III D. IV LDA CH₂l Br…
A:
Q: Open bodies of water can act as carbon sinks due to the ability of carbon dioxide to dissolve in…
A: Please find your solution below :
Q: How much heat (in kilojoules) is evolved or absorbed in the reaction of 2.90 gg of Fe2O3Fe2O3 with…
A:
Q: If we start the reaction below with 16.78 g of Ag2S (molar mass of 247.80 g/mol) and 6.780 g HCI…
A: To solve this problem we have to use the given balance chemical equation.
Q: The equilibrium constant, Ka, for a monopratic acid (propionic acid) is 1.3 ×10^-5. Which of the…
A:
Q: What is the concentration of H3O+ in a buffered solution made by adding 0.30 mol of acetic acid and…
A:
Q: 13. Please check to see if the following are balanced. If they are not, please balance them. a. K b.…
A: Q 13 balancing reaction will be as follows: a) 2K + Br2 → 2 KBr…
Q: Calculate the value of the free energy change, ΔG, for the reaction below at 225.0ºC when the…
A: Question is based on the concept of chemical thermodynamics we need to calculate Gibbs free energy…
Q: Use the following vapor pressure data to answer the questions: A B Liquid CH3COOC₂H5 C2H5NH2 Vapor…
A: Given that, We have to answer the following questions
Q: What is the hydroxide ion concentration of a solution prepared by dissolving 0.658 g of Mg(OH)2 in…
A: Given that - Mass of magnesium hydroxide Mg(OH)2 = 0.658 g Volume of solution (of water) = 333 mL…
Q: Fill in the following blanks. As the dye concentration increases, the absorbance of the light…
A: The relation between the concentration and absorbance of a sample can be explained with the help of…
Q: Br₂ hv A t-BuOK t-BuOH heat B HBr peroxides C NaSH D
A:
Q: 2. When the following pair of reactants are combined in the presence of a basic catalyst, a number…
A: in presence of base, carbonyl compound having enolisable alpha hydrogens, undergo aldol condensation…
Q: draw the structures of the geometric and optical isomers of the compound [Co(gly)3
A: We have been given an inorganic compound [Co(gly)3].We have been asked to draw the structures of…
Q: The oxidation of aqueous iodide by arsenic acid to give 13 and arsenous acid proceeds via the…
A: We need to write the rate expression for (a) decrease of initial rate for I3-, Δ[I3-]/Δt (b) [H+],…
Q: What is the K₂ reaction of HCN? Ka reaction: The K₂ of HCN is 6.2 x 10-10. What is the K, value for…
A: It is based on the concept of acid and base Here we are required to find the expression of acid…
Q: Consider the buffer made by preparing .650 moles of a weak acid HA with .725 of a weak base NA in…
A: Buffer is a solution which resists change in pH on adding small amounts of acid and base. we need to…
Q: Please do not round off intermediate calculations. Thank you.
A:
Q: Which of the following would successfully perform the shown reaction? (A) (B) (C) OH 1) NaH 2) EtBr…
A: Organic reactions are those in which organic reactant react to form organic products. In the given…
Q: Natural goldenrod pigment is a yellow dye that changes to a bright red color in the presence of a…
A: Given Natural goldenrod pigment is a yellow dye that changes to a bright red color in the presence…
Q: VSEPR theory can be used to predict the shapes that simple molecules form. First, explain the…
A: We have to determine the shape of the molecules using VSEPR theory.
Q: What is the pH of a 0.320 M solution of Ca(NO2)2 (Ka of HNO2 is 4.5 x 10-4)?
A:
Q: Be sure to specify states such as (aq) or (s). If a box is not needed leave it blank. Write the net…
A:
Q: Draw the major organic product for the reaction shown. Br 1. PPh3 2. n-butyllithium 3. Select Draw…
A: The carbonyl compounds on treatment with triphenyl phosphonium ylide forms alkene. This reaction is…
Q: Write the balanced net ionic equation for the precipitation of silver(I) sulfide from aqueous…
A: net ionic equation is a chemical equation where only those ions undergoing chemical changes during…
Q: onsider the reaction: 2 NH3(g) N₂(g) + 3H₂(g). At T = 740 K, the equilibrium constant Kc for this…
A: Kc is equilibrium constant and it is equal to concentration of product divide by concentration of…
Q: Which of the following statements most correctly describe(s) any chemical equilibrium? (A) At…
A: Given that, We have to select the most correct option for the any chemical equilibrium.
Q: What happens when HCI is added to a buffer composed of CH3COOH and CH3COO-? (A) There is no reaction…
A: The buffer is generally used to avoid the change of pH in a solution. The given buffer solution is a…
Q: 11 a b C Consider the hypothetical thermochemical equation 3A+B → 2C for which AH = 65.5 kJ/mol. X L…
A: Since you asked multiple questions so as per Q&A guidelines of portal I solved first three parts…
Q: Gaseous ethane (CH3 CH3) will react with gaseous oxygen (O₂) to produce gaseous carbon dioxide (CO₂)…
A:
Q: Find Molecular Equation, Ionic Equation, and Net Ionic Equation of Ammonium Carbonate + Phosphoric…
A: We have been given that Ammonium Carbonate + Phosphoric acid.We have been asked to find Molecular…
Q: Some chemical compounds are listed in the first column of the table below. Each compound is soluble…
A: The compounds which are formed by the complete transfer of electrons from one species to other are…
Q: Light fuel oil with an average chemical composition of C10H18 is burned with oxygen in a bomb…
A: This problem is based on Thermodynamics. The solution is given below.
Q: A sample of carbon dioxide is analyzed and determined to be 27.3% by mass carbon. If a 50.0 g sample…
A: Consider a compound AB Percent of A = (Mass of A/Mass of AB) x 100 Law of conservation of mass:…
Q: 6. As + NaOH →→ 5Na3ASO3 + 2H₂ Reactant Product Na = 15 As = 5 O= H = 4
A: Since, Balanced reaction means that both side number of atom present in equal number. Thus,
Q: 6. Write the simplest formula (ratio) for 0.080 mol Ba, 0.080 mol S, 0.320 mol O. (Hint: calculate…
A: The given compound contains 0.080 mol Ba, 0.080 mol S, 0.320 mol O. The simplest formula of the…
Q: 3. Predict the major organic product(s) and provide an arrow-pushing mechanism. Clearly identify the…
A: The question is based on organic reactions. we need to identify the product formed and explain…
Q: Balance the following chemical equation. If the chemical species needs a "1" in the blank to balance…
A: The given unbalanced chemical equations are Blank 1 HCl (aq) + Blank 2 Al(OH)3 (s) →Blank 3 AlCl3…
Q: Br 1) HNO3, H₂SO4 2) Zn, HCI ?
A:
Q: Classify the unbalanced reactions below. You will have to select the best answer if more than one…
A: Precipitation reaction: The reaction in which an insoluble solid is obtained as one of the product.…
Q: Consider the decomposition of lead(IV) oxide: PbO₂ (s) PbO(s) + O₂(g) At a certain temperature the…
A: Pressure exerted by individual gases in mixture of gases is known as partial pressure of that gases.
Q: What is the correct equilibrium constant expression for the following reaction? NH3(aq) + H₂O(/)…
A:
Q: Br 1. Nal, acetone 2. H₂O, 20 °C
A:
Q: Zinc reacts with HCI to form H2 gas. If you want to make 0.234 moles of H2 gas and have excess zinc,…
A: Balanced reaction Zn(s) + 2HCl(aq) ---> ZnCl2 (aq) + H2 (g) Moles of H2 produced = 0.234 moles…
Q: Use the following data to place the fictitious metals Q,R,T,X,A and E into an activity series
A: To solve this problem we have to arrange the given elements in activity series.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

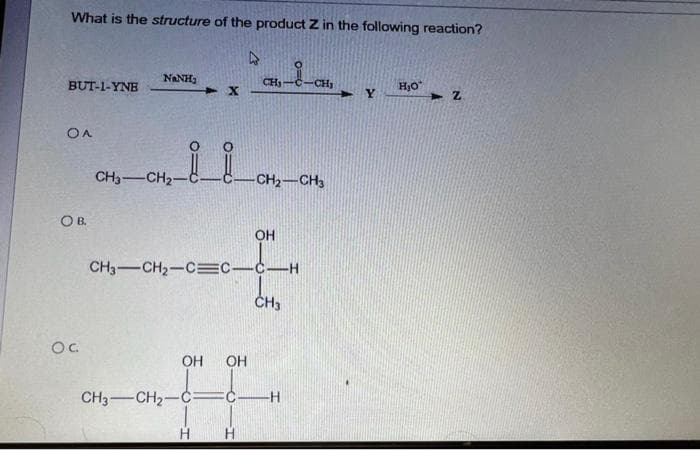
- Bicyclo[2.2.1]heptan-7-one + PCC (in CH₂Cl₂) => A.) Bicyclo[2.2.1]heptan-7-ol B.) Bicyclo[2.2.1]heptanoic acid C.) Cyclohexanecarbaldehyde D.) All the given choices are possible products E.) No reaction32. Which of the following is INCORRECT? A. CH3COOH H+ + CH3COO- B. HF H+ + F- C. H2O H+ + O2- D. C2HO4- H+ + C2O42-A. OsO4 and NMO B. Br2 and H20 C. Hg(OAc)2, H2O and NaBH4, NaOH D. RCO3H E. BH3-THF and H2O2, NaOH Which reagent will complete this reaction?
- 2. How many substitution product/s is/are formed when metabromo anisole is treated with ammonia?A. 0-no reactionB. 1C. 2D. 3What products are formed when each of the following reacts with HO-? a. CH3OH b. +NH4 c. CH3N+H3 d. BF3 e. +CH3 g. AlCl3 d. BF3 f. FeBr3 h. CH3COOHWhat product is formed when CH3OCH2CH2C ≡ CCH2CH(CH3)2 is treated with each reagent: (a) H2 (excess), Pd-C; (b) H2(1 equiv), Lindlar catalyst; (c) H2 (excess), Lindlar catalyst; (d) Na, NH3?
- a.What product(s) (excluding stereoisomers) are formed when Y is heated with Cl2? b.What product(s) (excluding stereoisomers) are formed when Y is heated with Br2? c.What steps are needed to convert Y to the alkene Z?d. Reaction with CH3MgI (excess), ether; then H+/H2O e. Reaction with LiAlH4, ether; then H+/H2O f. Reaction with DIBAL (diisobutylaluminum hydride), toluene, low temperature; then H+/H2ON(CH2CH3)3 + HNO3 --------> a.) rewrite the reaction using bond-line structure of reagents and products of the reaction b.) supply the curved arrows explaining the mechanism of the reaction

