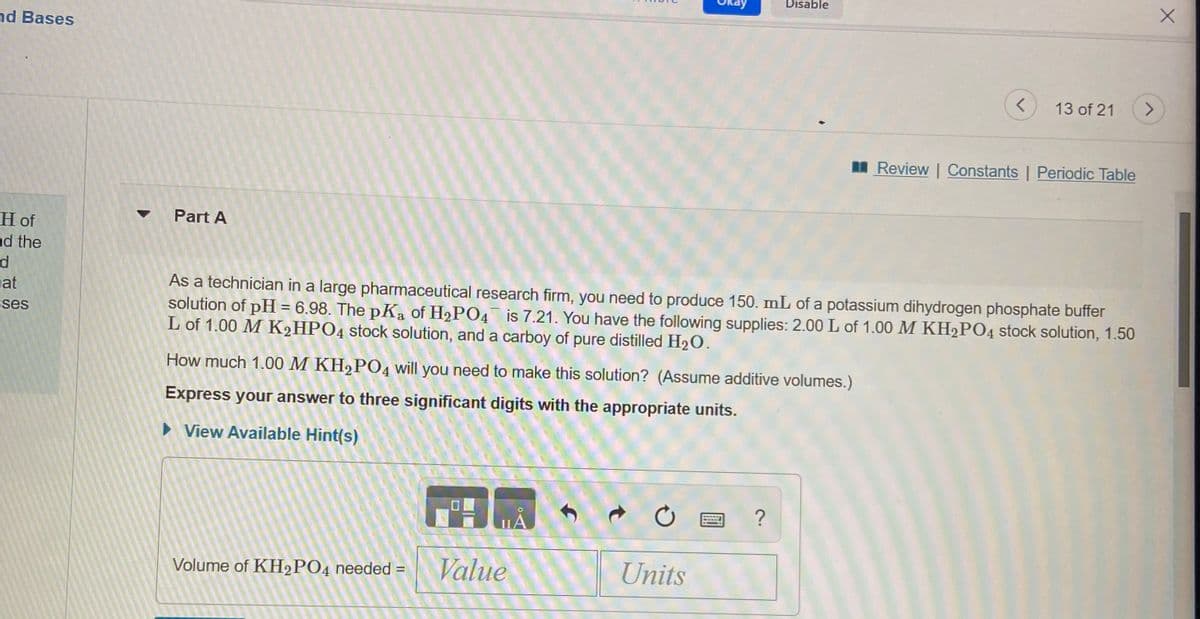
As a technician in a large pharmaceutical research firm, you need to produce 150. mL of a potassium dihydrogen phosphate buffer solution of pH = 6.98. The pKa of H2PO4¯ is 7.21. You have the following supplies: 2.00 L of 1.00 M KH2P04 stock solution, 1.50 L of 1.00 M K2HPO4 stock solution, and a carboy of pure distilled H2O. How much 1.00 M KH,PO4 will you need to make this solution? (Assume additive volumes.) Express your answer to three significant digits with the appropriate units. > View Available Hint(s)
As a technician in a large pharmaceutical research firm, you need to produce 150. mL of a potassium dihydrogen phosphate buffer solution of pH = 6.98. The pKa of H2PO4¯ is 7.21. You have the following supplies: 2.00 L of 1.00 M KH2P04 stock solution, 1.50 L of 1.00 M K2HPO4 stock solution, and a carboy of pure distilled H2O. How much 1.00 M KH,PO4 will you need to make this solution? (Assume additive volumes.) Express your answer to three significant digits with the appropriate units. > View Available Hint(s)
Chapter14: Principles Of Neutralization Titrations
Section: Chapter Questions
Problem 14.32QAP
Related questions
Question
Transcribed Image Text:Disable
nd Bases
13 of 21
<>
I Review | Constants | Periodic Table
Part A
H of
nd the
As a technician in a large pharmaceutical research firm, you need to produce 150. mL of a potassium dihydrogen phosphate buffer
solution of pH = 6.98. The pKa of H2PO4 is 7.21. You have the following supplies: 2.00 L of 1.00 M KH2PO4 stock solution, 1.50
L of 1.00 M K2HPO4 stock solution, and a carboy of pure distilled H2O.
at
ses
How much 1.00 M KH,PO4 will you need to make this solution? (Assume additive volumes.)
Express your answer to three significant digits with the appropriate units.
• View Available Hint(s)
Volume of KH2PO4 needed =
Value
Units
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Follow-up Questions
Read through expert solutions to related follow-up questions below.
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning