What mass of Fe,O, is produced when 17.3 L of O, gas react with FeS at 496 K and 5.76 atm. 4 FeS(s) + 70, (g) → 2 Fe,O,(s) + 4 SO,(g) First, use the ideal gas equation to calculate the moles of O,. mol O2 n = Then, calculate the mass of Fe,0, from the moles of O,. Show the conversions required to solve this problem. mol O, x g Fe, O, Answer Bank 4 mol SO, 1 mol O, 32.00 g O, 1 mol SO, 4 mol Fes 159.69 g Fe,O, 1 mol Fe,O, 64.06 g SO, 2 mol Fe,O, 7 mol O, 87.91 g FeS 1 mol Fes
What mass of Fe,O, is produced when 17.3 L of O, gas react with FeS at 496 K and 5.76 atm. 4 FeS(s) + 70, (g) → 2 Fe,O,(s) + 4 SO,(g) First, use the ideal gas equation to calculate the moles of O,. mol O2 n = Then, calculate the mass of Fe,0, from the moles of O,. Show the conversions required to solve this problem. mol O, x g Fe, O, Answer Bank 4 mol SO, 1 mol O, 32.00 g O, 1 mol SO, 4 mol Fes 159.69 g Fe,O, 1 mol Fe,O, 64.06 g SO, 2 mol Fe,O, 7 mol O, 87.91 g FeS 1 mol Fes
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter9: Gases
Section: Chapter Questions
Problem 77E: Ethanol, C2H5OH, is produced industrially from ethylene, C2H4, by the following sequence of...
Related questions
Question
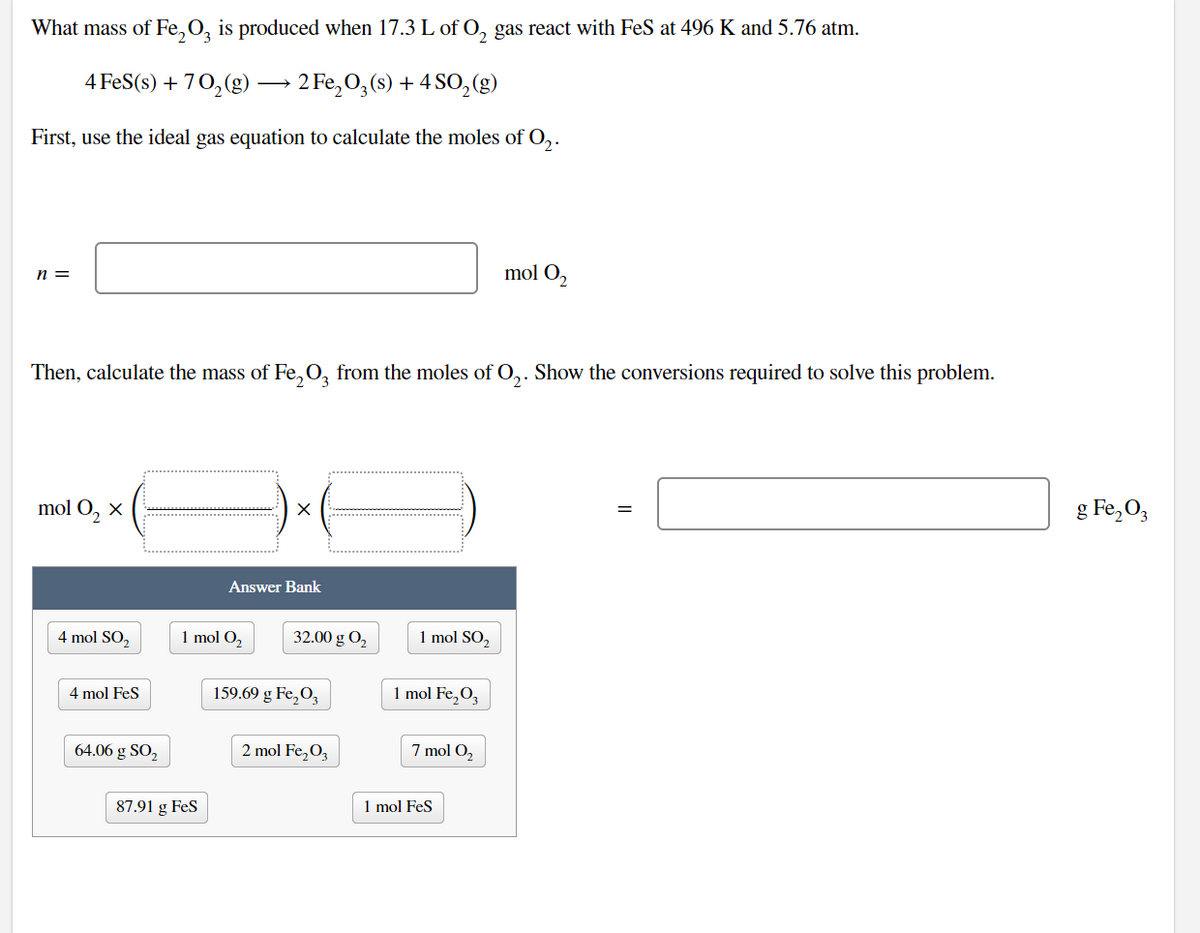
Transcribed Image Text:What mass of Fe,O, is produced when 17.3 L of 0, gas react with FeS at 496 K and 5.76 atm.
4 FeS(s) + 70,(g) -
2 Fe, 0,(s) + 4 SO,(g)
First, use the ideal gas equation to calculate the moles of O,.
n =
mol O2
Then, calculate the mass of Fe.,O, from the moles of O,. Show the conversions required to solve this problem.
mol O, x
g Fe, O,
Answer Bank
4 mol SO,
1 mol O,
32.00 g O,
1 mol SO,
4 mol FeS
159.69 g Fe,O,
1 mol Fe,O,
64.06 g SO,
2 mol Fe,O,
7 mol O,
87.91 g Fes
1 mol Fes
I|
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:
9781559539418
Author:
Angelica Stacy
Publisher:
MAC HIGHER

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:
9781559539418
Author:
Angelica Stacy
Publisher:
MAC HIGHER

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,