Q: Poly(vinyl alcohol), a hydrophilic polymer used in aqueous adhesives, is made by polymerizing vinyl…
A:
Q: The anionic polymerization of ε-caprolactam. Even if the reaction rate between caprolactam is slow,…
A: A question based on polymer chemistry, which is to be accomplished.
Q: Which monomer gives a greater yield of polymer, 5-hydroxypentanoic acid or 6-hydroxyhexanoic acid?…
A: Since we have a hydroxy group and carboxylic acid group present in the monomers. Hence there are two…
Q: Explain Markownikoff's addition and AntiMarkonikoff's addition with example.
A: Organic chemistry is branch of chemistry in which we deal with organic reactions and their…
Q: Chain branching occurs in cationic polymerization much as it does in free-radical polymerization.…
A: Concept Introduction: Cationic polymerization is the polymerization in which the reaction of…
Q: Urylon fibers are used in premium fishing nets because the polymer is relatively stable to UV light…
A: SOLUTION: Step 1: Urylon is a condensation polymer. The functional group present is polyurea. Urylon…
Q: Hexafluoropropene (as shown) can only be polymerized by free radical chemistry. A) Explain why…
A: Free-radical polymerization: it is a method of polymerization in which a polymer forms by the…
Q: Explain with more details, the polymerisation of methyl acrylate using three different…
A: The radical polymerisation of methyl acrylate is given as follows. For better understanding, if…
Q: Why does methyl methacrylate not undergo cationic polymerization?
A: In cationic polymerization the initiator is an electrophile that adds to the alkene monomer causing…
Q: FACULTY OF ENG Nylons are environment) and lose their strength in vve when implanted (not good in…
A: a) Nylons are non-absorbable and lose their strength in vivo when implanted. b) Degradation rate…
Q: Methyl acrylate and methyl methacrylate react with radical initiators (R•) as shown here. The…
A: The tendency of methyl acrylate (A) and methyl methacrylate (C) to react with radicals differ. Upon…
Q: When styrene (vinylbenzene) is commercially polymerized, about 1–3% of 1,4-divinylbenzene is often…
A:
Q: 2. 1-Methylstyrene (right) polymerizes under both cationic and anionic conditions a. Briefly…
A: A polymer is a macromolecule that is formed when a number of monomer units combine with each other.…
Q: When styrene (vinylbenzene) is commercially polymerized, about 1–3% of 1,4-divinylbenzene is often…
A:
Q: A3 Anionic polymerization of styrene is generally known to produce polymers with a narrow molecular…
A:
Q: What will be the major product for the following reaction according to the Markovnikov's rule?
A: According to Markonikov's rule, the electronegative atom (here Cl) gets attached to the carbon atom…
Q: How is alkyne prepared by Kolbe’s method?
A: Alkyne is referred to any chain of unsaturated hydrocarbons having triple bonds such as acetylene.
Q: two positions of anthracene sometimes react more like polyenes thanlike aromatic compounds.(a) Draw…
A: In case of anthracene, C9 and C10 involve in the diels-alder reaction.
Q: lumefantrine synthesis
A: Lumefantrine is an antimalarial agent which helps to treat acute malaria.
Q: Poly(n-butyl acrylate) has a lower Tg than poly(methyl methacrylate), because of: a) weaker…
A: Poly(n-butyl acrylate) and poly(methyl methacrylate) have the following structures:
Q: The polymer shown can be formed by Fischer esterification. What monomer(s) would be used to make…
A: In the polymerization , the key step is the selection of appropriate monomer to get the desired…
Q: Polycarbophil is a dietary fiber supplement. It is the calcium salt of a copolymer of acrylic acid…
A: Polycarbophil is a dietary fiber supplement. It is the calcium salt of a copolymer of acrylic acid…
Q: Give a single benefit for each polymerisation from carrying them out under a nitrogen atmosphere?…
A: Introduction: The polydispersity index (PDI) is dependent on the two parameters, i.e (I) number…
Q: Choose the best suiting monomers from the following pairs of chemicals to synthesize a polymer…
A:
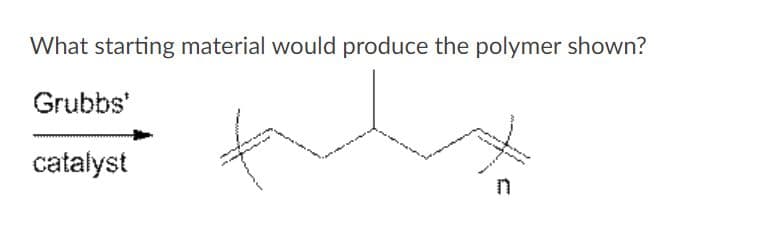
Q: ROMP F E
A: The first step of the polymerization reaction is shown as: In this step intermediate E is formed…
Q: - How does the molecular weight change vs % conversion in a step polymerization? Explain this…
A: Since you have posted a question with multiple questions, we will solve first questions for you. To…
Q: What will be the most suitable initiator used to polymerize the following monomers? Give ONE (1)…
A:
Q: 24. The cationic chain-growth, addition polymerization of a large excess of isobutylene (below)…
A: Topic--Cataionic Polymerization of the alkene (isobutene)
Q: Find the Molecular Weight (Polyhexylthiophene) untainted CH2(CH2).CH3 s, And polyhexylthiophene 25%…
A: In a polymerization reaction, monomers that are same or different combine to form larger molecule.…
Q: Delrin (polyoxymethylene) is a tough self-lubricating polymer used in gear wheels. It is made by…
A: a.
Q: Please provide the starting material
A:
Q: you react pentaeryhtritol with 4 equivalents of 2-bromo-2-methylpropionic acid bromide and now use…
A: given below.
Q: The polymer shown below is synthesized by hydroxide ion-promoted hydrolysis of a copolymer of…
A: a). Mechanism for the formation of polymer: The polymer can be formed through anionic mechanism.…
Q: Q) The monomer 1 can be polymerised via ring opening metathesis polymerisation. The resulting…
A: The given compound ‘1’ is cyclootatetraene (COT). It undergoes ring opening metathesis…
Q: Draw the starting structure that would lead to this polymer under radical conditions.
A: A question based on polymer chemistry, which is to be accomplished.
Q: Show the intermediate that would result if the growing chain added to the other end of the styrene…
A: Polystyrene is a polymer formed from the styrene monomer units. It follows addition polymerization…
Q: Terephthaloyl dichloride and p-phenylenediamine were polymerized to form a copolymer called Kevlar…
A:
Q: Show how the styrene can be prepared from benzene:
A: The reactant is benzene and product is styrene. The conversion of benzene to styrene can be…
Q: The diester shown below undergoes a Dieckmann condensation. Which product forms? C
A:
Q: Which compound undergoes the following Ruff Degradations and reactions to make all optically active…
A:
Q: Polybenzimidazoles are made by a two-stage step-growth melt polymerization: NH2 -H20 -$ OH NH2 x…
A: Given reaction:
Q: (i) Draw the structure of styrene. (ii) Markovnikov's rule. Outline the mechanism when styrene…
A: Introduction In Markovnikov's addition reaction, the negative part of the attacking molecules (like…
Q: The two hydrophilic monomers shown below can be copolymerized in an alternating fashion in aqueous…
A: Radical reactions are initiated by radical chain initiator such as tert-butyl hydroperoxide, AIBN…
Q: QUESTION 4: How would you synthesize the following polymer by anionic polymerization? Show each step…
A: Anionic addition polymerization is a form of chain-growth polymerization or addition polymerization…
Q: Delrin (polyoxymethylene) is a tough self-lubricating polymer used in gear wheels. It is made by…
A: Whether Delrin is a chain-growth polymer or step-growth polymer has to be given.
Q: What starting materials are needed to synthesize attached azo compound?
A:
Question 6 and please show the mechanism
Step by step
Solved in 2 steps with 1 images

- Show the structure of the polymer that results from heating the following diepoxide and diamine:The polymer shown below is synthesized by hydroxide ion-promoted hydrolysis of a copolymer of para-nitrophenyl methacrylate and acrylate. a. Propose a mechanism for the formation of the copolymer. b. Explain why hydrolysis of the copolymer to form the polymer occurs much more rapidly than hydrolysis of para-nitrophenyl acetate.Delrin (polyoxymethylene) is a tough self-lubricating polymer used in gear wheels. It is made by polymerizing formaldehyde in the presence of an acidcatalyst. Is Delrin a chain-growth polymer or a step-growth polymer?
- Delrin (polyoxymethylene) is a tough self-lubricating polymer used in gear wheels. It is made by polymerizing formaldehyde in the presence of an acid catalyst. a. Propose a mechanism for formation of a segment of the polymer. b. Is Delrin a chain-growth polymer or a step-growth polymer?Show the intermediate that would result if the growing chain added to the other end of the styrene double bond. Explain why the final polymer has phenyl groups substituted on every other carbon atom rather than randomly distributedWhat epoxy resin is formed by the following reaction sequence?
- (a) Draw the structure of the prepolymer A formed from 1,4-dihydroxybenzene and excess epichlorohydrin. (b) Draw the structure of the cross-linked polymer B formed when A is treated with H2NCH2CH2CH2NH2 as the hardening agent.One common type of cation exchange resin is prepared by polymerization of a mixture containing styrene and 1,4-divinylbenzene . The polymer is then treated with concentrated sulfuric acid to sulfonate a majority of the aromatic rings in the polymer. Q.) Show the product of sulfonation of each benzene ring.Which monomer and which type of initiator can you use to synthesize each of the following polymers?
- a) Draw the structure of the prepolymer A formed from 1,4 dihydroxybenzene and excess epichlorohydrin. (b) Draw the structure of the cross-linked polymer B formed when A is treated with H2NCH2CH2CH2NH2 as the hardening agent.(a) Hard contact lenses, which first became popular in the 1960s, were made by polymerizing methyl methacrylate [CH2=C(CH3)CO2CH3] to form poly(methyl methacrylate) (PMMA). Draw the structure of PMMA. (b) More-comfortable softer contact lenses introduced in the 1970s were made by polymerizing hydroxyethyl methacrylate [CH2=C(CH3)CO2CH2CH2OH] to form poly(hydroxyethyl methacrylate) (poly-HEMA). Draw the structure of poly-HEMA. Because neither polymer allows oxygen from the air to pass through to the retina, newer contact lenses that are both comfortable and oxygen-permeable have now been developed.A3 Anionic polymerization of styrene is generally known to produce polymers with a narrow molecular weight distribution. Explain the reason for this.



