Q: What product is formed when each compound undergoes thermal electrocyclic ring opening or ring…
A: In Electrocyclic reactions, one of the pi-bond of conjugated double bond converts into a ring…
Q: Hydrogenation of alkene A with Dz in the presence of Pd-C affords a single product B. Keeping this…
A: a.
Q: What cyclic product is formed when each decatetraene undergoes photochemical electrocyclic ring…
A: Structure of decatetraene is given below:
Q: The
A: We will write the reason of difference in reactivity
Q: Draw the products of radical chlorination and bromination of each compound. For which compounds is a…
A: “Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: Addition of HCl to alkene X forms two alkyt halides Y and Z. exocyclic C=C HCI CH3 CH2 -CH3 ci z a.…
A: Given : Addition of HCl to alkene X forms two alkyl halides Y and Z.
Q: c) Which of the following bicyclic ring system can be prepared by an intermolecular Robinson…
A: Organic reactions are chemical reactions which is involving in the organic compounds and the…
Q: What product is formed by the [3,3] sigmatropic rearrangement of each compound? a. b.
A: Given compounds,
Q: What product is formed by the [3,3] sigmatropic rearrangement of eachcompound?
A:
Q: One step in the synthesis of the nonsteroidal anti-inflammatory drug rofecoxib (trade name Vioxx)…
A: One step in the synthesis of the nonsteroidal anti-inflammatory drug rofecoxib (trade name Vioxx)…
Q: Synthesize B from A keeping stereochemistry in mind: N 111 A у ome HC
A: In this question, we will synthesized B from starting material A and also keeping stereochemistry in…
Q: With this in mind, draw the product of each intramolecular Diels-Alder reaction. ÇOOCH, a. b.
A: The reaction between conjugated diene with substituted alkene (dienophile) results in the formation…
Q: Taking into account anti periplanar geometry, predict the major E2 product formed from each starting…
A: The compounds in which the leaving group and the adjacent hydrogen atom are anti-periplanar to each…
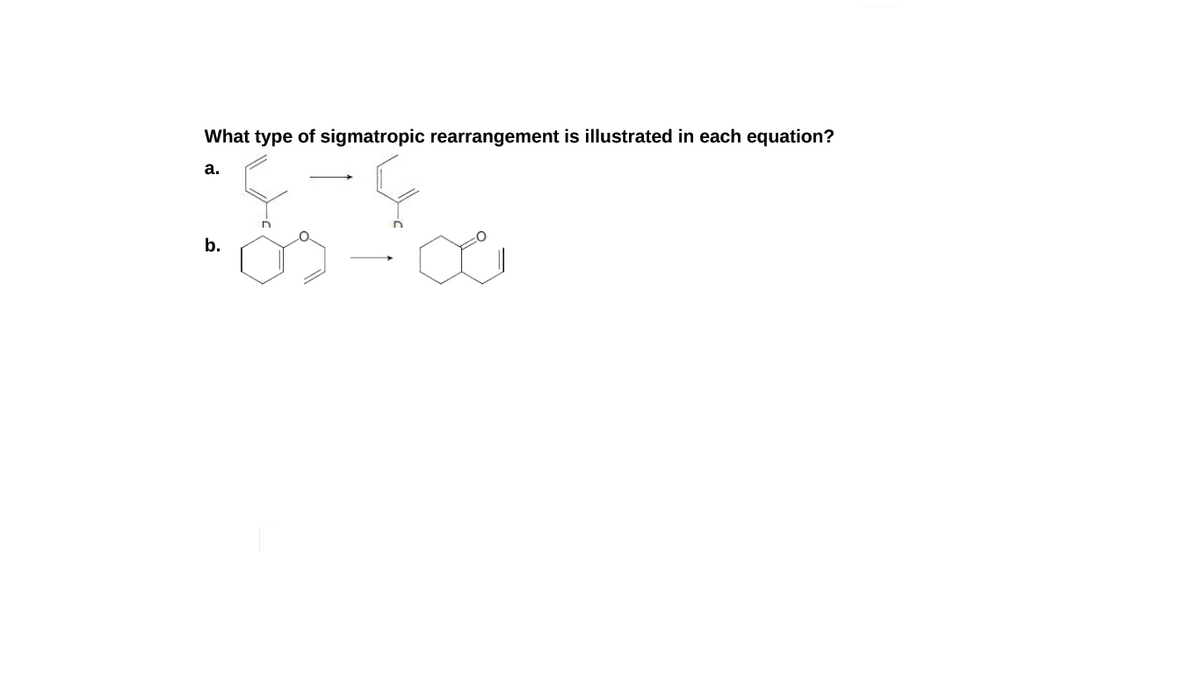
Q: What type of sigmatropic rearrangement is illustrated in each equation? a. b. D
A: Sigmatropic rearrangement reaction is one the important class of pericylic reactions. In this…
Q: Identify the more reactive dienophile in each of the following pairs.
A: Dienophiles are alkenes with an electron-withdrawing group. Dienophiles react with dienes in…
Q: Devise a synthesis of X from the given starting materials. You may useany organic or inorganic…
A:
Q: Rank the following alkenes in order of increasing stability:
A: The stability of alkene depends upon the degree of substitution and due to the hyperconjugation…
Q: What type of sigmatropic rearrangement is illustrated in each equation?
A: The first rearrangement is an example of sigmatropic [1,3] rearrangement. In this rearrangement, the…
Q: Synthesize B from A keeping stereochemistry in mind: N 11 0 A OMe не
A: In this question, we will synthesized the final product B from the starting material A. You can see…
Q: Attached compounds undergoes E2 elimination with strongbase? For compounds that undergo elimination,…
A:
Q: Which C–H bond in attached compound is most readily broken during radical halogenation?
A: a. During cleavage of C-H bond about 3 types of radical can be formed from the given molecule. Of…
Q: Write the IUPAC name and, where possible, the common name of each compound. Show stereochemistry…
A:
Q: What cyclic product is formed when each decatetraene undergoes thermal electrocyclic ring closure?
A: The number of electrons involved in the thermal electrocyclic ring closure or ring opening must obey…
Q: What compound undergoes metathesis to form each of the following compounds?
A: First break the double bond present in product made after metathesis reaction and add =CH2 to each…
Q: a. Which of the following compounds cannot be prepared by a Heck reaction?b. For those compounds…
A: Introduction: Heck reaction: Heck reaction is used to synthesize the substituted alkene. It consist…
Q: Classify each reaction as an electrocyclic reaction, a cycloaddition, or a sigmatropic…
A: Electrocyclic Reaction is a type of pericyclic reaction in which one pie bond is converted into one…
Q: (a) What product is formed when triene N undergoes thermal electrocyclic ring closure? (b) What…
A: If the number of electrons involves in thermal elelctrocyclic ring opening or ring closure obeys…
Q: Devise a stepwise synthesis of attached compound from dicyclopentadiene using a Diels–Alder reaction…
A: Our target molecule is
Q: What neutral nucleophile is needed to convert C to D? Imatinib, an effective treatment for certain…
A: Since in C and D, the difference is of and Cl part hence it means has attacked on the molecule and…
Q: What compound undergoes metathesis to form each of the following compounds?
A:
Q: What cyclic product is formed when each decatetraene undergoes thermal electrocyclic ring closure?
A:
Q: Dienynes undergo metathesis to afford fused bicyclic ring systems. (a Explain how A is converted to…
A: Metathesis: Inter change of the carbons of their double bonds when two alkenes react is known as…
Q: Consider the tetracyclic compound with rings labeled A–D. (a) Which ring is the most reactive in…
A: Benzene ring is activated towards electrophilic substitution reaction if there is an electron…
Q: Draw the product of the [3,3] sigmatropic rearrangement of each compound.
A: 3,3 sigmatropic rearrangement involves pericyclic reaction that proceeds via Huckel topology…
Q: 2. Draw the most stable enol tautomer for the following compounds b. C.
A: Ketones form keto - enol tautomers , which contain alkene functional group ( C=C ) and Alcoholic…
Q: What cyclic product is formed when each decatetraene undergoes photochemical electrocyclic ring…
A: a.
Q: What product is formed when each compound undergoes thermal electrocyclic ring opening or ring…
A: Incase of electrocyclic reactions, one of pi-bond of conjugated double bond is converted to ring…
Q: Dienynes undergo metathesis to afford fused bicyclic ring systems. (a)Explain how A is converted to…
A: Metathesis: Inter change of the carbons of their double bonds when two alkenes react is known as…
Q: Taking into account anti periplanar geometry, predict the major E2 product formed from each starting…
A: E2 reactions are the elimination reactions in which the reactant and the reagent or the nucleophile…
Q: a. Cycloheptyl methyl ether from cycloheptene and any other necessary reagents.…
A: Organic reaction mechanisms:
Q: 3. Deduce the structures of A, B and C and reagent(s) D in reaction scheme shown below. What is the…
A: 1. Given substrate is an alkyl chloride (2-Chloro butane) with bulkier base like tertiary butoxide…
Q: What dene and dienophile is needed to prepare the follow Choices a and b c and d e and f c and f b…
A: The reaction of both a and b reagents in option 'A' as well as c and d reagents in option 'B' will…
Q: Heating A results in two successive [3,3] sigmatropic rearrangements–Claisen reaction followed by…
A: Claisen rearrangement is an organic chemical reaction that offers a powerful method in the formation…
Q: Hydrogenation of alkene A with D2 in the presence of Pd-C affords a single product B. Keeping this…
A: a.
Q: Which C–H bond in each compound is most readily broken during radical halogenation?
A: a. During cleavage of C-H bond about 3 types of radical can be formed from the given molecule. Of…
Q: What alkene yields each set of oxidative cleavage products?
A: Gentle oxidative cleavage of the alkene in presence of KMnO4, O3 will give carbonyl products. The…
Q: By what mechanism is the following reaction likely to occur? (SN1, SN2, E1, E2) Put an "and" between…
A: In 1 reaction alkyl halide is 1°and SH- is strong nucleophile. SN2 reaction will takes place.…
Q: Addition of HCl to alkene X forms two alkyl halides Y and Z.a.Label Y and Z as a 1,2-addition…
A:
Q: Which of the following products is(are) formed by Wittig reaction of CH 3CH 2CH 2CHO with Ph 3P-CHCH…
A: We have given the organic reaction and we have to find the product of the reaction.
Q: Draw the products of radical chlorination and bromination of eachcompound. For which compounds is a…
A: Bromination is more selective than chlorination and secondary halogenation is preferred in case of…
What type of sigmatropic rearrangement is illustrated in each equation ?
Step by step
Solved in 2 steps

- What type of sigmatropic rearrangement is illustrated in each reaction?Suzuki coupling of aryl iodide A and vinylborane B affords compound C,which is converted to D in the presence of aqueous acid. Identifycompounds C and D and draw a stepwise mechanism for the conversionof C to D.Addition of HCl to alkene X forms two alkyl halides Y and Z. (A) Label Y and Z as a kinetic or thermodynamic product and explain why. (B) Explain why the addition of HCl occurs at the exocyclic C=C, rather than the other C=C
- Draw the major organic product of the reaction. Ignore stereochemistryHeating A results in two successive [3,3] sigmatropic rearrangements–Claisen reaction followed by Cope reaction–to afford β-sinensal, a component of mandarin orange oil. What is the structure of β-sinensal?Devise a stepwise synthesis of attached compound from dicyclopentadieneusing a Diels–Alder reaction as one step. You may also use organiccompounds having ≤ 4 C's, and any required organic or inorganicreagents.
- Identify F in the following reaction sequence. F was converted in several steps to the antidepressant paroxetine (trade name Paxil; see also Problem 9.9).Interestingly, reduction of the complex shown above mostly gives thealcohol enantiomer shown below after workup. a. Draw the other minor diastereomeric alcohol product. b. Briefly explain the diastereoselectivity of the reduction – why is oneenantiomer heavily favored? Draw a structure or structures to support youranswer.What is the major organic product obtained from the following reaction under low temperature conditions? A, B, C, or D?
- 1. Draw the organic product (ignoring stereochemistry) 2. State whether the process is overall oxidation, overall reduction, or neither.Bromide Bhas normal activity (for a secondary bromide) towards SN1 substitution, but A has much higher reactivity and Chas much lower reactivity.What product is formed by the [3,3] sigmatropic rearrangement of each compound?



