Q: Drug name: p-cresol
A:
Q: Choose the most acidic compound from Compounds l-IV. H. O,N-CH,-C-CH; OFt II III IV
A: The given compounds are : We have to determine which compound is the most acidic.
Q: A 38.0 g sample of hot copper is carefully placed into an insulated container containing 172 g of wa...
A: Find the complete solution given below.
Q: Given the image below chose the best options that’s analyze the content of the image
A: Given : The content of image. To find : Best options in the given blanks brackets. Solution : As we...
Q: Balance the equation in basic aqueous solution Al(s) + MnO4 (aq) → MnO2(s) + Al(OH)4(aq) What are th...
A: Redox reactions are those involving oxidation of one species and reduction of another. Redox reactio...
Q: What is the pH of a buffer solution made by dissolving 0.10 mol of formic acid, HCOOH, and 0.10 mol...
A: pH of an acidic buffer solution: pH = pKa + logHCOO-HCOOH pH = 3.75 + log 0.100.10 pH = 3.75 + log 1...
Q: For the chemical reaction: Cu + 2AGNO3 → 2Ag + Cu(NO3)2 If 100 moles of Cu were to react with 5 mole...
A:
Q: How many moles of MgCl2 are produced from 35.0g of Mg and 35.0g of Mg + 2HCI → MgCl2 + H2 O 0.956 mo...
A: Since you have Posted multiple Question. We will solve the first question for you. To get the remain...
Q: Identify the major product(s) for the following reaction. Select all that apply. If the reaction doe...
A: When Toluene is treated with N - Bromo succinimide ( NBS ), in presence of sunlight, then there is b...
Q: True/False The change in entropy (Delta S) for the surroundings is equal to the negative of the enth...
A: We are given the question statement. The correct option is: a. True
Q: 100.0 mL of a 0.500 M solution of KBr is diluted to 500.0 mL. What is the nw concentration of the so...
A: If a solution having M1 concentration and V1 volume and it is diluted to M2 Concentration and V2 Vol...
Q: Regarding an aromatic cycle, the -NH-C(=DO)CH3 group has an electronic effect : Veuillez choisir une...
A: Both the electronic effect are explained as follows;
Q: Explain how the periodic table of the elements is organized.
A: Modern periodic law states that the properties of elements are periodic functions of their atomic nu...
Q: Write a chemical equation (not the mechanism) for the reaction of 1-butanol with each of the followi...
A: Answer: a. b. c.
Q: A chlorine (Cl) atom is adsorbed on a small patch of surface (see sketch at right). This patch is kn...
A: Change in Entropy for the adsorbed site can be given as follows: ∆S=KBlnXXo where KB: Boltzmann Cons...
Q: Which of the following electron configuration pairs would most likely result in covalent bonding? A...
A: Covalent bond formation take between non metals and metalloids or metalloids to non metal.
Q: 10. Based upon your observations of the dissolution of NH,Cl in exothermic or endothermic? Briefly e...
A: We above to explain about enthalpy of dissolution of ammonium chloride
Q: What is the free energy change in kJ for the reaction below?Cl2(g) + 6 H2O(l) + 5 Sn2+(aq) → 2 ClO3−...
A: E°=-1.606V ∆G°rxn= -nFE°cell
Q: Determine the [H+] or [OH-] concentrations using the Product Ion Constant of Water at 25 C: [H+] [...
A: Answet is given as follows
Q: You may have noticed that the "solvent of choice anard reagents is dried diethyl ether or tetrahydro...
A: Alkylmagnesiumhalide is called "grignard reagent " It is an organo metallic compound since Carbon-Me...
Q: . Why is it important in diluting a concentrated acid that you add acid to water, not water to acid?
A: The reason for adding acid to water and not water to acid while diluting the acid is as follows:
Q: Please look at pic
A: Lattice energy It defines the strength between the opposite charge ions that form Lattice. More stro...
Q: The following reaction is exothermic: 2NO (g) + Br2 (g) ⇌ 2NOBr (g) After it reaches equilibrium, ...
A: We have the following reaction 2NO (g) + Br2 (g) ⇌ 2NOBr (g) This equilibrium reaction is exothermic...
Q: Write the molecular formula for the following molecule. Use the format CxHyOzCxHyOz. For example...
A: Molecular formula denotes total number of atoms present in one molecule
Q: Which reaction, when at equilibrium, would be unaffected by a change in pressure? A. N2(g) + O2 (g...
A: The answer is given Asfollows
Q: How many moles of nitrogen monoxide (NO) will be produced from 8.00 moles of NH3 and 8.00 moles of O...
A: Ammonia reacts with oxygen to form nitrogen oxide and water. The equation for the balanced chemical ...
Q: Which of the following liquid substances has the highest vapor pressure at its normal boiling point?...
A: Given datas are: A) CH₃OH (b.p. = 65°C) B) C₆H₆ (b.p. = 80°C) C) C₂H₅OH (b.p. = 78°C) D) H₂O ...
Q: H* Key A H* A- H* Dissociated H* ion A H* H* A- Dissociated A ion H* A- A - A H НА Does the diagram ...
A: We have a diagram representing the dissociated H+ , dissociated A- and HA. We are asked to determine...
Q: For the reaction below, 0.333M A and 0.500M B are placed in an container. At equilibrium, the concen...
A: We have to calculate concentration of B at equilibrium.
Q: i What mass of aluminum sulfide is produced when 3.00 moles of aluminum reacts with excess sulfur ac...
A: e) 225 g Given reaction; 2Al(s) + 3S(s) ----> Al2S3 (s) moles of Aluminum given = 3.0 mol
Q: Which of the following laws describe the relationship between gap pressure and gas volume? A) Boyle...
A: We are given that we have a relation between the gas pressure and volume . We have to predict which ...
Q: Which one of the following would have the highest heat of vaporization? A) CH₃OH B) CH₂F₂ C)...
A: Heat of vaporisation is affected by H- bonding. Heat of vaporisation is directly proportional to H- ...
Q: adhesive vs cohesive forces in a liquid and capillary
A: Forces that are present in same type of molecules i.e. attractive forces that act between same type ...
Q: 5. When you take 20.0 mL of 6.0M HNO3 and dilute to a final volume of 300.0 mL?
A:
Q: Direct methanol fuel cells (DMFCS) have shown some promise as a viable option for providing "green" ...
A:
Q: Which image above correctly represents the Lewis dot structure of antimony
A: We have to choose correct structure of antimony
Q: A bicycle tire is filled to a pressure of 110. psi at a temperature of 30.0oC. At what temperature w...
A: Given : P1 = 110 psi P2 = 105 psi T1 = 30.0°C
Q: Which of these aqueous solutions is expected to have the highest pH at 25 °C? 0 1.0 М Са(ОН)2 0 1.50...
A: pH of Acid is below 7 and pH of base is above 7.
Q: 7. If an element, “X" has two isotopes with the following masses and abundances: 46X = 45.974 amu , ...
A:
Q: experiment on the table below. Calculate the H* and OH concentrations of these solutions, and indica...
A: Since you have asked a question with multiple sub-parts, we will solve first three sub-parts for y...
Q: The value of AS° for the catalytic hydrogenation of acetylene to ethene, C2H2 (g) + H2 (g) → C2H4 (g...
A: Ans is -112j/kmol
Q: Calculate the amount of oxygen, nitrogen, and carbon dioxide that are produced or consumed by the ba...
A: The amounts of reactants and products in a reaction can be compared using the stoichiometry of the i...
Q: A + B → C + D is of order 0. As an experimenter, which parameters can you make vary to influence the...
A: The variation in the following parameters will affect the rate of zero order reaction.
Q: the equilibrium constant of the reaction 2A + B ⇌ C is 9.4 x 10^9 Which of the following is true? ...
A: For Reaction :: 2A + B ⇌ C Kc = 9.4 x 109
Q: What happens when too much NaSCN is prepared in solution of mixtures of standard solutions of Fe(NO3...
A: Fe3+ (from Fe(NO3)3 and SCN- ions (from NaSCN) react to form a complex.
Q: For the chemical reaction: Cu + 2AgNO3 → 2Ag + Cu(NO3)2 If 5 moles of Cu were to react with 100 mole...
A: Limiting reagent is the reactant that is fully consumed during the reaction. It is present in lesse...
Q: A sample of a cleaning product containing household amm of 11.50. Determine the hydroxide ion concen...
A: We will calculate concentration of OH-.
Q: Describe Lewis Acids and BasesLewis Acids and Bases?
A: The terms that is Lewis acids and Lewis bases are given by Lewis theory of acid- base reactions. And...
Q: Determine the pH and equilibrium concentrations of all species of a 25 M H2SO3 solution at 25oC
A:
Q: The image below is a representation of which one listed below a nuclear charge Electronegativity I...
A: The correct answer is Electron shielding.
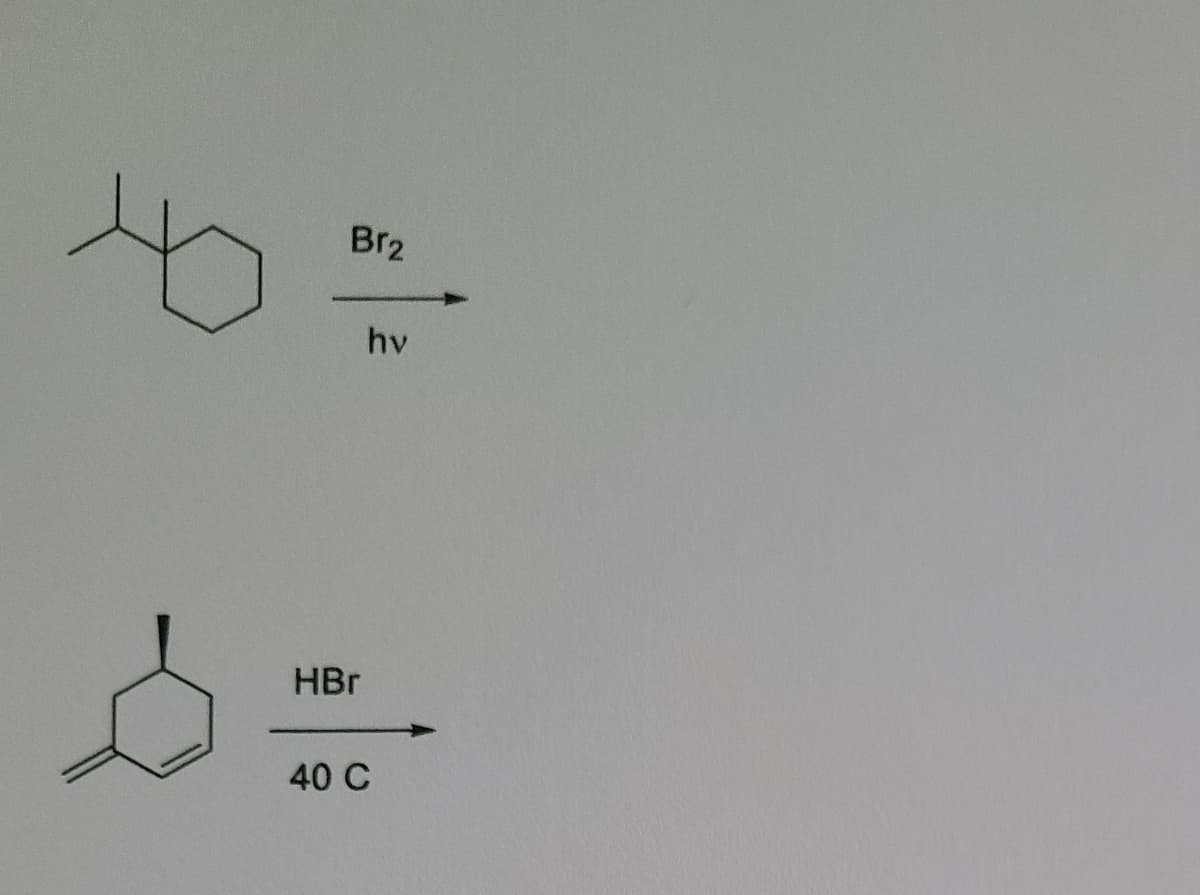
Step by step
Solved in 2 steps with 1 images


