When an aqueous, neutral NiCl₂ solution is electrolyzed with 20 dc voltage, Cl₂ gas is the primary product in the anode, while H₂ gas will be the primary product in the cathode. Consider that the overpotentials for the production of gases Cl₂, O₂ and H₂ are negligible (~0.0 V). Eº Cl₂/Cl-: +1.36 V Eº O₂, H+/H₂O: +1.23 V E° H*/H₂: 0.00 V E° Ni+2/Ni: -0.23 V E° H₂O/H₂, OH-: -0.83 V Select one: O True O False
When an aqueous, neutral NiCl₂ solution is electrolyzed with 20 dc voltage, Cl₂ gas is the primary product in the anode, while H₂ gas will be the primary product in the cathode. Consider that the overpotentials for the production of gases Cl₂, O₂ and H₂ are negligible (~0.0 V). Eº Cl₂/Cl-: +1.36 V Eº O₂, H+/H₂O: +1.23 V E° H*/H₂: 0.00 V E° Ni+2/Ni: -0.23 V E° H₂O/H₂, OH-: -0.83 V Select one: O True O False
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter24: Coulometry
Section: Chapter Questions
Problem 24.1QAP
Related questions
Question
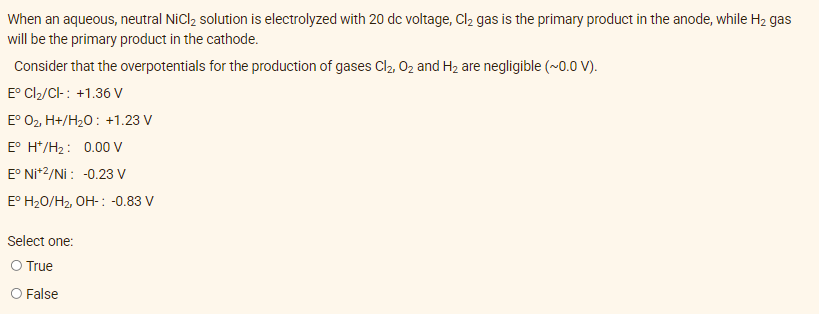
Transcribed Image Text:When an aqueous, neutral NiCl₂ solution is electrolyzed with 20 dc voltage, Cl₂ gas is the primary product in the anode, while H₂ gas
will be the primary product in the cathode.
Consider that the overpotentials for the production of gases Cl₂, O₂ and H₂ are negligible (~0.0 V).
Eº Cl₂/Cl-: +1.36 V
Eº O₂, H+/H₂O: +1.23 V
E° H*/H₂: 0.00 V
E° Ni+2/Ni: -0.23 V
E° H₂O/H₂, OH-: -0.83 V
Select one:
O True
O False
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning