When calcium carbonate is added to hydrochloric acid, calcium chloride, carbon dioxide, and water are produced. CaCO, (s) + 2 HCl(aq) - CaCl, (aq) + H,O(1) + CO,(g) > How many grams of calcium chloride will be produced when 26.0 g of calcium carbonate is combined with 14.0 g of hydrochloric acid? mass of CaCl,: | 320 g Which reactant is in excess? HCI CACO,
When calcium carbonate is added to hydrochloric acid, calcium chloride, carbon dioxide, and water are produced. CaCO, (s) + 2 HCl(aq) - CaCl, (aq) + H,O(1) + CO,(g) > How many grams of calcium chloride will be produced when 26.0 g of calcium carbonate is combined with 14.0 g of hydrochloric acid? mass of CaCl,: | 320 g Which reactant is in excess? HCI CACO,
Living By Chemistry: First Edition Textbook
1st Edition
ISBN:9781559539418
Author:Angelica Stacy
Publisher:Angelica Stacy
ChapterU5: Fire: Energy , Thermodynamics, And Oxidation-reduction
Section: Chapter Questions
Problem SII2RE
Related questions
Question
I understand the beginning but I get lost near the end of converting the moles of CaCl2 to grams.
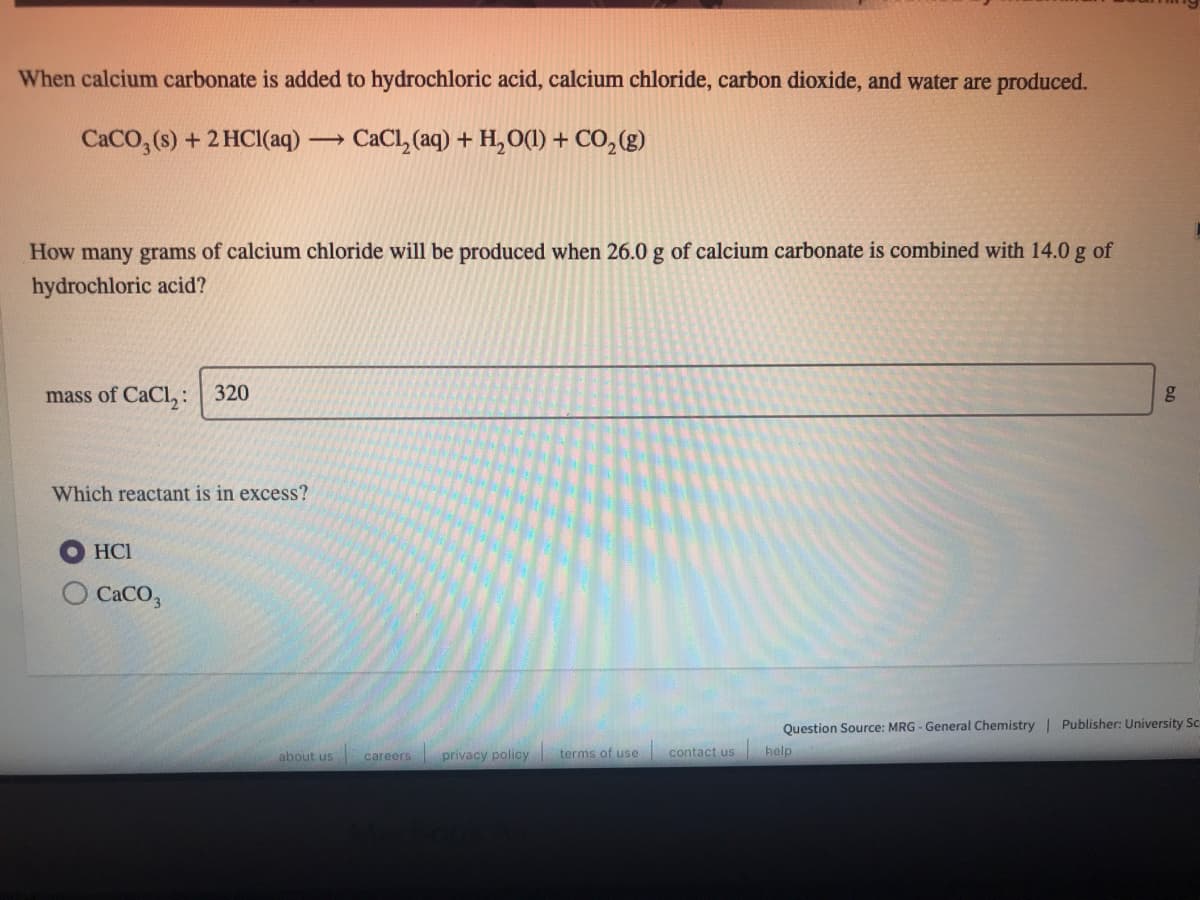
Transcribed Image Text:When calcium carbonate is added to hydrochloric acid, calcium chloride, carbon dioxide, and water are produced.
CACO, (s) + 2 HCI(aq)
CaCl, (aq) + H, O(1) + CO,(g)
How many grams of calcium chloride will be produced when 26.0 g of calcium carbonate is combined with 14.0 g of
hydrochloric acid?
mass of CaCl,:
320
g
Which reactant is in excess?
HCI
CACO,
Question Source: MRG - General Chemistry | Publisher: University Sc
about us
privacy policy terms of use
contact us
help
careors
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:
9781559539418
Author:
Angelica Stacy
Publisher:
MAC HIGHER

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:
9781559539418
Author:
Angelica Stacy
Publisher:
MAC HIGHER

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning