When ethanol is added to water at 25 °C and 1 bar, the total volume of the solution changes according to the empirical function cm x - 0.36394 cm (mol EtOH) = ** V = 1002.93 cm + 56.6664 mol EtOH cm (mol EtOH) + 0.028256- where x is the number of moles of ethanol in the solution. As ethanol is added to the solution, the partial molar volume of ethanol decreases, reaches a minimum, and then increases. This behavior is shown in the figure below. Find the function for (ma) find the value x = ng 56 Ong TP How many moles of ethanol are present when the partial molar volume of ethanol in the solution is minimum? How many moles of water are present if the mole fraction of ethanol with this amount of ethanol present is 0.0952? 53 5. *- nmol 10 Is the partial molar volume of water increasing, decreasing with increasing x, at a maximum or at a minimum? Explain based on an equation based on an equation from the lecture slides. Partial melar volume, VAcm mot
When ethanol is added to water at 25 °C and 1 bar, the total volume of the solution changes according to the empirical function cm x - 0.36394 cm (mol EtOH) = ** V = 1002.93 cm + 56.6664 mol EtOH cm (mol EtOH) + 0.028256- where x is the number of moles of ethanol in the solution. As ethanol is added to the solution, the partial molar volume of ethanol decreases, reaches a minimum, and then increases. This behavior is shown in the figure below. Find the function for (ma) find the value x = ng 56 Ong TP How many moles of ethanol are present when the partial molar volume of ethanol in the solution is minimum? How many moles of water are present if the mole fraction of ethanol with this amount of ethanol present is 0.0952? 53 5. *- nmol 10 Is the partial molar volume of water increasing, decreasing with increasing x, at a maximum or at a minimum? Explain based on an equation based on an equation from the lecture slides. Partial melar volume, VAcm mot
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter2: Electrical Components And Circuits
Section: Chapter Questions
Problem 2.12QAP
Related questions
Question
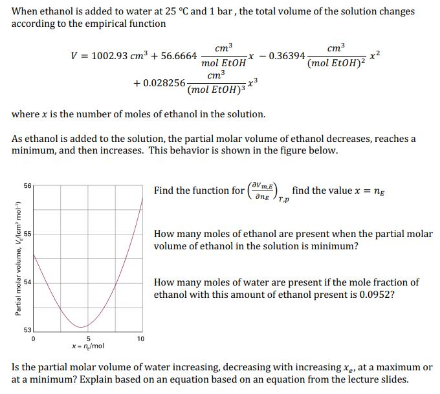
Transcribed Image Text:When ethanol is added to water at 25 °C and 1 bar, the total volume of the solution changes
according to the empirical function
cm
x - 0.36394
cm
(mol EtOH)
= **
V = 1002.93 cm + 56.6664
mol EtOH
cm
(mol EtOH)
+ 0.028256-
where x is the number of moles of ethanol in the solution.
As ethanol is added to the solution, the partial molar volume of ethanol decreases, reaches a
minimum, and then increases. This behavior is shown in the figure below.
Find the function for (ma) find the value x = ng
56
Ong TP
How many moles of ethanol are present when the partial molar
volume of ethanol in the solution is minimum?
How many moles of water are present if the mole fraction of
ethanol with this amount of ethanol present is 0.0952?
53
5.
*- nmol
10
Is the partial molar volume of water increasing, decreasing with increasing x, at a maximum or
at a minimum? Explain based on an equation based on an equation from the lecture slides.
Partial melar volume, VAcm mot
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 5 steps with 4 images

Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning