When limestone (solid CaCO3) is heated, it decomposes into lime (solid CaO) and carbon dioxide gas. This is an extremely useful industrial process of great antiquity, because powdered lime mixed with water is the basis for mortar and concrete - the lime absorbs CO₂ from the air and turns back into hard, durable limestone. A Suppose a limekiln of volume 750. L is pressurized with carbon dioxide gas to 25.5 atm, and heated to 700.0 °C. When the amount of CO₂ has stopped changing, it is found that 8.46 kg of CaCO3 have appeared. olo Calculate the pressure equilibrium constant K, this experiment suggests for the equilibrium between CaCO3 and CaO at 700.0 °C. Round your answer to 2 significant digits. Note for advanced students: it's possible there was some error in this experiment, and the value it suggests for K does not match the accepted value. 4 K = ? 0.0
When limestone (solid CaCO3) is heated, it decomposes into lime (solid CaO) and carbon dioxide gas. This is an extremely useful industrial process of great antiquity, because powdered lime mixed with water is the basis for mortar and concrete - the lime absorbs CO₂ from the air and turns back into hard, durable limestone. A Suppose a limekiln of volume 750. L is pressurized with carbon dioxide gas to 25.5 atm, and heated to 700.0 °C. When the amount of CO₂ has stopped changing, it is found that 8.46 kg of CaCO3 have appeared. olo Calculate the pressure equilibrium constant K, this experiment suggests for the equilibrium between CaCO3 and CaO at 700.0 °C. Round your answer to 2 significant digits. Note for advanced students: it's possible there was some error in this experiment, and the value it suggests for K does not match the accepted value. 4 K = ? 0.0
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter5: Gases
Section: Chapter Questions
Problem 78QAP
Related questions
Question
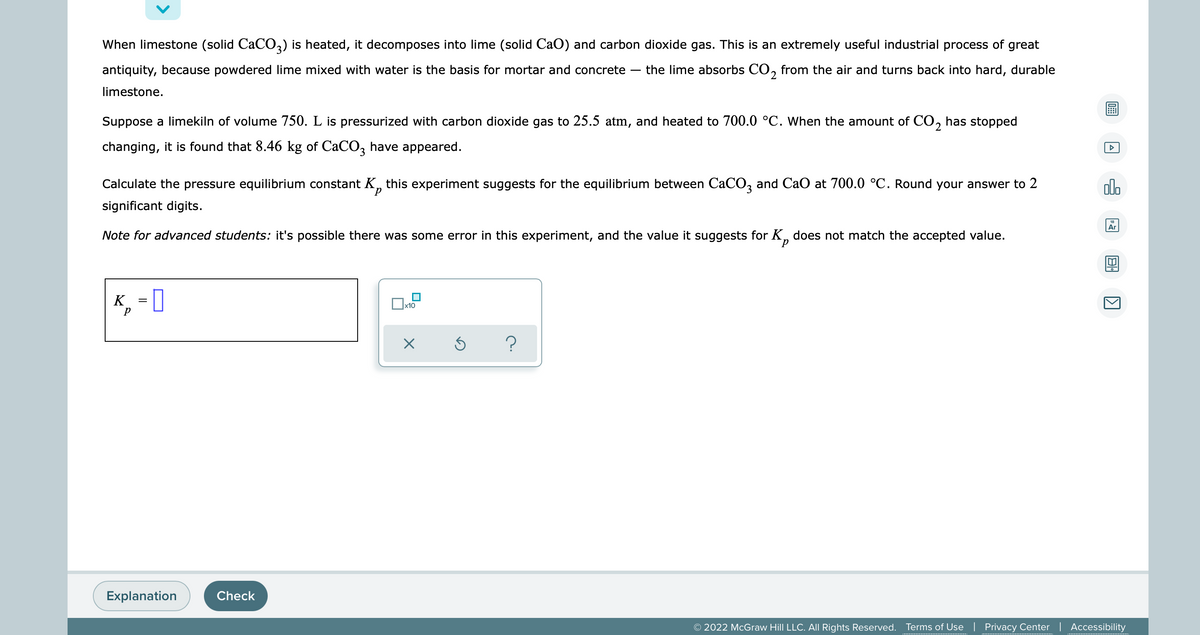
Transcribed Image Text:When limestone (solid CaCO3) is heated, it decomposes into lime (solid CaO) and carbon dioxide gas. This is an extremely useful industrial process of great
antiquity, because powdered lime mixed with water is the basis for mortar and concrete the lime absorbs CO₂ from the air and turns back into hard, durable
limestone.
Suppose a limekiln of volume 750. L is pressurized with carbon dioxide gas to 25.5 atm, and heated to 700.0 °C. When the amount of CO₂ has stopped
changing, it is found that 8.46 kg of CaCO3 have appeared.
Calculate the pressure equilibrium constant K this experiment suggests for the equilibrium between CaCO3 and CaO at 700.0 °C. Round your answer to 2
significant digits.
alo
Ar
Note for advanced students: it's possible there was some error in this experiment, and the value it suggests for K does not match the accepted value.
р
K₁ =
x10
р
X
Ś ?
Explanation
© 2022 McGraw Hill LLC. All Rights Reserved. Terms of Use
Check
A
B
K
Privacy Center | Accessibility
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning