Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter21: Surface Characterization By Spectroscopy And Microscopy
Section: Chapter Questions
Problem 21.10QAP
Related questions
Question
2. I need help
Transcribed Image Text:S.
HW Check :Intermolecular Force X
org/assignment/4622623085/assessment
y Scho.
Home-WA ISD 129...
Home Schoology
Zoom!
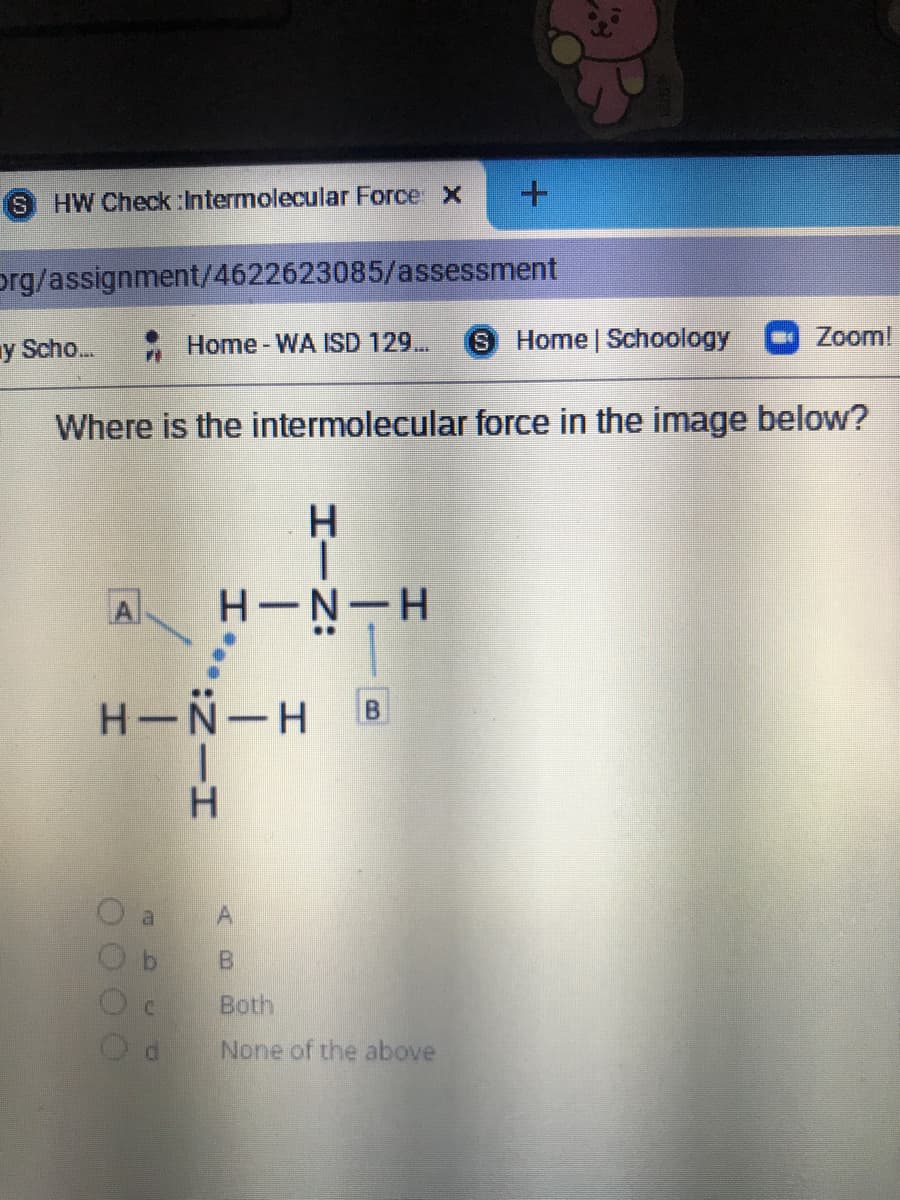
Where is the intermolecular force in the image below?
A
H-N-H B
H.
al
Both
None of the above
HIN:
Expert Solution
Step 1
Correct answer is (A).
Intermolecular force is between two molecules and it is denoted by dotted line.
It is hydrogen bond between NH3 molecules.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning