Q: absorbance 0.8 0.7 y 1.4x- 0.01. 0.6 0.5 0.4 0.3 0.2 0.1 0.1 0.2 0.3 0.4 0.5 0.6 Concentration…
A: In the absorbance vs concentration graph : x-axis represents the concentration of sample. y-axis…
Q: QUESTION 20 A sample run through a GC gave a peak with retention time of 2 431 minutes, a peak…
A:
Q: II rome File Edit View History Bookmarks Profiles Tab Window Help Watch Gilmore Girls | Netflix (2…
A:
Q: What is sampling in analytical chemistry?
A: Analytical chemistry is a branch of chemistry in which we analyze the sample by different…
Q: What is the most accurate volume that the graduations can measure in this instrument? Pinch and zoom…
A: Measuring cylinder (also known as graduated cylinder) is used to quantify a certain volume of liquid…
Q: What is the role of the fixer in black and white photography?
A: The role of the fixer in black and white photograph – The fixer stabilises the image, removing the…
Q: Which of the following is a relative dating method technique? Group of answer choices Carbon-14 K/Ar…
A: Relative dating is determining whether an object or event or is older or younger than other objects…
Q: Page 17: Choose all of the following extensive properties density volume color mass melting point O…
A: A multiple choice question about extensive properties, which is to be accomplished.
Q: www は Expl Set Col Bkg Col Smp Орen Save Print Stack Spe Ful Sc Cmn Scl Aut Bsin Ady ATR Subtract…
A: To explain: Need to fill the table.
Q: A balance high readability is necessarily equated to high accuracy. Select one: a. True b. False
A: Given statement, A balance high readability is necessarily equated to high accuracy.
Q: 6. List common analytical apps for SPE? 7. What is sample preparation in analytical chemistry?. WHY…
A: Solid Phase Extraction(SPE) is a sample preparation technique that makes use of a solid adsorbent…
Q: What is the importance of the following analytical methods in chemical analysis? i. UV-vis…
A:
Q: 200 180 160 140 120 100 80 60 40 20 CDS-04-752 ppm H2N- ppm Int. 标记碳 167.40 146 1 151.41 253 2…
A: We have to discuss the functional groups identified in this NMR spectrum of following given…
Q: the VT of a column is 30 cm3 , and VS is 20 cm3 , and Vo is 7 mL, find Vi (in mL).
A:
Q: Please can you help me out with the missing columns on the first photo.
A: The missing columns on the first photo are as follows:
Q: The following data were obtained for 1.00 cm samples of a particular chemical. What is the…
A: In the given graph, x-axis represents concentration of the sample. y-axis represents absorbance of…
Q: A 1.058-g sample of a copper unknown was dissolved in water and diluted to a final volume of 100.0…
A: The calibration curve equation is 8.58 M = A Given : Absorbance = 0.529 = A Hence 0.529 = 8.58 M…
Q: need
A: According to the question, we need to complete the reaction. Reaction 01: C2H4(g) + H2O(aq)…
Q: How does the perspective of the grid change in the first set of photographs?
A: Given How does the perspective of the grid change in the first set of photographs
Q: Calculate the Rf value for The spot on the TLC plate shown below. Measure distances in mm (with…
A:
Q: Mark the statements which are correct. (Select all that apply.) 103 mg = 1010 ng 106 mg = 1012 ng…
A: 1 liter = 1000ml 1gm = 10^9 ng 1L =1dm^3
Q: b) ہنڈا F Br xxx Sug g) Br etco Fall 2021 Spring 2022 Screen Shot 2022-0...49.34 PM Organic…
A: Name the following compounds. only part f,c and g.
Q: What should you do if you get a chemical on your skin? For the toolbar, press ALT+F10 (PC) or…
A: If the chemical drops coincidentally get on the skin the skin may burn if the chemical is in…
Q: Considering the placement of Cu and I in the periodic table. explain why Cu is used for electrical…
A: Considering the placement of Copper (Cu) and Iodine (I), the reason for employing copper for making…
Q: For the data below, Write the linear equation of the line (use 5 Decimal places and don't add…
A:
Q: A 15X ocular lens used with a 100X objective lens would produce a total magnification of A. 25x…
A: Option D is correct answer. Simply the total magnification is given by the product of magnification…
Q: -3 (e) Jika 0.1 mol dm asid hidroklorik digunakan untuk mentitrat 50.00 cm' larutan kalium…
A:
Q: He+ Al → n+ 2 13 01:23:26 Multiple Choice 16 31 14 to 15 3 14 30 15 Next> 20 of 35
A:
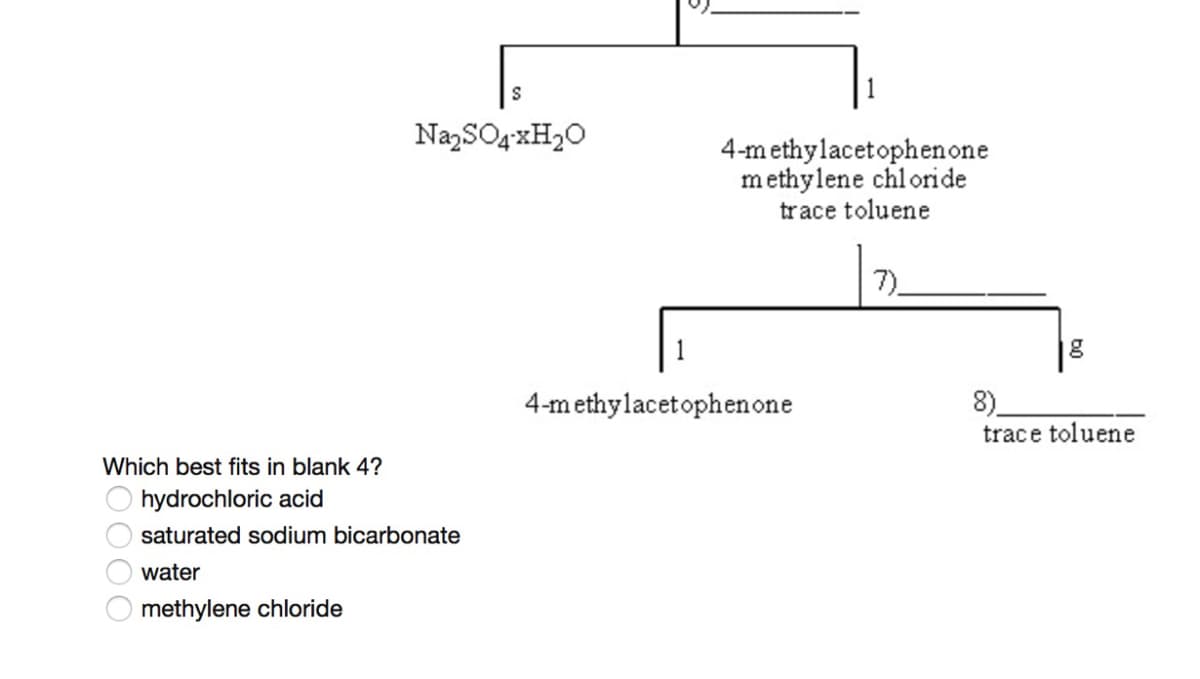
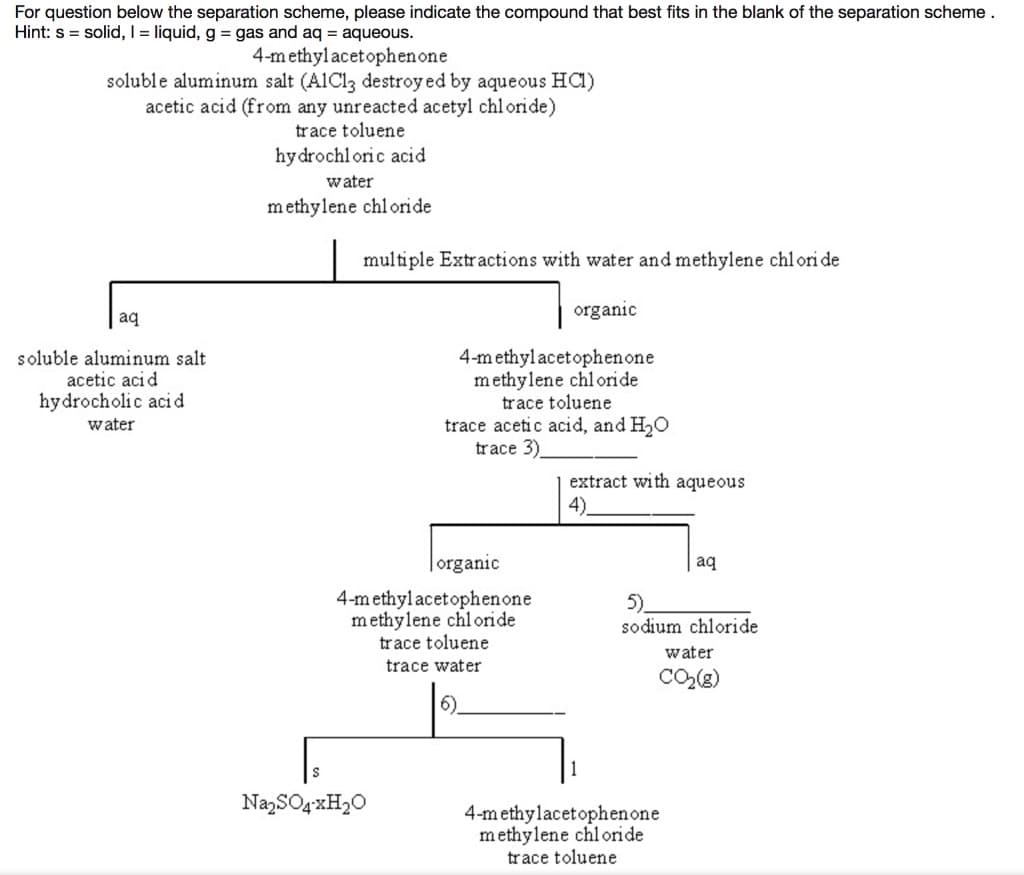
Q: Which option is a correct representation of the missing information in Figure 8? * 1- A…
A: In an organic reaction mechanism, a curved arrow represents the transfer of electrons. In an organic…
Q: TRUE OR FALSE When a star’s absolute and apparent magnitudes are known it becomes a standard…
A: True
Q: The trendline for a calibration curve is y=4.299x+-0.0001. The absorbance of a solution of the…
A: The concentration of the unknown solution is 0.0189 uM.
Q: A little change creates a big response reproducibility Sensitivity Selectivity Detection limit
A:
Q: 07 - Air Matte... CK 12 License Exploration Worksheet Page
A: The answer of the following questions are : 8- d. CH4 9- d. Nitrogen 10- a. Xenon,Xe 11- b.…
Q: 1. Name the three classifications of compound microscope. 2. Differentiate magnification and…
A:
Q: How should you read 3.50 m
A: To measure length, scales such as centimeter, meter, and kilometer are used. 1 kilometer is equal to…
Q: Regarding TLC plates: a) When spotting a TLC plate, are small spots or large spots better? Or does…
A: Ans Spots are not too large or too small. If spot is large then overlaps with other Components . So…
Q: Which of the following pairs is correctly matched? A hotplate : supplies flame B beaker : measures…
A: The options given are,
Q: Identify one pro and one con for each observation (visual and UV/Vis)
A: A question based on concentration terms that is to be accomplished.
Q: In this lab, you will construct a calibration curve using absorbance and concentration data. Which…
A: A calibration curve is: Standard graph which shows the change in the response of an analytical…
Q: enumerate the 12 safety hazard in the photo.
A:
Q: The TLC technique can be used to: a. Check the purity of a compound. b. Determine the number of…
A: To solve this problem we have to know about the use of TLC .
Q: products Select Draw Rings More Erase H +1 equiv OH¯ about us careers privacy policy terms of use…
A:
Q: You determine that your sample has a mass of 386 g and a volume of 20ml it also is fairly unreactive…
A: Mass of sample =386g Volume of sample = 20ml Density = Mass/volume =386/20…
Q: ( give explanation with diagram)
A:
Q: The length of one pencil is measured with a ruler, the following measurements are obtained; 5.49…
A: Arranging the obtained length in arranging order we get: 3.28 , 5.34 , 5.38 , 5.41 , 5.49 Finding…
Q: Modified True or False. Analyte interacts with UV –Vis radiation UV radiation is between 200-300 nm…
A:
Q: Discuss the importance of keeping a laboratory notebook (logbook).
A: Logbook is an important documentation of all the experimental progress in the lab in a systematic…
Q: Which of the following chemical formulas are NOT written correctly? (a) F3P (b)NH4+ (c) Fe(NO3)2…
A: The answer to the following question is-
Q: Prepare two graphs, %T vs. Concentration and Absorbance vs. Concentration, using the data for the…
A: According to Beer-Lambert law the amount of light absorbed or transmitted when passed through a…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- The separation and purification processes given below and the method used against them are given. Which or which of these pairings are correct? I. Brewing of tea - ExtractionII. Purification of water impurities - ChromatographyIII. Separating olive pomace from olive oil while producing olive oil-DecantationIV.Petroleum fractions obtaining - Ordinary distillationV. Obtaining essential oils and using them in perfume making - Water-steam distillationA. I, II, III, IVB. I, II, III, IV, VC. I, IV, VD. I, II, III, VE. I, II, VTransfer 3-4 drops or a pinch of the test compound (Acetanilide) to 3 ml of the solvents, 5% NaOH and 5% HCL Shake the mixture thoroughly. The time required for the solute to dissolve in the solvent should not be more than 2 minutes. Indicate with (+) or (-) if the test compound is soluble or insoluble in the solvent. What is the solubility class of Acetanilide in 5% NaOH? (Very soluble, freely soluble, soluble, sparingly soluble, slightly soluble, very slighlty soluble, practically insoluble) What is the solubility class of Acetanilde in 5% HCL? (Very soluble, freely soluble, soluble, sparingly soluble, slightly soluble, very slighlty soluble, practically insoluble)Which of the following is expected to achieve the best recovery of an organic compound from an aqueous solution? 1.Three extractions with 20 mL of ethyl acetate per extraction (60 mL total) 2.Two extractions with 30 mL of ethyl acetate per extraction (60 mL total). 3.One extraction with 60 mL of ethyl acetate. 4.In theory these extractions yield identical recoveries of the organic compound.
- Transfer 3-4 drops or a pinch of the test compound (Acetanilide) to 3 ml of the solvents, 5% NaOH and 5% HCL Shake the mixture thoroughly. The time required for the solute to dissolve in the solvent should not be more than 2 minutes. Indicate with (+) or (-) if the test compound is soluble or insoluble in the solvent. Is Acetanilide solube or insolube in 5% NaOH? _______ Is Acetanilde soluble or insolube in 5% HCL? _______1-A bottle of Na2EDTA.2H2O is labeled with an assay of 100.5%, what is this implied?What is the result of heating to 100 ºC?A mixture of pure BaCO3 and pure Na2CO3 weighs 1.000 g and has the total neutralizing power of 15.37 meq of CaCO3. Calculate the percentage of combined CO2 in the mixture and the weight of Li2CO3 that has the same neutralizing power as 1.000 g of the above mixture. Note: include up to 4 decimal places Please explain each step
- ALL INFORMATION NEEDED IS PROVIDED, I NEED FIRST QUESTIONS ANSWERED FIRST BEFORE OTHERS CAN BE ANSWEREDRate = -(Delta[RCl])/Delta t andln[RCl]t=-k*t+ln[RCl]0 < [R-Cl]-time equation Test tube A = 0.30 of 0.10M NaOH(aq), 6.70ml DI H2O & 1 drop of bromophenol bue Test Tube B = 3.00M of 0.10M R-CL in acetone. Mix together in 3 water baths Bath A Trial 1= 20C, 53.6s Trial 2= 20C, 49s for color to changeBath B Trial 1= 32C, 18s Trail 2= 32C, 20s Bath C Trial 1= 10C, 204s Trial B= 10C, 200s average out both trials for each bathcolor change happens after 10% of R-Cl has reacted so In terms of R-Cl this means that 10% of the R-Cl has reacted and ____ of the R-Cl is left Q1. at the time of the color change [R-Cl]t=____[R-Cl]o (Fill in the blank) Q4. Substitute the value of [R-Cl]t into appropriate equation listed above and solve for k in terms of [R-Cl] and t. Q5. Use average time for each temp and determine the value of k (with units) at each temperature Q6. Rearrange the Arrhenius…I just need help figuring out the values to do a calculation. Part of the lab relevant... "Prepare standards on the same day they will be used. Using a 100 mL volumetric flask, add x mL (vide infra) of the 0.050 mg/mL phosphate standard, 20 mL of th e molybdate-antimoney reagent, 1 mL of ascorbic acid, and dilute to volume with ultrapure water. Do this procedure for x = 0 mL, 1 ml, 2 ml, 5 ml, and 10 ml using volumetric pipettes. To be clear, we will be making 5 different solutions. The equation that was given was Concentration of initial standard (mg/mL) multiplied by volume added (mL) = concentration of created standard multiplied by volume of volumetric I dont need you to do the calculations, I just need help finding what values go in equation. we are trying to find the concentration of created standardTim is interested in analyzing for Pyraclostrobin (pesticide) in strawberries using QuECHers method for extraction and clean up before analysis with HPLC-uv. The concentration of the Pyraclostrobin in the fruits that he sampled is thought to be between 1-10 ng pesticide per g fruit fresh weight. The HPLC-uv instrument he is using has a LoQ of 5 ppb for Pyraclostrobin. Tim is starting out with 500 mg of the sliced fruit
- Please help The petition coeficiente between benzene and water for carta iLife is approximately 3.9 .you are given a 0.35 g sample of acetanilide dissolved in 100ml of water Which of the following extraction techniques will give q better recovery of.acetanilide from water A) one extraction with 60ml of benzene or b) two successive extractions with 30ml portions of benzene? Show calculationPlease type all of the parts to this question occordingly to the instructions. Here is an example of a formal procedure based off of the experiment presented in Mohrig:Sodium hydroxide (4.6 g) was dissolved in water (25 mL). This solution was added to a 100 mL round bottom flask along with methyl salicylate (2.0 mL) and a stir bar. The mixture was heated at reflux for 15 min and then progressively cooled to 5 °C. The solution was acidified with 3M sulfuric acid (15-20 mL), and the resulting solid was collected by vacuum filtration. The crude salicylic acid was recrystallized from water to yield ???? g of final solid. The melting point and IR were taken to assess purity. Calculate the molarity of the solution of sodium hydroxide that is used.a. M = b. If the original procedure is the base scale (100%), how far are we scaling down the reaction when using 0.75 mL of methyl salicylate? (Example: the original uses 2.0 mL, if we were to use 1.0 mL, we would be scaling the reaction down…The purpose of this experiment is to use both extraction and crystallization techniques to separate a solid mixture of organic compounds. You will receive a packet with 1 g of a unknown (pH is basic) amide solid sample which will contain three components, one from each group: Group 1 (~50% wt): ethyl 4-aminobenzoate, ortho-toluic acid, or benzoic acid Group 2 (~40% wt): fluorene, 1,4-dibromobenzene, benzil, benzoin Group 3 (~10% wt): fluorene, 1,4-dibromobenzene, benzil, benzoin (but not the same as Group 2 compound) Create a lab procedure that will allow you to successfully isolate, purify, and identify the two major (Group 1 and Group 2) components of the unknown amide sample. reagents that can be used are 1M HCl, 6M HCl, 1M NaOH, 6M NaOH, diethyl ether, and methylene chloride

