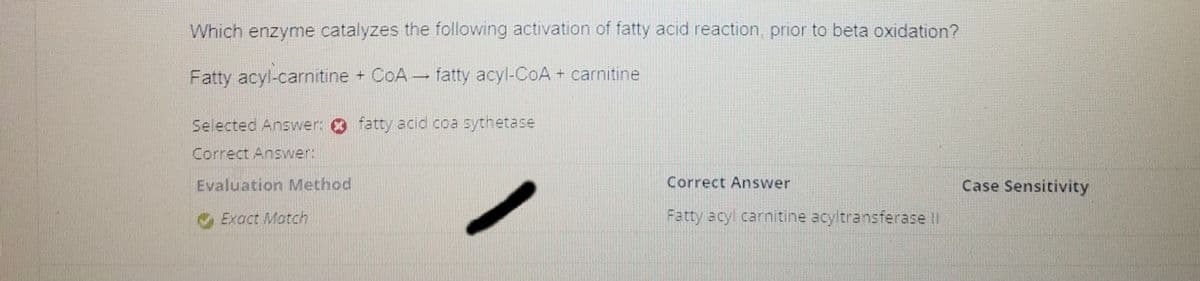
Which enzyme catalyzes the following activation of fatty acid reaction, prior to beta oxidation? Fatty acyl-carnitine + CoA fatty acyl-CoA + carnitine Selected Answer: fatty acid coa sythetase Correct Answer: Evaluation Method Exact Match Correct Answer Fatty acyl carnitine acyltransferase II Case Sensitivity
Q: The activation energy of an uncatalyzed reaction is 91 kJ/mol. The addition of a catalyst lowers the…
A: The rate of a chemical reaction is generally given by the Arrhenius equation:k = A *…
Q: Which of the following reactions required dihydrolipoyl dehydrogenase? Oxidative decarboxylation of…
A: In oxidative decarboxylation, the carboxyl group (-COO) is removed from the substrate in a redox…
Q: Which of the following metabolic processes require both biotin and nicotinamide? -Conversion of…
A: Enzymes catalyse biochemical reactions. Enzymes are proteins. Enzymes sometimes require non-protein…
Q: Explain the biochemical basis and clinical implications of atherosclerosis with molecular and…
A: Atherosclerosis is a chronic inflammatory disease characterized by the buildup of plaque within the…
Q: The kcat and KM for chymotrypsin-catalysed cleavage of a synthetic substrate, S, were determined to…
A: For a one-substrate enzyme-catalyzed reaction, the Michaelis-Menton equation shows the quantitative…
Q: Which of the following properties of lipids makes it hard to digest? (A) Their molecules need an…
A: Lipids are a group of biomolecules that includes oils, fats, steroids and phospholipids. It acts as…
Q: What is the parameter in the enzyme measurements?
A: Enzyme measurements provide various parameters which are used to quantify and characterize the…
Q: Under anaerobic conditions E. coli can use Nitrate (NO3) as an electron acceptor in a pathway to…
A: Before going into the final answer, let me explain to you the significance of redox loops in…
Q: 61. Which pairs of restriction enzymes would allow the cloning of the HGH cDNA 3' (downstream) of…
A: Nucleases are enzymes that degrade nucleic acids. They can be classified intoRNase : enzymes that…
Q: 165. Explain why the direction of movement of fluid in the manometer was different during yeast…
A: Respiration, the fundamental process by which living organisms generate energy, has revealed…
Q: Which of the proteins: the CAP or the lac repressor, does the eukaryotic nuclear receptor for the…
A: There are steroid hormones (compounds with characteristic sterol rings). Steroid hormones can be…
Q: 40. Propose that all three carbon atoms in glycerol is labeled with 14C. The labeled glycerol is…
A: Glycerol is a molecule commonly found in biological systems and serves as a precursor for various…
Q: Why does FAHD2 oxidation give 1.5 ATP while NADH oxidation gives 2.5 ATP? Explain this in relation…
A: The differing entrance locations of NADH and FADH2 at which they contribute electrons in ETC,…
Q: 45. Choose between the light dependent reaction or the Calvin cycle steps of photosynthesis.
A: Plants, algae, and some bacteria convert sunlight into chemical energy through photosynthesis. These…
Q: 5. Which of the following statements is true for the shown reaction? HS-COA CH₂ LAE A B COOH Cool…
A: The product of glycolysis, pyruvate is converted to acetyl CoA. Acetyl CoA enters the cycle by…
Q: When one categorizes a protein's structural classification, they sort different proteins primarily…
A: Amino acids are defined as organic compounds that form the core structure of proteins and gives them…
Q: 27. Explain the full signal transduction mechanism for intracellular-mediated responses (the actions…
A: Lipophilic hormones (steroid or thyroid hormones) signal intracellular-mediated responses.…
Q: Using the amino acids glyceric acid and aspartic acid show how a peptide bond is formed showing the…
A: Before going into the solution, we need to point out an error in the question. Glyceric acid is not…
Q: Match the description on the left with the terms on the right. A component of cytochromes Exist in…
A: Both hemoglobin (Hb) & myoglobin (Mb) are globular proteins.Hemoglobin help to transport oxygen…
Q: The bonds that are important inthe secondary structure of a protein are a)hydrogen bonds…
A: Proteins are complex biomolecules composed of long chains of amino acids that are linked together…
Q: Shown below is a common lipid. To which of the following lipid groups does this molecule belong? CH₂…
A: It is asked to find the type of lipid groups of the given molecule.Lipids: Lipids are fatty acids…
Q: Which of the following are true about the glyoxylate cycle? (select all that apply) Group of answer…
A: 1.Certain organisms have a metabolic mechanism called the glyoxylate cycle that helps them meet…
Q: 852. what is the difference between maltose maltase, sucrose-sucrase and lactoselactase?
A: The body uses carbohydrates for energy. From simple sugars to complex starches, they are found in…
Q: Table 2 Absorbance vs CoCl2 concentration Test Tube Number Cobalt Chloride Concentration…
A: Spectrophotometry or colorimetry is an analytical technique that is used for the quantification of…
Q: iii) Draw a structural diagram of a hydrogen bond between β-D-glucose and the side chain of any…
A: iii) Firstly we need to understand the polar residues of a protein. The polar residues of a protein…
Q: Match each stated goal to the most relevant method, technique, or procedure for achieving that goal.…
A: Separation of proteins is the process of isolating and purifying individual proteins from a mixture…
Q: BIOCHEMISTRY Why glycogen in muscle cells cannot be stained blue-black by iodine solution?
A: Muscles store glucose as glycogen. Iodine solution stains starch, another glucose storage molecule,…
Q: List two denaturants and one reducing agent that are commonly used in protein chemistry.
A: Denaturants interacts with the non-covalent interactions within the native structure of proteins…
Q: Which of the following factors do not stabilize the 3-D structure of a folded protein? O Disulfide…
A: The total 3-D configuration of a protein's secondary structures and other components is referred to…
Q: A specialized lysine-linked co-enzyme often participates in the transfer of carbon fragments within…
A: The small non-protein organic molecules which work along with enzymes in various catalytic reactions…
Q: Draw tripeptide Lys-Thr-Glu at pH 7.4 and circle the six atoms that are restricted in the same…
A: Amino acid is a biomolecule that contains both amino and carboxylic acid…
Q: In general, when the nonbicarbonate buffer concentration is normal, an acute PaCO2decrease of 10 mm…
A: The scenario provided states that the non-bicarbonate buffer concentration is normal, and there is…
Q: Much of the stability of the double-stranded DNA structure is the result of.... A. the angle of…
A: Nucleic acid can be of 2 types:DNA (deoxyribonucleic acid)RNA (ribonucleic acid)Nucleic acids are…
Q: • The Predator's Mb has a higher affinity for O₂ than the Alien's Mb O True False
A: Ans- falseAs the myoglobin curve for Alien achieves P50 much before the predator, it means Mb of…
Q: BIOCHEMISTRY Cell culture has become one of the most fundamental techniques for modeling…
A: Several methods can detect protein expression. Here are some common methods with pros and…
Q: Based on your knowledge of how living organisms can do this chemistry list the cofactor or subclass…
A: Fatty acids metabolism involves β-oxidation. In β-oxidation, the fatty acids are oxidized on the β…
Q: Lipid bilayers comprising high concentrations of gangliosides are very rare. Which of the following…
A: A lipid bilayer is a fundamental structural component of cell membranes in living organisms. It is…
Q: Which of the following statements is true? a. Plotting the rate of facilitated transport against…
A: Facilitated transport, also known as facilitated diffusion, is a mechanism of passive transport that…
Q: Cyanogen bromide is a common sulfhydryl-modifying reagent that can generally be used to break…
A: The amino acids in a protein are linked by various linkages such as amide, hydrogen bonding, sulfide…
Q: Which of the following is NOT part of the Q cycle? O coQH₂ transfers two electrons to Complex III O…
A: Coenzyme Q or ubiquinone or CoQ is an electron carrier that accepts electrons from NADH and FADH2,…
Q: In the clinical case scenario provided, match the defective enzyme in Column A with the consequence…
A: Defective enzymes leads to compromised metabolism within individuals. This would lead to health…
Q: The Hill coefficients for NAD+ binding to the T. tenax GAPDH in the presence and absence of NADP+…
A: Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) can be either phosphorylating or…
Q: Which methodologies can be used to detect the expression of a given protein? Please provide pros and…
A: As per the central dogma of molecular biology, the information on protein synthesis is stored in the…
Q: Biosynthesis of fatty acid 20:D6 from acetyl-CoA occurs in the __________ of mammalian cells.…
A: Fatty acids are carboxylic acids with a hydrocarbon chain ranging from 4 carbon to 36 carbons. Based…
Q: Below is an image of how biotin is used in the reaction that converts pyruvate to oxaloacetate, an…
A: Biotin, which is also referred to as vitamin B7 or vitamin H, is a water-soluble vitamin categorized…
Q: What kinds of forces hold the double strands of a DNA double helix together?
A: The double strand of DNA, often referred to as a double helix, is a fundamental structural feature…
Q: BIOCHEMISTRY Most important nucleophiles in the cell?
A: Several nucleophiles—substances that donate an electron pair to form a chemical bond—are important…
Q: 68. Describe the basic biomolecularstructure of the 5 chemical classes of chemical messengers. Know…
A: Chemical messengers help regulate cell and tissue communication. They help cells communicate,…
Q: Draw one example of an interaction the enzyme could provide that would stabilize this transition…
A: Enzyme catalyzed reaction proceeds through transition state(s). A transition state is a very short…
Q: 5. Given the following TLC and melting point data for three unknown samples sample melting point…
A: Title: Comparative Analysis of Unknown Compounds through Melting Point and TLC Techniques:Chemical…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

- Long explanations are not needed. Direct answers would suffice. a. Ribose-5-phosphate is produced by oxidative decarboxylation of 6-phosphogluconate catalyzed by the enzyme glucose-6-phosphate dehydrogenase. I. True II. False b. The pentose phosphate pathway occurs in the mitochondrion of tissues actively engaged in synthesis of fatty acids. I. True II. FalseA patient who has been drinking large amounts of alcohol for long periods of time shows thefollowing symptoms: apathy, loss of memory, and a rhythmical to-and-fro motion of the eyeballs.Which of the following reactions are most likely to be affected in the patient? A. Conversation of pyruvate to acetyl-CoA B. Conversation of a-ketoglutarate to succinyl-CoA C. Both A and B D. Neither A nor BLong explanations are not needed. Direct answers would suffice. a. High concentration of glucose 6-phosphate is inhibitory to the enzyme hexokinase. I. True II. False b. Which compound is formed when NAH reduces pyruvate (anaerobic condition)? I. lactate II. alpha-ketoglutarate III. glucose IV. oxaloacetate
- Under aerobic conditions of high ratios of NADH/NAD+ and ATP/ADP, as pyruvate is utilized for its carbon skeleton, which molecules would you expect to see significant radiolabeling in the liver? Select all that apply. **Please note some molecules contain more details, including not only molecule name, but location of the label. Pick the options that are accurate for the above situation. 1. Glucose C2 & C5 2. Glucose C1 & C6 3. Glucose C2 only 4. Pyruvate C1 5. Lactate C2 for export 6.CO2 from TCA cycle shows some radiolabel 7. Label is halved over many TCA cycles 8. Oxaloacetate 9. MalateIndicate what will happen ( increase, decrease or no effect) tothe activity of the enzyme or rate of the metabolic pathway in the given conditions a. release of glucagon in the blood to the activity of carnitine acyl transferase 1 b. high malonyl CoA to the activity of carnitine acyl transferase I C. Epinephrine to the activity og glycogen synthase d. high citrate to the activity of acetyl CoA carboxylase e. high acetyl CoA to ketogenesisIn relation to Carbamoyl Phosphate Synthetase enzyme, answer the following: A- What are the two isoforms of this enzyme, explain why there are two isoforms? B- What are the clinical manifestations associated with the deficiency of these two enzymes? C- Write down the biochemical reaction and the name of the metabolic pathway that these two isoforms are involved in, and how many ATP is utilized by these two isoforms?
- How many more acetyl CoA are generated from stearic acid than from linoleic acid during beta oxidation? Enter numerical answer onlyName some HMG CoA reductase inhibitors? Please answer at your own words.What is(are) the only active site(s) not used in the second round of fatty acid synthase? Select all that apply. Group of answer choices Acetyl-CoA ACP Transacylase Beta-Ketoacyl- ACP Synthase Beta-Ketoacyl- ACP Dehydrase Palmitoyl thioesterase Malonyl-CoA ACP Transacylase Enoyl-ACP Reductase
- In the context of fatty acid oxidation to acetyl CoA, indicate whether each of the following substances is involved in (1) fatty acid activation, (2) fatty acid trans-port, or (3) b-oxidation pathway. More than one choice may be correct in a given situation. a. AMp b. FAD c. Acyl CoA d. H2OWhat enzyme is inhibited by Acetyl-CoA carboxylase? a)Acetyl-CoA carboxylase b)Citrate lyase c)E3 ubiquitin ligase d)Carnitine acyltransferase IEnzyme Triacylglycerol Lipase Rennin Catalase Hexokinase Enzyme Official Name (write N/A if not applicable) Enzyme Official Number (4 digits) Simple or Conjugated Enzyme (specify cofactor if applicable) Type of Reaction Catalyzed Substrate Optimum pH Optimum Temperature Function/s Disease (give 1 and describe briefly)

