Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
ChapterNW4: Nomenclature Worksheet 4: Carbonyl Compounds
Section: Chapter Questions
Problem 31CTQ
Related questions
Question
For each factor (on the left list) u can choose one of the three options (on the right). U can choose each option as many times as u like
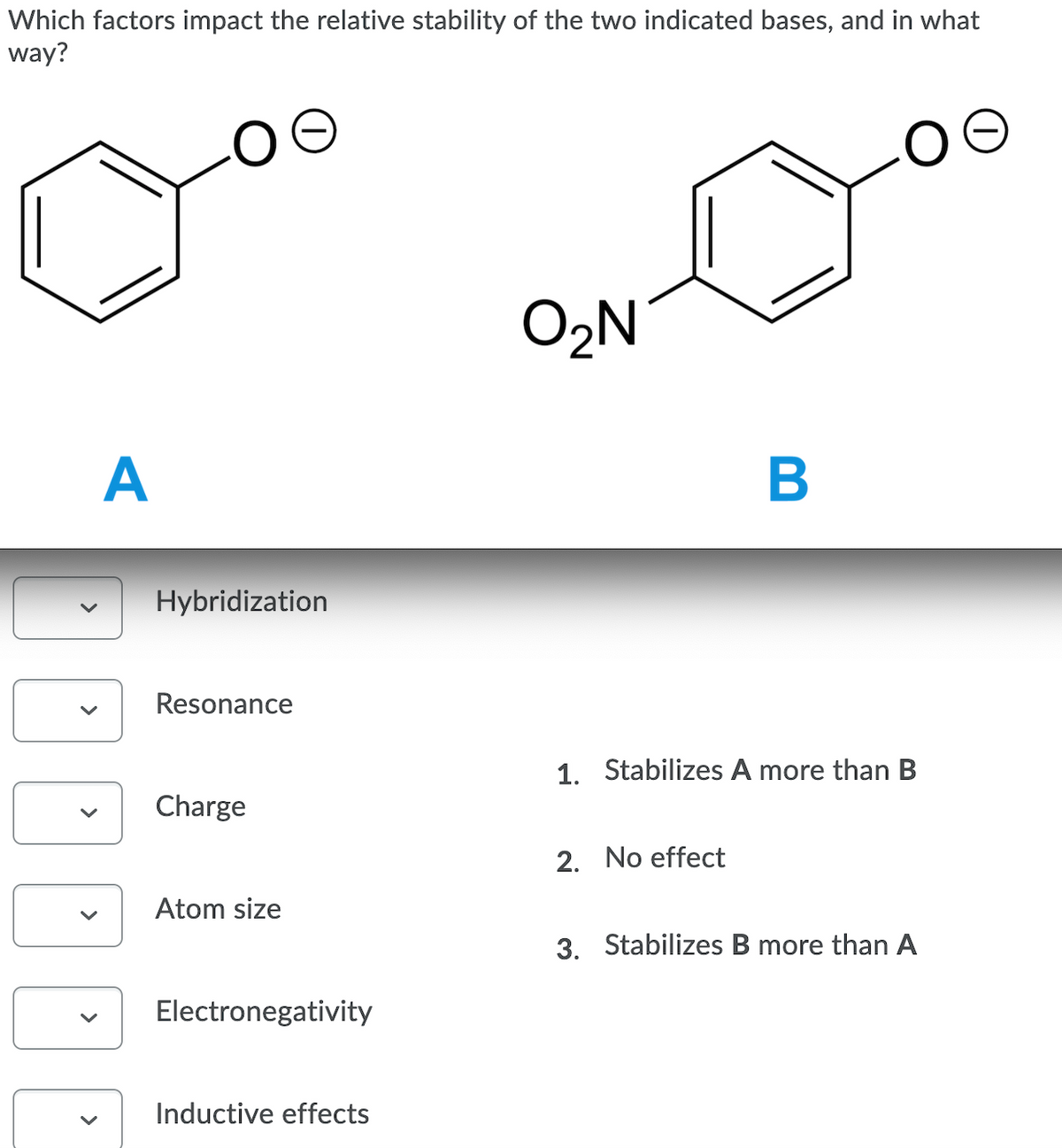
Transcribed Image Text:Which factors impact the relative stability of the two indicated bases, and in what
way?
O2N
A
В
Hybridization
Resonance
1. Stabilizes A more than B
Charge
2. No effect
Atom size
3. Stabilizes B more than A
Electronegativity
Inductive effects
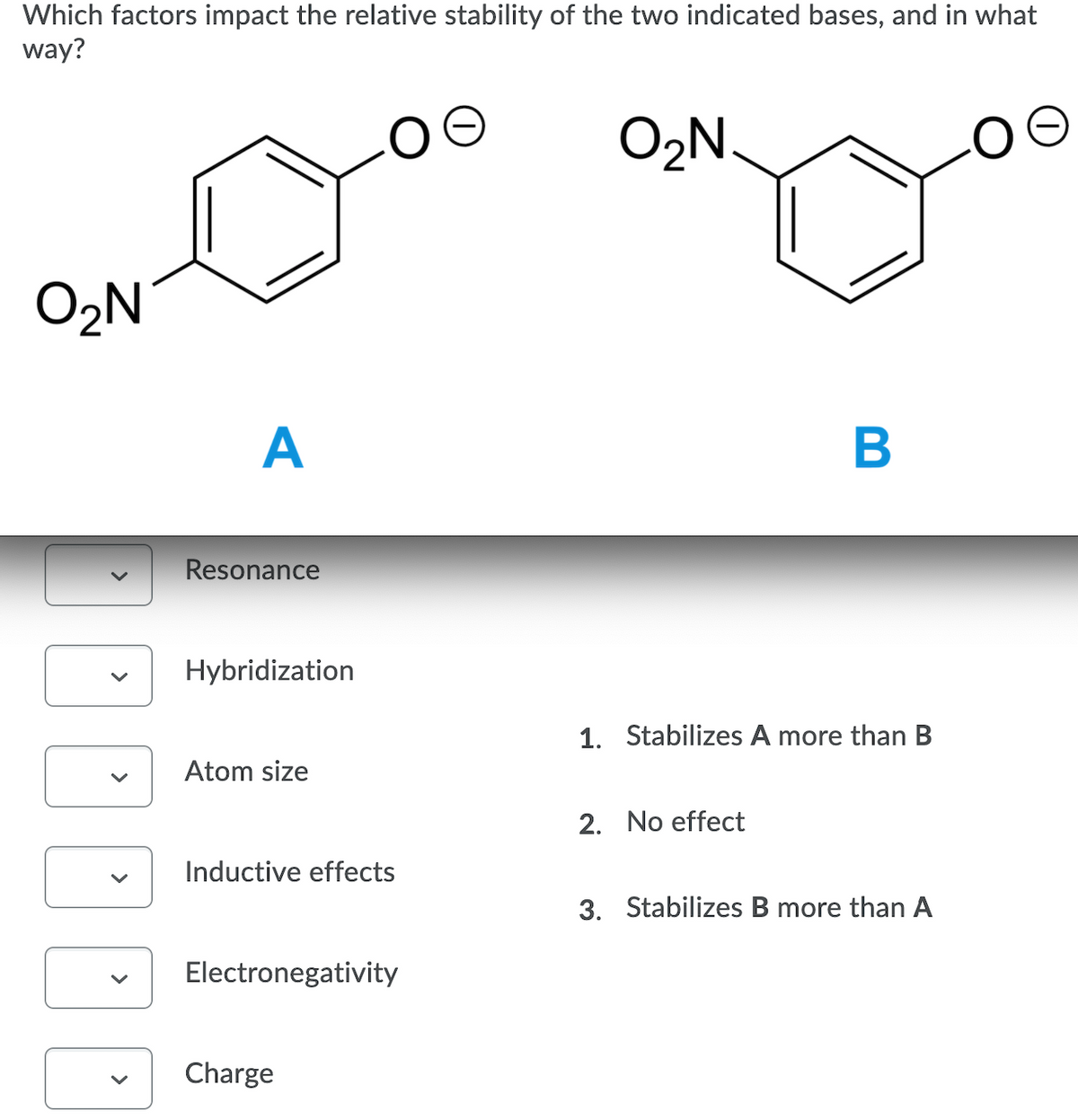
Transcribed Image Text:Which factors impact the relative stability of the two indicated bases, and in what
way?
O2N.
O2N
A
Resonance
Hybridization
1. Stabilizes A more than B
Atom size
2. No effect
Inductive effects
3. Stabilizes B more than A
Electronegativity
Charge
>
>
>
<>
>
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 2 images

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning
