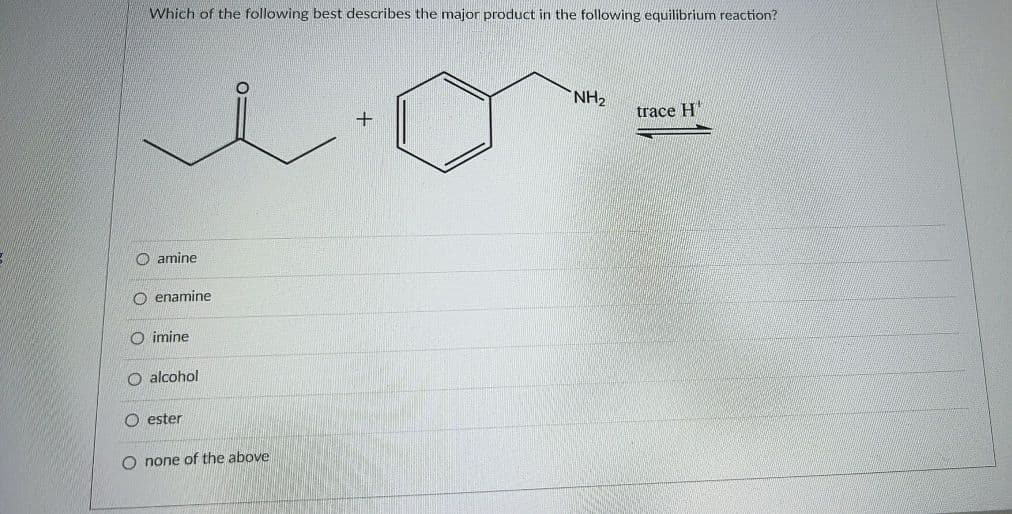
Which of the following best describes the major product in the following equilibrium reaction? NH2 trace H" O amine O enamine O imine O alcohol O ester none of the above
Q: A chemist determined by measurements that 0.0600 moles of gold participated in a chemical reaction. ...
A: • The values provided in the question are:- i) Number of moles of gold participated in a chemic...
Q: If the pKa of a weak acid HA is 3.84, what is the pkb of its conjugate base A-
A: Given Data : pKa = 3.84 To Find : pKb
Q: 46. Which of the following bonds is least polar? a) C-O b) H-C c) P Cl d) Na-Cl
A: More the electronegativity difference, more is the polarity.
Q: Calculate the pH of the following solutions. - 0.10 M pyridine
A:
Q: An environmental chemist analyzed the effluent (the released waste material) from an industrial proc...
A: Given : Volume of sample = 3.52 ml Volume of NaOH solution = 10.59 ml Concentration of NaOH solut...
Q: Types of corrosion are. Nine. Eight. Seven.
A: Types of corrosion are given below.
Q: write the rate law for the following reactions. (assume each reactions follow an elementary rate law...
A:
Q: Finding mole ratios from chemical formulae This is the chemical formula for methyl tert-butyl ether ...
A:
Q: The following molarities were calculated from replicate standardization of a solution: 0.5022, 0.502...
A: Given data :- Set of molarities calculated from replicate standardization of a solution are : 0.5022...
Q: 3-D contour surfaces
A: In this question we have to draw the 3-D contour for a 4p orbital.
Q: A chemist determined by measurements that 0.045 moles of calcium participated in a chemical reaction...
A: Mole can be defined as a standard unit for measuring large quantities of very small entities such as...
Q: A mixture of gaseous reactants is put into a cylinder, where a chemical reaction turns them into gas...
A: Given data :- Amount of energy absorbed = 393 KJ Work done by the piston on the system = 194 KJ
Q: Reaction of 2-butanone with CH3MBBR/ether followed by H3O* gives a, 4° alcohol O 1° alcohol O 3° alc...
A:
Q: Give ONE (1) example each for natural and synthetic polymers.
A: Natural polymers occur in nature and can be extracted. They are often water-based. Synthetic Polyme...
Q: 89% copper oxide What is the
A: Given percentage of Copper Cu=89% Given percentage of oxygen=(100-89)=11%
Q: (Q100) Consider the hypothetical reaction, which has an equilibrium constant (K,) of 15.3 when the r...
A: 2W(g) + X(g) ------->3Y(g) Kp =PY3/(PW2.PX) ...(1) Kp = equilibrium constant = 15.3 PY = parti...
Q: Need the mechanical properties of the polymers
A: The polymer is a larger macro-structure that is composed of smaller molecular units repeated over a ...
Q: Sulfur dioxide and oxygen react to form sulfur trioxide, like this: 2 SO,(9)+O2(g) –→2 SO3(g) The re...
A: Recall the given reaction, 2 SO2 (g) + O2 (g) 2 SO3 (g) is an exothermic...
Q: Br KCN Select one: Primary alkyl halide; SN1 reaction mechanism. Primary alkyl halide; SN2 reaction ...
A:
Q: Gaseous butane (CH;(CH,) CH,) reacts with gaseous oxygen gas (0,) to produce gaseous carbon dioxide ...
A:
Q: Sodium bicarbonate is added to water and allowed to come to equilibrium according to the reaction be...
A: Given reaction : NaHCO3- (aq) + H2O (l) →H2CO3 (aq) + NaOH (aq) Give that, At equilibrium, pOH = 12....
Q: Calculate the mass of Kr in a 8.57 L cylinder at 96.6 °C and 4.48 bar. mass: g
A:
Q: What is a protecting group in a multi synthesis reaction in organic chemistry
A: Protecting groups are used in synthesis to temporarily mask the characteristic chemistry of a functi...
Q: The thermochemical equation which is associated with AH, the standard enthalpy of formation for acet...
A:
Q: Calculate the equilibrium concentrations of all relevant C species if an action releases methane (CH...
A: We have to determine the value of equilibrium concentrations of all relavent C species if an action ...
Q: ryour units!) 2. 500.0 liters of a gas are prepared at 700.0 mm Hg and 200.0 °C. The gas is placed i...
A:
Q: Kindly complete the table below and show how it was done. Final answers must be reported in proper u...
A: 25.3 mL of glacial acetic acid is dissolved in water to form a 50 mL solution. Here, we have to ...
Q: .Titrant is usually in In a conical flask. In beaker. O
A: burette
Q: What is the IUPAC name for the following compound?
A: IUPAC rules: Choose the longest chain rule While numbering functional group (ketone) should ge...
Q: Compute the sulfide-lon concentration of a 0.5F HC2H3O2 SOIUTion which has been saturated with H2S a...
A: Answer and explanation is given below hope you like it.
Q: The amount of iron in ore can be quantitatively determined by titrating a solution of the unknown wi...
A: Given values -> Volume of iron(3) = 25.0ml (V1) Volume of dicromate =18.0ml (V2) Molarity of dicr...
Q: Calculate the concentration of all ions present in each of the following solutions of strong electro...
A:
Q: When an equilibrium mixture of A(aq) and B(ag) is placed in a hot water bath, the solution turns blu...
A: Le Chatelier's principle: This principle is used to predict the effect of a change in conditions on ...
Q: After completing the experiment, a student finds that the equilibrium concentrations of the reactant...
A:
Q: The initial enrichment of uranium in LWRS is 3% and the final (at discharge after 3 years in the cor...
A:
Q: Question 8 0. Predict a possible product (if one is possible) for the following Friedel-Crafts Alkyl...
A: Given reaction is intramolecular Fridel craft reaction.
Q: Determine the pH of the solution given after the addition of the common ion. The Kw of water is 1.0 ...
A: NH4Cl ----> NH4+ + Cl- 0.25 M ...
Q: Write the pressure equilibrium constant expression for this reaction. 2 CH;OH(1)+3 O,(9)→2 CO,(g)+4H...
A:
Q: Draw the condensed formula or skeletal structural formula for the following compounds: (a) 2-methyl...
A: (a) 2-methylpentanoic acid (b) Ethyl butanoate
Q: (b) Calculate Amax for the following compounds. () (i)
A:
Q: Write the formulas for the following compounds.
A: Molecular compounds are named with the first element first and then the second element by using the ...
Q: Predict the products of each reaction below. Indicate regiochemistry and stereochemistry when releva...
A: Kmno4 is strong oxidising agent. Over oxidation takes place.
Q: blö 5 How many grams of Na3PO4 required * ?to prepare 8 N in 500 ml 323.6 gm. 218.6 gm. O 656 gm. O
A: Given that , Normality of the solution = 8N Volume of solution is = 500 ml. we have to calculate how...
Q: At a certain temperature, the equilibrium constant K for the following reaction is 1.3: CL(e) + CHCI...
A:
Q: Part A Nitric oxide reacts with chlorine gas according to the following reaction: 2 NO (g) + Cl2 (g)...
A: The given chemical reaction is as equilibrium and the reaction is as follows: 2NOg + Cl2g ⇌ 2NOClg A...
Q: Decide how the objects in the following table would be listed, if they were listed in order of incre...
A: Please find your solution below : Kinetic energy is the energy of motion. It is a form of energy tha...
Q: What is the pOH of a buffer made of 1.7E-1 M (CH3)3N(aq) and 5.4E-1 M (CH3)3NHCl(aq)? K for (CH3)3N ...
A:
Q: Synthesize the folowng compnd stavting fom bezere (3 (A) (B) CHs Hz N
A: Organic reaction mechanisms:
Q: Draw the condensed structural formula or skeletal formula of the hemiacetal and acetal products form...
A: Hemiacetal and acetal are protected forms of aldehyde carbonyl group by reaction with alcohol i...
Q: Which one hardness type that remain after boiling process. * Temporary hardness. O Permanent hardnes...
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Which reagents are best used to produce 4-methyl-2-pentanol from a ketone? a. amins and trace acid, followed separetely H2/PD/C b. sodium cyanide followed by HCl c. sodium borohydridd followed by acidic work up d. hydrogen cyanide followed by HCl e. none of the abovewhich of the following compounds shows the weakest acidity A-ch3oh B-(ch3)3coh C-h2o D-h2co3Predict the major products of the following reactions. p@methylanisole + acetyl chloride + AlCl3
- Predict the major products of the following reactions. (a) the tosylate of cyclohexylmethanol + excess NH3(b) n-butyl tosylate + sodium acetylide, H¬C‚C:- +NaChoose the answer that best represents the products of the following reaction. Potassium carbonate is reacted with phosphoric acid. A. 2 K3PO4 (aq) + 3 H2O (l) + 3 CO2 (g) B. K2CO3 + H3PO4 C. 3 K2CO3 + 2 H3PO4 D. 2 K3PO4 (aq) + 3 H2CO3 (aq)B, we saw how acetylide ions add to carbonyl groups in much the sameway as Grignard and organolithium reagents. Acetylide ions also add to ethylene oxidemuch like Grignard and organolithium reagents. Predict the products obtained by addingthe following acetylide ions to ethylene oxide, followed by a dilute acid CH3CH2¬C‚C≠-
- Choose the best reagents to complete the following reaction. There's another answer choice not shown on screen: E) 1. CH3Li 2. H3O+ Hint: The correct answer is not option C.Identify the best reagents to console the following reactionWhich substrate will you use to synthesize 1-propanol from propanal? A.) NaBH₄ B.) O₃ C.) KMnO₄ D.) CH₃MgCl E.) None of the given answer
- Benzillic Acid lab Q1. Show how to prepare the following compounds, starting from the appropriate ketone1) LiAlH4; ) H3O+ will do which of the following reactions? 3-Phenyl-butanoic acid → 3-phenyl-butan-1-ol Benzene → cyclohexane Benzoic acid → benzaldehyde 2-Phenyl-hexanoic acid → benzoic acid and carbon dioxide 2,3-dimethyl-pent-1-yne → 2,3-dimethyl-pent-1-enePredict the major products of the following reactions. p@nitrotoluene + Zn + dilute HCl

