Q: 1. (a) Draw an energy cycle and use the Standard Enthalpies of combustion values in the table below ...
A:
Q: Describe the formation of metal complex with octahedral geometry using all the orbitals of same ener...
A: The formation of an octahedral complex using the orbitals of same energy level. Since you have as...
Q: Theoretical yield
A:
Q: The peptide below is placed into a buffer with a pH = 1.54 -NH-CH-C HO- H;N-CH- CH2 -NH -CH-C -NH-CH...
A: We have to find out the direction of movement of peptide at pH=1.54
Q: Is the gas temperature inside the syringe the same as the temperature of the water bath used in the ...
A:
Q: What is the pH of a 0.10 M solution of NaCN? The Ka of HCN is 4.9×10–10.
A:
Q: the Calculate the AHn for this reaction using CH.(g) + 2 H,O(g)→ 4H2(g) + CO2(g) Bond Energy (kJ/mol...
A: CH4 + 2H2O ->4H2 + CO2
Q: Answer the following questions about the energy profile diagram below. Reaction Progress Number of e...
A:
Q: Determine the ionic strength, 4, for each of the solutions. Assume complete dissociation of cach sal...
A: The ionic strength can be calculated as follows
Q: A chemist is studying the following equilibirum, which has the given equilibrium constant at a certa...
A:
Q: Combined Gas Law 1. A gas has a volume of 800.0 mL at-23.00 C and 300.0 torr. What would the volume ...
A: Given, The volume of a gas at -23.00 °C and 300.0 torr is 800.0 mL and the volume of a gas at 227....
Q: The concentrations of enzymes in cells are usually quite small. What is the biological significance ...
A: The enzymes are used in biological reactions. These biological reactions are taking place in the liv...
Q: A brick has a mass of 4.0 kg and the Earth has a mass of 6.0 x 1o g. Use this information to answer ...
A: Please find your solution below : The mole is a quantity that defines the amount of a substance and ...
Q: Identify the best reagents to complete the following reaction.
A:
Q: Write the corect Lewis structure and assign a formal charge to each atom in the fulminate ion, (CNO)...
A: Given :- formula of fulminate ion = (CNO)- To write :- correct lewis structure of fulminate ion and...
Q: a. Write the electron configuration for Rb*. (Express your answer as a series of orbitals, in order ...
A:
Q: The monohydrogen phosphate ion HPO3-2 has a Pka of 12. One could best describe this ion as a a) very...
A: Given :- pKa for HPO3-2 = 12 To identify :- nature of ion i.e. a) very strong acid or b) moder...
Q: A solution contains 1.47x10-2 M lead nitrate and 1.01x102 M silver acetate. Solid sodlum lodide is a...
A: Answer: This question is based on the concept of solubility product, according to which a compound s...
Q: Sucralose, an artificial sweetener commonly used by diabetic patients, has a formula of C12H19Cl3O8....
A:
Q: Directions: For each of the following reactions, indicate whether you would expect the entropy of th...
A: Please find your solution below : Entropy is a measure of dispersal of energy in a system. It is def...
Q: Match the most likely product with each reaction vPhenol plus excess 1/2 iodine in HNO3 yields A. 2,...
A:
Q: 5. A container containing 30.0 g of propane, C3H8, is burned with 75.0 g of oxygen. a. Outline the s...
A:
Q: What are the Safety Measures/ Precautions in the Science laboratory and classroom while doing a chem...
A: Science can be defined as the application of knowledge of natural world using a methodology which ...
Q: Which of the following 1.00 mole of fossil fuel when burned (combustion in open atmospheres) will ha...
A: Work done by any system or on the system can be calculated by the formula , w = -P∆V
Q: ng kinetic energy
A:
Q: The compound potassium cyanide is a strong electrolyte. Write the reaction when solid potassium cyan...
A: When strong electrolyte is added to water , it breaks into its corresponding ions . equation is give...
Q: entify reagents that can be used to accomplish the following transformation: 5-8 OH Ме Он he transfo...
A: We have given alcohol which is cyclopentanol and we have to synthesis 1-methylcyclopentanol.
Q: ewis structures shown contains one polar covalent bond. Identify the bond, and use th olarity.
A: Polar covalent bond is formed when atoms having different electronegativity share electrons in a cov...
Q: 1point You got a new lego set on your birthday and you build an air plane. The change in the entropy...
A: 3 1 pont You got a new lego set on your birthday and you build an air plane. The change in the entro...
Q: 0.035x10-mol M of Ca2+. 0.012M 2.92x10-3L 0.07x10-mol Calculated Ksp 0.069x103 0.063x103 0.053x103 M...
A: % error can never be negative as it is the mode value i.e., negative is not considered. % error = | ...
Q: write the ionization reaction for each of these KOH CH3NH2
A: To write the ionization reaction for each of these 1. KOH 2. CH3NH2
Q: Draw the major product of the reaction shown below. Ignore inorganic byproducts. ОН 1. КОН 2. (CH3)2...
A:
Q: thermometer A 50.8 g sample of aluminum is put into a calorimeter (see sketch at right) that contain...
A: Given: Mass of Aluminium = 50.8 gm Mass of water = 300 gm Initial temperature of water = 21.0°C ...
Q: Which of the following would have the greatest dipole? a) Cl-Br 47. b) N-F c) 0-F d) P-S
A: • The given bonds are- Cl-Br , N-F , O-F and P-S • We need to determine which bond has gr...
Q: The Lewis structure of the molecule, NH,O*, contains a) b) c) d) 1 double bond and 4 single bonds 5 ...
A: The Lewis structure of a molecule describes the distribution of valence shell electrons around each ...
Q: Solve only letter A. Please show the complete solution clearly.
A:
Q: What is the standard enthalpy of reaction per mole of either HCl or NaOH when 50.00 cm3 of 0.1 moldm...
A:
Q: Is the gas temperature inside the syringe the same as the temperature of the water bath used in the ...
A: In the given problem we are provide with the temperature of a water bath which is in contact with a ...
Q: A mixture of xenon and neon gas is compressed from a volume of 100.0 L to a volume of 99.0 L, while ...
A:
Q: Calculate the mass of camphor (C1,H1,0) that contains a billion (1.000 x 10°) carbon atoms. Be sure ...
A: We have to calculate the mass of camphor (C10H16O) containing 109 carbon atoms.
Q: Calculate the mass of diphosphorus pentoxide (P,0,) that contains a million (1.000 x 10°) phosphorus...
A: Given that, no. of P atoms= 1.000*106 atoms mass of P2O5=? Molar mass of P2O5=283.889 g mol-1
Q: i) BH3, THF i) H,О,NaOH ? CH3 CH3 -CH3 FHO- он -CH3 HO + + enantiomer enantiomer I II III IV
A:
Q: The combustion of propane may be described by the chemical equation C3H8(g)+5O2(g)⟶3CO2(g)+4H2O(...
A: Balance chemical equation : C3H8(g)+5O2(g)⟶3CO2(g)+4H2O(g) Given data : Mass of propane = 14.7 g ...
Q: Explain the process of isolation and characterization of Enalapril in tablets for the commercial dru...
A: Given:- Explain the process of isolation and characterization of Enalapril in tablets for the commer...
Q: Calculate the pH of a 0.859 M NaF solution. Assume Ka(HF) = 1.0 x 10-4
A:
Q: What is the corresponding heat required if 324.0 grams of CaCO3(s) is to be decomposed? Leave answer...
A:
Q: A mixture of xenon and hydrogen gas is compressed from a volume of 66.0 L to a volume of 54.0 L, whi...
A:
Q: Ammonia (NH3) chemically reacts with oxygen gas (02) to produce nitric oxide (NO) and water (H,O). W...
A:
Q: Name the following compounds: 1.[Cr(NH3)6Cl3 , K(Co(EDTA)] and Ni(en)2Cl2 2.[Pt(py)4[PtCl4] and [...
A: Rules to follow in naming transition metal complexes: - If the complex is a salt, the cation is name...
Q: shows The NMR brum abueaption Sinyle Coela live at - 21°c but in tensity :3) at - |18°c explonatien
A: the given problem in based on inorganic NMR
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

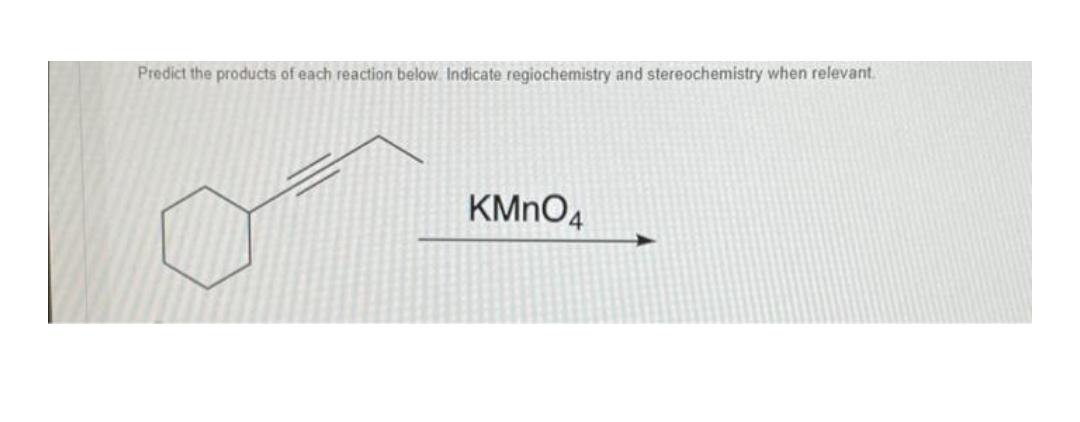
- Predict the products of each reaction below. Show stereochemistry where necessary.Predict the major products of the following reactions, including stereochemistry where appropriate. cyclooctanol + NaOCl>HOAc (d) cyclopentylmethanol + CrO3 # pyridine # HClPredict the major products of the following reactions, including stereochemistry where appropriate. cyclopentanol + HCl>ZnCl2
- Predict the major products of the following reactions, including stereochemistry where appropriate. ) cyclopentylmethanol + Na2Cr2O7>H2SO4Predict the Product of the reaction and indicate regiochemistry and stereochemistry when relevantPredict the major products of each reaction and its stereochemistry. Write NR if none found.
- Predict the major products of the following reactions, including stereochemistry where appropriate. (a) cyclooctanol + NaOCl/HOAc (b) cyclopentylmethanol + CrO3 . pyridine . HClPredict the major products of the following reactions, including stereochemistry where appropriate. ) cyclopentylmethanol + CrO3 # pyridine # HClPredict the major products of the following reactions. Include appropriate stereochemistry in the product structures. S-1,3-dimethylcyclohexene + aqueous Hg(OAc)2Hg(OAc)2 followed by treatment with NaBH4
- Predict the major products of the following reactions, including stereochemistry.(a) cyclohexene + KMnO4>H2O (cold, dilute)Predict the products and show the mechanisms for the following reactions. Please indicate the correct stereochemistry where necessary.Predict the products of the following reactions, showing stereochemistry where appropriate.

