Q: Which of the following carbocations would be expected to rearrange? O None of these would be…
A: The given compounds are as follows.
Q: Q9. Arrange the following alkenes in order of increasing stability (least stable to most stable). II…
A: Given : Structure of Alkene is given To find : Stability order
Q: c) What is the "order of stability"(most to least) of carbocations? Use the following list. methyl…
A: Tertiary Benzyl > Secondary Benzyl > Primary Benzyl > Secondary Diallyl > Primary Allyl…
Q: A terminal alkyne (RC=CH) is exposed to excess HBr. What rule should be followed to determine the…
A:
Q: Which of the following is the most likely structure of the following carbocation after rearrangment…
A:
Q: Which of the followings is the MOST stable carbocation?
A:
Q: 4. (2 pts) Circle the most stable carbocation and cross out the least stable carbocation among the…
A:
Q: Arrange the alkenes in terms of increasing stability. Drag the choices to their correct positions.…
A: The stability of alkenes increases with increase in substitution on olefinc carbon. Note: According…
Q: Identity the carbocation that is the most stable? a. CH2CH3 CH O b. CHCH, CH, с. CH d. CH,CH2 CH;
A: Carbocation is species in which carbon is carrying positive charge on carbon. In carbocation, carbon…
Q: Rank the following alkenes in terms of increasing stability. (A) (B) (C) OC<A<B OA< C < B OA < B < C…
A: Stability of the alkenes increases on increasing the substitution which means highly substituted…
Q: The following carbocations are listed in increasing order of stability (least to greatest stability)
A: Stability of carbocation can be explained by inductive effect Carbocations are electron deficient…
Q: (а) or II I (b) or nag
A: Carbocation is species in which carbon carries positive charge.
Q: Which of the following carbocations ( A or B) is more stable? Explain your choice.
A:
Q: Which of the following carbocations would rearrange after reaction with HX? a. Хон b. ҳ CA ОН 'OH
A: -> When alcohol reacts with hydrogen halide Reaction proceeds through formation of carbocation…
Q: Draw a structural formula for the most stable carbocation with each molecular formula. Q.) C4H9+
A:
Q: Which of the followings is the most stable alkene? CH3CH2C=CH2 b. CH3-CHCH=CH2 а. CH3 CH3 CH3CH2 CH3…
A:
Q: Which of the following carbocations is(are) likely to rearrange? CH3 CH3 CH3 CH3 CH3, OCH2 CH3 CH3…
A: Structure is given The carbocation likely to rearrange = ?
Q: Which of the following carbocations (A or B) is more stable? Explain your choice.
A: In the given two carbocations, the stability is decided by the electronegativity factor of O and…
Q: ČH2 CH3 CH3 CH3
A: The carbocations given are,
Q: Which one of the following carbocations is the ?most stable one II III IV :Select one a. II b.I с.…
A: A chemical reaction is a symbolic representation of the conversion of substances to new substances.…
Q: Question 8 Rank the following carbocations from most stable to least stable: II II O III >I> II OI>…
A: Given that, Three carbocations have been given,we have to find out order of stability?
Q: A In each blank, type the letter of the more stable alkene in each pair (type A or B). A blank 1…
A: The stability of an alkene depends on the substituted groups around the double bond. It will also…
Q: Rank each of the following carbocations in order of stability (1 = least stable and 4 = most…
A: A carbocation can be defined as a molecule in which a carbon atom has a positive charge and three…
Q: The highest energy and be not stable of the following carbocations is: CH2 CH3 CH3 CH3 I II III IV…
A: The carbon atom bearing positive charge is known as carbocation.
Q: The carbocation below is significantly more stable than a typical primary alkyl carbocation. Explain…
A:
Q: Which carbocation is more stable? a. H2C=CH+ or HC=C+ b. HC=C+ or CH3CH2+
A: a) H2C=CH+ or HC=C+ b) HC=C+ or CH3CH2+ More stable carbocation has to be given below,
Q: Rank the following carbocations in terms of stability from lowest to highest. A C D D<C<B<A B<D<A<C…
A: In this question we have to tell the stability of carbocation in the term of stability.
Q: Rank the following carbocations in order of decreasing stability (most → least): CH₂CHCH₂…
A:
Q: Which of the following carbocations is the least stable? D. X С. Е. A. N А. В. H. Oc OA E B
A: We know the stability order of carbocation: 3°>2°>1°>CH3
Q: Identify the secondary carbocation from the following: o (CH3),CHCH2* O CH3CH2* o (CH3),CH* O CH3*
A:
Q: Rank the following carbocations in decreasing stability (most stable to least stable). [Note: your…
A: Carbocations are carbon ions which have a positive charge on it and its hybridization is sp2.
Q: Which one of the following alkenes is the most stable? H3C CH3 H3C CH3 H3C CH2-CH3 ÇH3 C= H3C. CH2…
A: More substituted alkenes are more stbale.More substituted alkenes can attain more hyperconjugation…
Q: a) CH3 b) CH3 – CH – CH- CH3 CH3 с) СНЗ — СН2 —- CH - СHЗ
A:
Q: 3. Rank the following carbocations in order of increasing stability. (1 = least stable, 5 = most…
A: The delocalization of lone pair of electrons or pi-bond electrons from one bond or atom to another…
Q: Which of the following alkenes is most stable? A) I B) II C) III D) all of them
A:
Q: Arrange the following alkenes in order of increasing stability (least stable to most stable). II II…
A: More substituted alkene has the greater number of hypercojugative C−HC-Hbonds and so greater is the…
Q: Rank the following carbocations in terms of their energy. A Highest Energy Lowest Energy
A:
Q: Which of the following radicals are allylic? B D. only D A, B, and C B and C
A: The radicals given are,
Q: Which carbocation will rearrange? +) +) II A. Tonly B. Il only C. both I and II D. neither I or II
A: Answer :- I have explained each solution below hope this will help you !!!!! Note - There will be…
Q: Rank the following carbocations in terms of their energy. + A В Highest Energy Lowest Energy
A:
Q: Rewrite each of the following stereospecific transforms as a synthetic step in the forward…
A: Since you have posted a question with multiple sub-parts, we will solve the first three sub-parts…
Q: Draw a structural formula for the most stable carbocation with each molecular formula. Q.) C3H7+
A: A chemical reaction mechanism represents the way to form and break the chemical bonds between…
Q: Which one of the following carbocations is the most stable one? I II III IV
A: The species in which the carbon atom bonded with three sigma bonds and has a positive charge is…
Q: (а) or II + I (b) or + I II (с) or + II +
A: (a) ( I ) carbocation is unstable because it is vinylic carbocation inwhich positive charge on sp2…
Q: Rank the following alkenes from MOST to LEAST stable. II II a. I>II>III b. III> II>I c. II>III>I d.…
A: Hydrocarbons are the molecules formed by the carbon and hydrogen atoms. They are of two types-…
Q: Draw the starting alkene that would lead to this the major product (and its enantiomer) under these…
A: The Starting Alkene that would lead to final product is given below
Q: Which of the following carbocations has the highest energy: * CH2 CH3 CH3 CH3 II II IV
A: The energy is related to the stability of the compound. The higher stability of the compound, the…
Q: Identify the least stable and the most stable of the following carbocations B A
A: Carbocation is species in which carbon carries positive charge.
Q: Which of the following is the most stabilized carbocation? 1 OMe a) OMe OMe b) OMe OMe d) a O e
A: We have to choose the most stabilized carbocation as follows in step 2
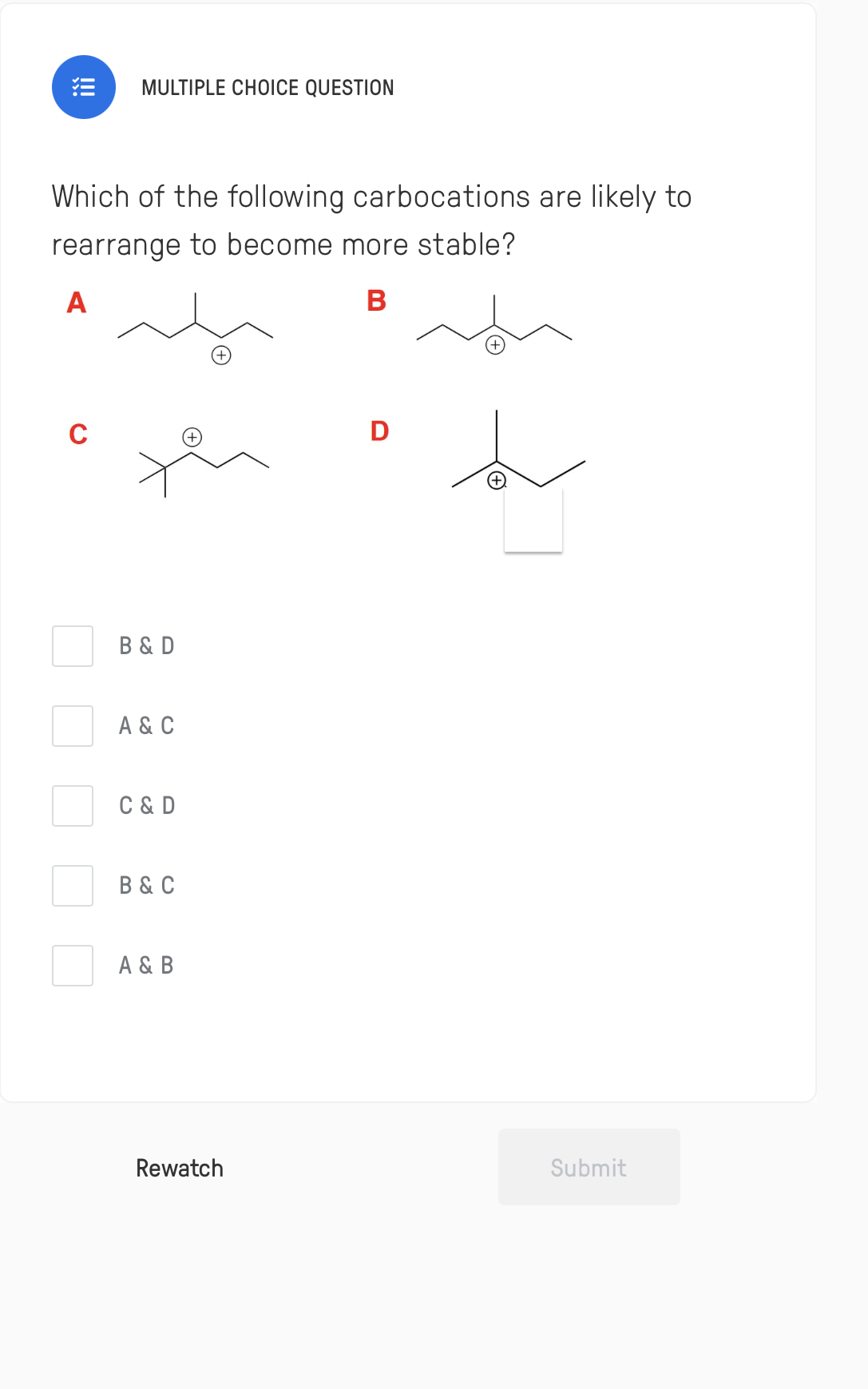
Q: Which of the following carbocations would you expect to rearrange?
A: A chemical reaction mechanism represents the way to form and break the chemical bonds between…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Consider the molecule 1-bromo-2-methylbutane. C3 and C4 should be drawn as Et as in theexample. This group is called an ethyl group and can be considered a sphere about twice the sizeof a methyl group. Draw the following Newman projections sighting down the C1C2 bond... a. The lowest potential energy conformation. b. The highest potential energy staggered conformation.Rank the following groups in order of decreasing priority. −H, −CH3, −Cl, −CH2Clwat is degree unsaturation ? & draw 2 structures per number that meet the following
- onsider the methyl group at the junction of rings A and B. Is it axial or equatorial to ring A? Is it axial or equatorial to ring B?Please draw the line structures for the C8H17+ carbocations that have the carbon skeleton below And please circle the most stable carbocationName the alkene below.Use only E/Z designators to indicate stereochemistry.
- Indetify the molecule that is (2S,3R)-3-chloro-2-methylbutan-1,2-ol. (A, B, C or D)Will the cyclopropane formed be facing same direction as substituents or away?(will it have dashed wedges)?Acoording to Organic Chemistry 3rd edition (Klein), the dienophile in the image is trans. Why is the following molecule trans and not cis?
- 1,4-Pentadiene (CH2=CH-CH2-CH=CH2) is a liquid at room temperature and has a density of 0.66 g/mL and molar mass of 68.12 g/mol. In a laboratory experiment, 3.80 mL of this compound was treated with 4.80 mL of conc. H2SO4 (100% w/w; molar mass 98.08 g/mol). Note that the density of conc. H2SO4 is 1.84 g/mL. The resulting sulfate ester was then treated with 1.20 mL of water (molar mass 18.02 g/mol) affording, after work- up, 2,4-pentanediol (molar mass 104.15 g/mol) as the crude product. The crude product was then purified by simple distillation, which yielded 2.00 g of pure product. What is the theoretical yield of 2,4-pentanediol expressed in grams? Show calculations. What is the percentage yield of pure 2,4-pentanediol?1,4-Pentadiene (CH2=CH-CH2-CH=CH2) is a liquid at room temperature and has a density of 0.66 g/mL and molar mass of 68.12 g/mol. In a laboratory experiment, 3.80 mL of this compound was treated with 4.80 mL of conc. H2SO4 (100% w/w; molar mass 98.08 g/mol). Note that the density of conc. H2SO4 is 1.84 g/mL. The resulting sulfate ester was then treated with 1.20 mL of water (molar mass 18.02 g/mol) affording, after work- up, 2,4-pentanediol (molar mass 104.15 g/mol) as the crude product. The crude product was then purified by simple distillation, which yielded 2.00 g of pure product. a. Provide a balanced chemical equation to show the reaction between 1,4-pentadiene and sulfuric acid. Do not use molecular formulas in the chemical equation except for sulfuric acid. b. What reactant is the limiting reagent in this chemical equation? Show calculations to support your answer.how do i determine degree of unsaturation for an equation if it includes elements such as N, O, F. when each of these are present in the formula do i subtract, add, or leave it the same from the amount of carbons present. example includes C6H7N or C4H7Br

