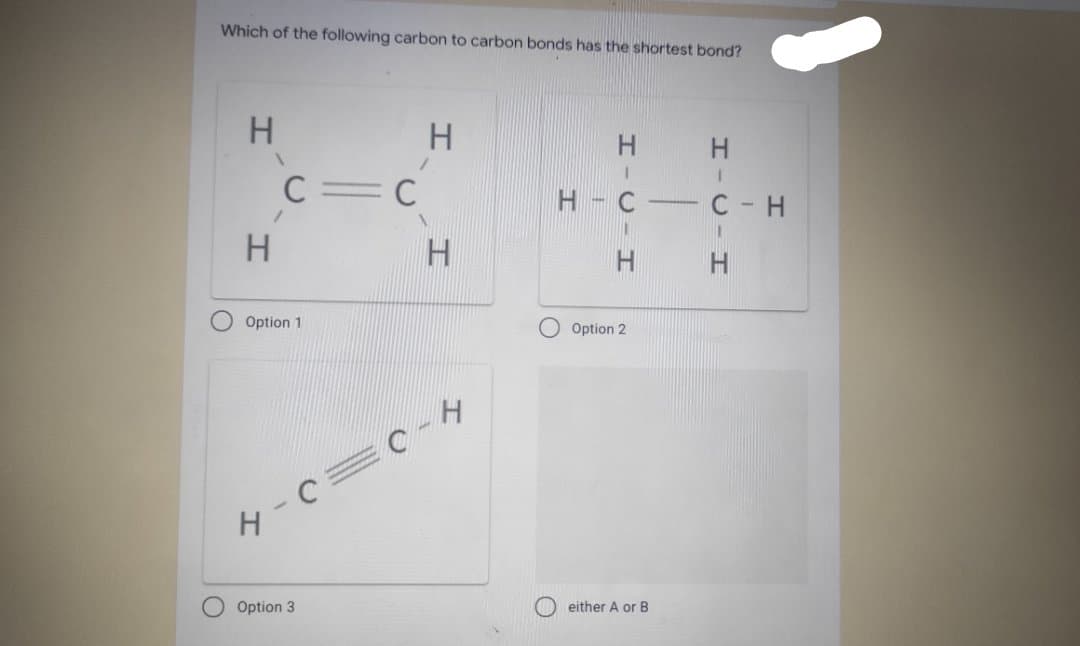
Which of the following carbon to carbon bonds has the shortest bond? H C=C H. H. O Option 1 O Option 2 O Option 3 O either A or B HICIHI HICIHI
Q: Isotretinoin, a fatty acid, is a medication used to treat severe acne that has not responded to…
A: The E and Z configuration for the olefin carbon is as shown:
Q: Which of the following bonds will be most polar? Group of answer choices C-O N-F N-O C-F Cl-F
A: The most polar bond from the given choices = ?
Q: Which of the following bonds will be the weakest? Select one: a. Ge-Ge b. Si-Si c. C=C d. C-C
A:
Q: Choose the more stable, i.e., less reactive, molecule (or ion) in each of the three pairs of…
A: molecule 1st will be less stable than molecule 2nd, because carbonyl group of 2nd molecule is…
Q: (a) Arrow story – indicate which bond(s) are broken and/or formed according to each indicated arrow…
A: In an organic reaction mechanism, a curved arrow represents the transfer of electrons. In an organic…
Q: 20. Which is the most polar bond? (а) С-С (b) С-Н (c) N-H (d) О-н (e) Se-H
A: Relationship between polarity of bond and electronegativity difference (∆EN) Polarity of bond can be…
Q: Hydrophilic or Hydrophobic? 1: Ionic or 2: Polar or Positive Atoms end? non-polar covalent bond?…
A: a) C and C : Since C and C forms non polar covalent bonds.
Q: Choose the most stable structure. + 5
A: The above given structures are chair conformation of dimethyl decalin. Decalin can exist in two…
Q: 3- The bonds that would be expected for atoms forming H2O molecule and joining H2O molecules…
A: What type of bond formed in H2O and joining H2O molecules are ?
Q: H-ċ-o-H H-N-c-H H-C-CEN: H-c-N=ö Which of the structures shown below has 4 sigma (o) bonds and 1 pi…
A:
Q: A C H H H H-c=C-c-CEC-H H
A: We are to discuss which carbon-hydrogen bond is easier to break in the given structure.
Q: Below is one of the two resonance forms for the acetate ion. What is true about the bond rotations…
A: Lewis structure of a molecule can be defined as the arrangement of valence shell electrons in all…
Q: The structures below show the step-wise bond making and bond breaking in this reaction. Draw curved…
A: Given a step- wise bond making and bond breaking reaction. we are asked to draw the curved arrows to…
Q: Which are diastereomers?
A: diastereomers given in step 2
Q: 13 What is the approximate value of the H-C-H bond angle in methane, CH Select one: Da 120 Ob. 109.5…
A: In methane molecule, there are four bond pairs of electrons and to reduce the repulsion between bond…
Q: Draw the resonance hybrid of CH3NO2 in Problem 1.53(a).
A: Two Resonance structures can be made on the given compound but there will be only one structure…
Q: Rank the polarity of the indicated bonds in structures I-III from most polar to least polar. A) C-CI…
A: Bond with more electronegativity difference is more polar.
Q: 2. Draw the important resonance forms of the following free radicals. Indicate how the resonance…
A:
Q: Which of the following bonds is most polar? Group of answer choices H-F H-O H-Cl H-N
A:
Q: What is the configuration, Z or E, of each of the following double bonds? (a) Br (b) D Br (c) -NO2…
A: Hello. Since the question contains more than three sub-parts, the first three sub-parts shall be…
Q: Which compound has a longer C¬Cl bond?CH3CH2Cl CH CHCl 2at one time, it was used as arefrigerant, an…
A: The given compounds are CH3CH2Cl and CH2=CHCl2 as shown below:
Q: How many moles of bonds between which pairs of atoms are broken during the combustion of 3 moles of…
A: The balance chemical reaction of combustion of methane gas is
Q: Draw a circle the compound below with the strongest C-X bond and draw a rectangle around the…
A:
Q: CH Rank the following in order of decreasing importance as a contributing resonance structure to the…
A: Rules :- Rule 1: The most significant resonance contributor has the greatest number of full octets.…
Q: Structure of compound A: H CH3
A: To find : several small molecules from the compound and to give the reason for the breakage of bond.…
Q: Think about the Lewis structure for the CH;N molecule (shown below). (Gray = C; white =H; red = 0;…
A:
Q: Rank the following bonds in order of increasing strength:
A: As we move down the group the electronegativity( tendency to attract shared pair of electrons)…
Q: Which compound would you predict to be highest in energy? OH H20 step 1 H-CI + step 2 step 3…
A: Interpretation: The compound among A,B, C and D having highest energy is to be predicted. Concept…
Q: Lewis Structures and Formal Charge 1.42 Give the formal charge on the highlighted carbon in each…
A:
Q: REPRESENTATIONS OF ORGANIC MOLECULES Identifying isomers and resonance structures Determine the…
A: Here we have to find the relationship between structure A and structure B-
Q: O REPRESENTATIONS OF ORGANIC MOLECULES Identifying isomers and resonance structures Determine the…
A: Identify the relationship between given structures A & B = ?
Q: Part 2: Interpret each chemical formala below. Determine hoe many atoms of cach element make up the…
A: 6 ) Na+ and Cl- Means 1 atom of Na (sodium) with one atom of Cl ( Chlorine) to make Nacl.
Q: Draw a circle the compound below with the strongest C-CI bond and draw a rectangle around the…
A: The compound is mostly stabilized by the resonance effect . More is the resonance effect , more will…
Q: Answer the following questions about erlotinib and terbinafine. Erlotinib,sold under the trade name…
A: Hybridization is a conceptual process in which atomic orbitals are combined together to furnish a…
Q: Which bond is most polar? O H-S H-H O H-P O H-CI
A: Given :- H-S H-H H-P H-Cl To determine :- The most polar bond
Q: 3. 3 Draw resonance structures for the following: (a) HCO CH (sttog 01)loo olfot sai o ds HC CH CH3…
A: In resonance structures charges or radical are delocalised due to the presence of double bond or…
Q: Which of the following bonds would be the least polar? A. B-F B. O-F C. F-F D.C-F
A: POLARITY: When a chemical structure has an unlike electron density arrangement, it is a polar…
Q: Select the correct value for the indicated bond angle in each of the compounds. O−S−O angle of SO2…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: What is the molecular geometry of the molecule below? い-0-1 | O Linear O tetrahedral bent O trigonal…
A: In dichlorine monoxide (Cl2O) the central atom oxygen undergo sp3 hybridization.
Q: What is the approximate O-C-H bond angle in acetaldehyde CH;CHO? 180° O 109.5° 120° 90°
A: Since you asked multiple questions so as per Q&A guidelines of portal I solve first question…
Q: 14. Which is the odd one out? OH C. CH3 for OH o
A: 14.Carboxylic acid is a class of organic compound with general formula RCOOH, where is any alkyl or…
Q: Consider the three compounds shown below and then answer the question that follow: H. H. H H. N: H H…
A: Pi bonds are those bonds which are formed by the lateral overlapping of the orbitals between the two…
Q: Which bond is most polar? H-H H-I H-Br H-Cl
A: Interpretation: We have to tell which among the following is more polar.
Q: (CH3CH2)2NH, TSOH Atoms, Bonds and Rings Charges Draw or tap a new bond to see suggestions. Undo…
A:
Q: (e) (c) Хон H H H (b) (d) H H A
A:
Q: Which of the following bonds is least polar? Select one: a. B-H b. Al-H c. P-H d. C-H e. N-H
A:
Q: 1. Arrange in the order of increasing bond C - C lengths (lowest to highest) C2H2, C2H4, C2H6…
A: 1. Answer - The correct option is (e) According to the question The given compound are - ethyne ,…
Q: What are the approximate H-C-H bond angles expected for the carbanion whose structure is given in…
A: According to the valance shell electron pair repulsion theory (VSEPR), the valance electron pairs…
Q: Which of the following bonds would be considered the most polar? O A) P-C B) P-N C) O-F D) S-O E)…
A: The more the electronegativity difference between the atoms, the more will be the polarity of the…
Step by step
Solved in 2 steps

- __ Al(OH)3 + ___ HBr ------> ___ AlBr3 + ___ H2O Can yo balence this for meeach reagant must have 3 or less carbons . one image is the directions and one is the question.How do you do number 112? I think the greater the stengtg of the h bond, the more energy relaesed , the more stable, thus greater expthermic. Thst makes f + fhf the strongest h bond as f is most electroneg
- What is the approximate H−C−O bond angle in formaldehyde, H2C=O? a. 90° b. 109° c. 120° d. 180° Group of answer choices a b c dBreak compound A into several small (less or equals to 6 carbon atom) molecules. You need to explain the reasons why you choose to break those bonds.Is bond formation endothermic or exothermic? Write a + or sign above the arrow in the previousquestion to represent the sign of the energy change associated with the arrow.
- Consider the following orbital representation of HCCH (ethyne). a. Answer the same three questions (a-c) from the previous exercise. b. Label each CH bond in the drawing above. c. What is the total number of a bonds found in ethyne?…. bonds? d. How many p orbitals are there on a single carbon of ethyne? e. How many hybrid orbitals are there on a single carbon of ethyne?Organic chemistry is a bit like cooking. Later in this course we will study “recipes” for preparingnew molecules. A common instruction in such recipes is “add a strong acid” or “add a strongbase” (rarely are they added at the same time since a violent reaction ensues). You will see theseterms frequently from now on, and are expected to be able to recognize them or draw theirstructures from memory. Memorize them before next class.Answer numbers 4, 5, and 6 Find : (a) pOH (b) [H3O+] (c) [OH-]
- A. Find the moleular formula of diazomethane. b. Draw iits lewis structure and resonance forms as neccessary c. Explain why diazomethane is not very stable26.) How many sigma and how many pi bonds are there in molecule below? H3C-CH=CH-CH3 Write your answer as _____ sigma and _____ pi. For example, 6 sigma and 7 pi (no period at end).Which of the following funds will be released polar? A. B-F B.O-F C. F-F D. C-F D. C-F

