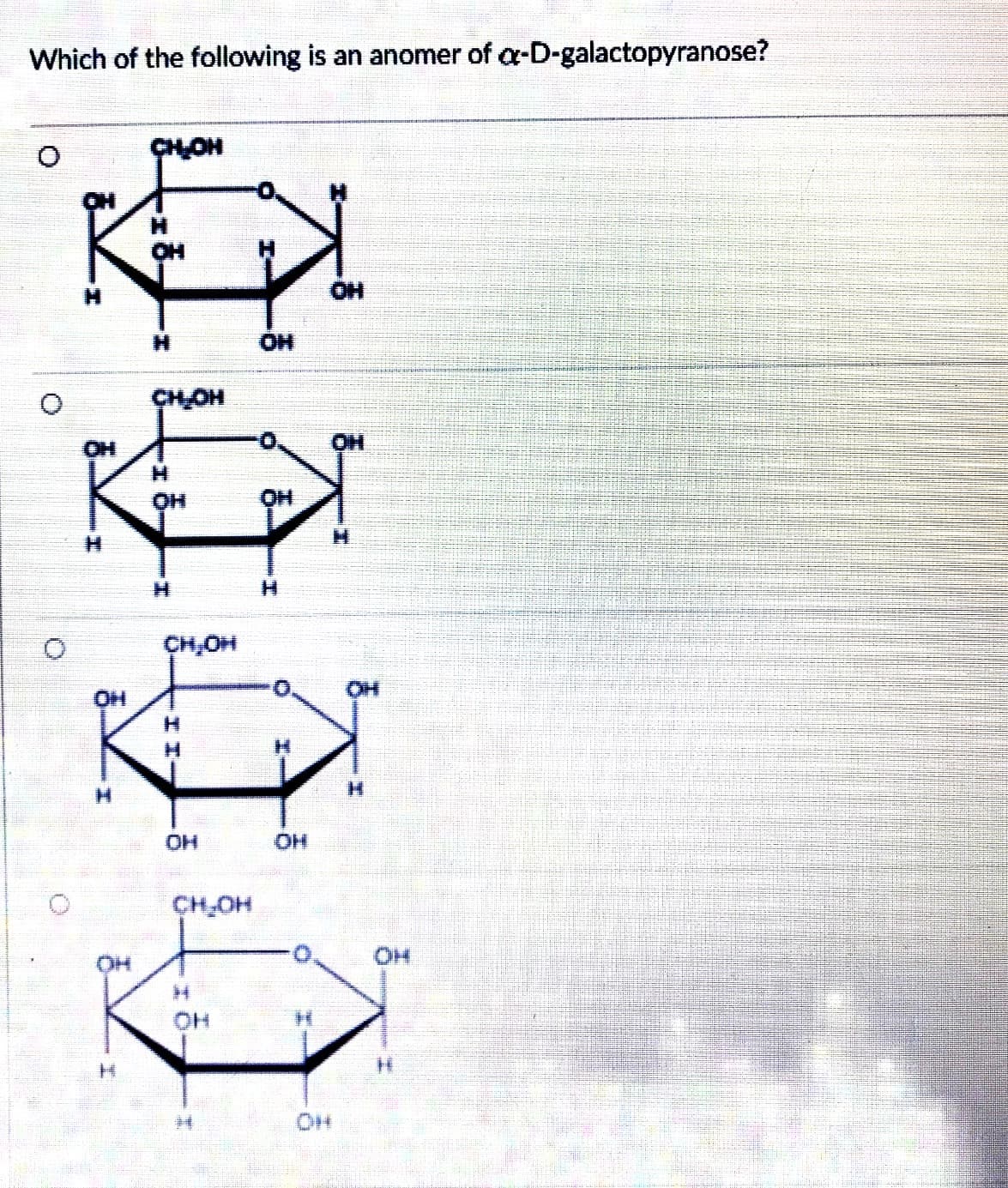
Which of the following is an anomer of a-D-galactopyranose?
Q: How to synthesize aspirin without using acetic anhydrid
A: Aspirin, which is also called as acetylsalicylic acid (ASA), it is a medication used in reduce…
Q: Which of the following enzyme catalyzes a reaction that introduces reduced nitrogen ito cellular…
A: Nitrogen metabolism is characterized by the coordinated expression of a large number of enzymes…
Q: Complete the table
A: Polysaccharides is one of the important component in living organisms, which is found many diverse…
Q: D-Galactose.
A: ''Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Regulation of metabolic pathways most typically occurs at enzyme steps that are thermodynamically…
A: Regulation of metabolic pathways occur by controlling the Enzymes either by modifying it's pre…
Q: A 44-year-old man diagnosed with acute tubular necrosis has a blood urea nitrogen of 60 mg/dL and a…
A: A heart attack or a heart stroke can cause the tubular necrosis. This is a condition in which the…
Q: A protein that has been reversibly denatured has Multiple Choice temporarily lost part or all of its…
A: Each protein has its own distinct sequence of amino acids, and the interactions between these amino…
Q: Long explanations are not needed. True or False a. One NADH is produced for every pyruvate…
A: The pentose phosphate pathway is an alternative pathway for glucose oxidation. It provides NADPH for…
Q: How many molecules of Pyruvate can form from Glycerol metabolism as a by-product of fatty acid…
A: The major precursors of gluconeogenesis are lactate, glycerol, alanine, ketone bodies like…
Q: Polyunsaturated fatty acids a). are solid at room temperature b). have carbon-carbon double bonds…
A: Fatty acids are carboxylic acids with long hydrocarbon chain. There are two types of fatty acids -…
Q: How many hydrogen bonds exist between this DNA strand and its complementary strand? 5 -TGTCCCA-3'…
A: Nucleic acids are the biomolecules composed of nucleotide units. A nucleotide unit is composed of a…
Q: Calculate the frictional coefficient of a molecule of DNA of 20 base pairs in water at 20C; assume…
A: DNA or deoxyribonucleic acid is a polynucleotide chain made of monomeric units of nucleic acids.…
Q: 1. Are small biomolecules that are solubilized by the Cl atoms present: a. CH3(CH2)10CH(OH)2 b.…
A: Electrophoresis is a technique for seperating charged molecules in a mixture under the influence of…
Q: what are the effects of PSTMB shown and why is it important to show effects of PSTMB in vitro…
A: Introduction: According to the Warburg hypothesis, cancer cells undergo a high rate of intracellular…
Q: An unsaturated fatty acid produces more energy than the analogous saturated fatty acid. A) True B)…
A: Saturated fatty acids contain hydrocarbon chains that are only linked by single bonds. There are one…
Q: Total Number of NADH Obtained from one Pyruvate Molecule Total Number of FADH2 Obtained from one…
A: In respiring bacteria pyruvate is further oxidized after it is transported into mitochondria that is…
Q: evidence of Bial's test to show a positive result for the presence of a specific sugar
A: Bial's test is a chemical test used to test for Pentose sugar. It includes chemicals like orcinol,…
Q: first half of the Q cycle results in
A: The mechanism of Q-cycle is proposed by Peter Mitchell. Q-cycle terms as proton motive Q cycle. It…
Q: 1. What are the different classes of lipids? State their function and identify their chemical…
A: A group of organic compounds includes lipids that are insoluble or poorly soluble in…
Q: How does the DNA hold information?
A: Deoxyribonucleic acid (DNA) is a polymer made up of two polynucleotide chains that coil around each…
Q: QUESTION 21 Consider the following template strand of DNA: AAT TCA CCC 3' 5 The mRNA made from this…
A: DNA stands for Deoxy-ribonucleic acid. It is a molecule which carries the information of a proteins.…
Q: what color will cellulose turn for iodine test?
A: Cellulose is an important component of cell wall of plants. It is a Polysaccharide made up of beta D…
Q: If a cell sample is placed in pure water, what is the concentration of the salt inside the cell?…
A: The water move inside the cell and outside the cell through the process of osmosis. If a cell is put…
Q: What are the different solid phases that antibody or antigen can bind to, in ELISA?
A: Quantitative immunological techniques that use a solid phase can be defined as solid-phase…
Q: 2. Use your knowledge of amino acids (and the R groups) and tertiary structures of proteins to…
A: The disease, which is referred to as sickle cell anaemia is caused because of mutation. In this…
Q: What does the last number in the numeric designation of enzymes refer to?
A: Each enzyme is allocated a four digit EC number,an enzyme Commission number does not specify enzymes…
Q: Describe the whole process and the principle behind DNA extraction.
A: Deoxyribonucleic acid (DNA) is a polymer made up of two polynucleotide chains that coil around each…
Q: True or False: 1. Electron-deficient chlorophyll molecules of PS II receives electron from PS I.…
A: Introduction: The light-absorbing pigment of thylakoids is arranged in functional arrays called…
Q: A patient is suspected of having low stomach acid, a condition known as hypochloridia. To determine…
A: The gastric sample here is acidic. The equation of titration of an acid (sample in this case) with…
Q: Ribose-5-phosphate is produced by oxidative decarboxylation of 6-phosphogluconate catalyzed by the…
A: Above questions is from PENTOSE PHOSPHATE PATHWAY.
Q: 3. Explain the importance of lipid content in the structure of biological membranes
A: The lipids which it encircle membrane proteins in biomembranes have a significant impact on their…
Q: The succinyl CoA to citrate pathway of the citric acid cycle is shown. Identify the missing…
A: Succinyl Co-A and the reactions given in the question are part of the TCA cycle. The TCA cycle is a…
Q: 2. The following graph illustrate the binding of 02 to proteins: myoglobin and hemoglobin ( one…
A: Hemoglobin is an oligomeric conjugated protein with four peptide chains joined by a…
Q: Which of the following is the complementary strand of a DNA strand made up of 32% cytosine? A.…
A: Two strands of DNA are complementary to each other , if we know the sequence of one strand, we could…
Q: Think of a metaphor or analogy of the light-dependent reactions similar to the “construction…
A: Light-dependent reactions occur withinside the thylakoid membrane of the chloroplasts and arise…
Q: Long explanations are not needed. Direct answers would suffice. a. The penultimate carbon in an…
A: Aldohexoses are carbohydrates with six carbon atoms and an aldehyde group. Glucose is an example of…
Q: 5 Write equilibrium expressions for these reactions. (a) 2H,0,(g)=2H,0(g) + O,g) (b) 2N,0,(g)=…
A: Introduction: A number that relates the concentrations of the beginning compound and products of a…
Q: 5'- What will be the Sanger products of the DNA with base sequence ACGTCGACTCCGGTC-3'
A: DNA sequencing is the biochemical method used for determining the order of nucleotide bases, A, G,…
Q: What is the maximum number of possible intra-peptide disulfide bonds for any given peptide form?
A: The side chains of Cysteine residues in proteins undergo oxidation to form disulfide linkages.
Q: The lipase substrate emulsion contains 0.500 mg of olive oil per 3 mL Also, the molar mass of the…
A: The number of moles of a substance is calculated by using the equation, n=mM, where, "n" is the…
Q: 2. Use your knowledge of amino acids (and the R groups) and tertiary structures of proteins to…
A: Sickle cell disease is a disorder of red blood cells that are inherited. This is a disease that is…
Q: The reason for the decrease in the rate of enzyme reaction as the temperature is increased beyond…
A: An enzyme is a substance which serves as a catalyst in living things, governing the rate of chemical…
Q: State the significance of HMP shunt
A: The pentose phosphate pathway is also called the hexose monophosphate shunt pathway.
Q: Analyte Results Reference ranges total protein 9.40 g/dl 6.0 – 8.0 g/dl albumin OD=0.09, []=? 3.7…
A: Solve it by using Lambert beers law. Which states that Absorption is directly proportion to…
Q: Which of the following is FALSE about glycolysis? a. The pathway axidizes two moles of NADH to NAD*…
A: Glycolysis is the technique that breaks down the 6-carbon glucose into 3-carbon pyruvate that is a…
Q: Multiple Choice - Explain your answer in 3-5 sentences. - answer properly QUESTION: The allosteric…
A: Proteins that behave as physiological catalysts are known as enzymes. Catalysts speed up the…
Q: What are two important compounds that lie at crossroads of major metabolic pathways? Select both…
A: Metabolic pathway engineering in microbial hosts for heterologous biosynthesis of commodity…
Q: "Match the gluconeogenic precursor to the type of reaction(s) that bring it to gluconeogenesis as…
A: Gluconeogenesis is the synthesis of glucose from a non-carbohydrate source. The precursors for…
Q: What is expected to happen to a suspended protein when the pH of the solution is adjusted to the…
A: The pH impacts the precise nature of protein interactions by altering the protonation state of…
Q: An unknown amino acid labeled BAYOKEM has an IpH value equal to 5.97. 1. If its pka2 is equal to…
A: pH is the measure of the strength of H+ ion or Hydronium ions in solution. pOH is the…
Step by step
Solved in 2 steps with 1 images

- Draw the α-l-glucopyranose sugars using Haworth projections:Draw the Haworth projection of β-D-Altopyranose given the structure of D-Altrose.Given a tripeptide Cys-His-Lys, Cys: Pk1 = 1.71; Pk2 = 10.78; PkR = 8.33 His: Pk1 = 1.82; Pk2 = 9.17; PkR = 6.0 Lys: Pk1 = 2.18; Pk2 = 8.95; PkR = 10.53 draw the protonic equilibria for the tripeptide what is the IpH?
- Draw the following sugars using Haworth projections:a. b-d-galactopyranose b. a-d-tagatopyranose c. a-l-glucopyranoseDuring your experimental investigation of the conversion of glutamine to proline, you have labeled the γ-carbonyl group of glutamine with 14C. What carbon in the proline product will be labeled?Draw Haworth projection formulas for the b-anomer of monosaccharides with each of the following Fischer projection formulas
- The pH at which the Lysine R group is 20% dissociated?What is the melting temperature and G/C content of the following primers? a.) 5’ GAAATAATTTTGTTTAACTTTAAG 3’ b.) 5’ GTAACTCAGCTTTCAGGTCG 3’Draw the following compounds: a. α- and β-anomers of D-galactose b. aldonic acid, uronic acid, and aldaric acid derivatives of galactose c. galactitol d. δ-lactone of galactonic acid

