Which of the following is (are) the most likely reaction product(s) in the monobromination of cyclohexanone by bromine in the presence of light? Select an answer and submit. For keyboard navigation, use the up/down arrow keys to select an answer. a 2-Bromocyclohexanone Your answer 2-Bromocyclohexanone, 3-bromocyclohexanone 2-Bromocyclohexanone, 3-bromocyclohexanone, 4-bromocyclohexanone d 3-Bromocyclohexanone e 4-Bromocyclohexanone
Which of the following is (are) the most likely reaction product(s) in the monobromination of cyclohexanone by bromine in the presence of light? Select an answer and submit. For keyboard navigation, use the up/down arrow keys to select an answer. a 2-Bromocyclohexanone Your answer 2-Bromocyclohexanone, 3-bromocyclohexanone 2-Bromocyclohexanone, 3-bromocyclohexanone, 4-bromocyclohexanone d 3-Bromocyclohexanone e 4-Bromocyclohexanone
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter16: Aldehydes And Ketones
Section: Chapter Questions
Problem 16.47P: The base-promoted rearrangement of an -haloketone to a carboxylic acid, known as the Favorskii...
Related questions
Question
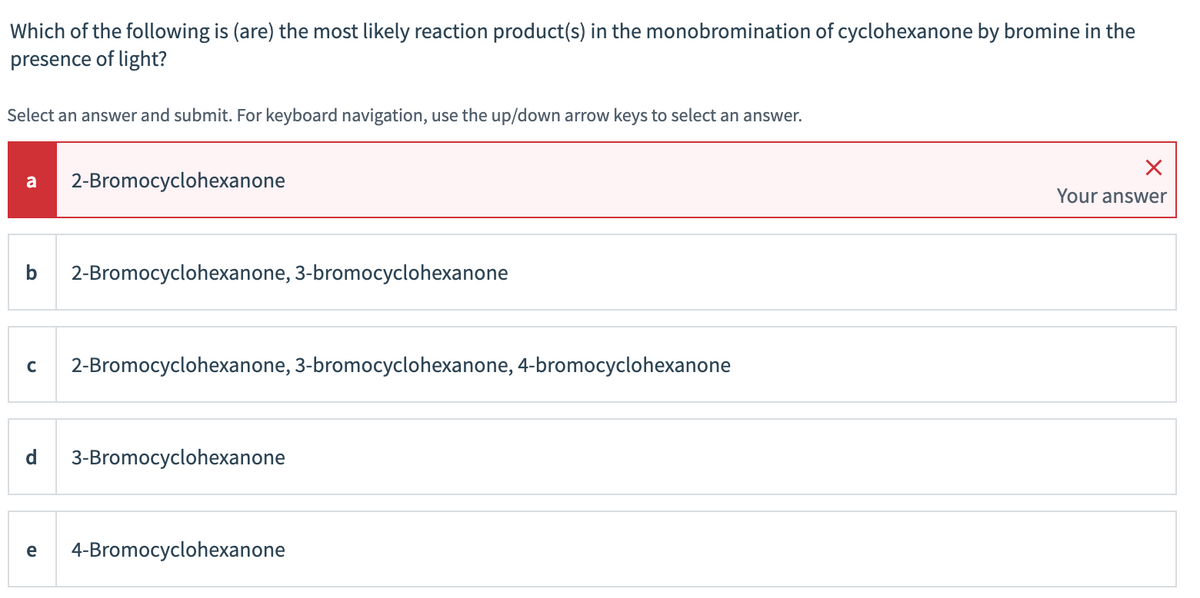
Transcribed Image Text:Which of the following is (are) the most likely reaction product(s) in the monobromination of cyclohexanone by bromine in the
presence of light?
Select an answer and submit. For keyboard navigation, use the up/down arrow keys to select an answer.
a
2-Bromocyclohexanone
Your answer
b
2-Bromocyclohexanone, 3-bromocyclohexanone
2-Bromocyclohexanone, 3-bromocyclohexanone, 4-bromocyclohexanone
3-Bromocyclohexanone
e
4-Bromocyclohexanone
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning