Which of the following statements is correct? A 0.1245 M HCI solution contains 0.1245 moles HCl per ml of solution. A solution that contains 0.3176 mmoles KCI/mL has the same concentrations as 317.6 moles/L KCI. A solution that contains 0.03571 moles NAOH/L is also the same as 35.71 M NaOH D In 100.0 mL of 0.2456 M NaCl solution, it contains 24.56 mmoles NaCl.
Which of the following statements is correct? A 0.1245 M HCI solution contains 0.1245 moles HCl per ml of solution. A solution that contains 0.3176 mmoles KCI/mL has the same concentrations as 317.6 moles/L KCI. A solution that contains 0.03571 moles NAOH/L is also the same as 35.71 M NaOH D In 100.0 mL of 0.2456 M NaCl solution, it contains 24.56 mmoles NaCl.
World of Chemistry, 3rd edition
3rd Edition
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Chapter15: Solutions
Section: Chapter Questions
Problem 21A
Related questions
Question
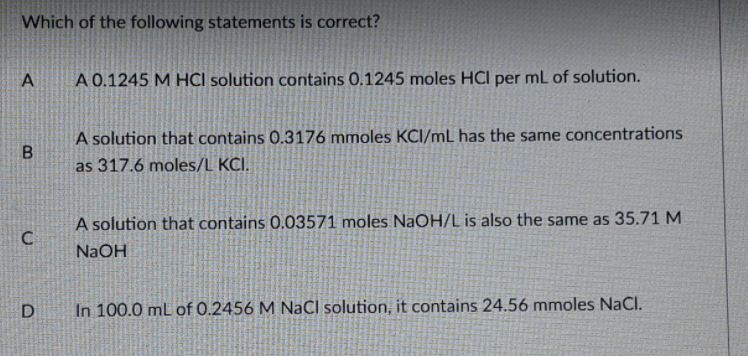
Transcribed Image Text:Which of the following statements is correct?
A 0.1245 M HCI solution contains 0.1245 moles HCl per mL of solution.
A solution that contains 0.3176 mmoles KCI/mL has the same concentrations
as 317.6 moles/L KCI.
A solution that contains 0.03571 moles NaOH/L is also the same as 35.71 M
C
NaOH
D
In 100.0 mL of 0.2456 M NaCl solution, it contains 24.56 mmoles NaCl.

Transcribed Image Text:Consider an aqueous solution of Na,COa that contains 2.5467 g Na,CO, (M.M.
105.99) dissolved in 250.0 mL solution. Which of the following statements is
correct?
A
The solute is NazCO3 while the solvent is H2O.
The solute is Na2CO3 but the solvent was not defined.
The solvent is NazCO3 and the solute is water.
The solution is Na,CO3 and the solvent is water.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning