Which of the following statements is FALSE? (You can choose more than one answer) Gas doesn't have a definite volume or shape. The temperature at which the solid start changing to liquid is the boiling point. Temperature is the average kinetic energy of particles in a substance. ON. Diamond is an example of amorphous solid. V. Sublimation is the change of state from liquid to gas. The density of water increases when you cool it down from 4"C 0°C. Evaporation takes place only at the surface of a liquid. V The formation of clouds in the sky is an example of condensation.
Which of the following statements is FALSE? (You can choose more than one answer) Gas doesn't have a definite volume or shape. The temperature at which the solid start changing to liquid is the boiling point. Temperature is the average kinetic energy of particles in a substance. ON. Diamond is an example of amorphous solid. V. Sublimation is the change of state from liquid to gas. The density of water increases when you cool it down from 4"C 0°C. Evaporation takes place only at the surface of a liquid. V The formation of clouds in the sky is an example of condensation.
Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Mark S. Cracolice, Ed Peters
Chapter2: Matter And Energy
Section: Chapter Questions
Problem 48E: Questions 47 and 48: Samples of matter may be classified in several ways, including gas, liquid, or...
Related questions
Question
100%
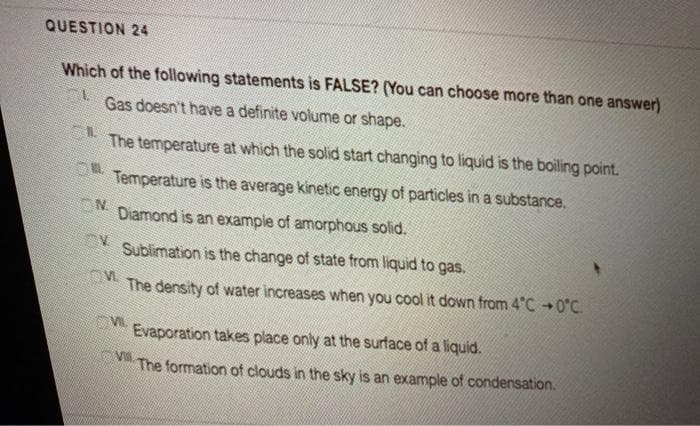
Transcribed Image Text:QUESTION 24
Which of the following statements is FALSE? (You can choose more than one answer)
Gas doesn't have a definite volume or shape.
The temperature at which the solid start changing to liquid is the boiling point.
Temperature is the average kinetic energy of particles in a substance.
ON
Diamond is an example of amorphous solid.
Sublimation is the change of state from liquid to gas.
The density of water increases when you cool it down from 4°C +0°C.
Evaporation takes place only at the surface of a liquid.
VII
The formation of clouds in the sky is an example of condensation.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning