Q: A molecule cannot be polar if: It does not have a C5 principal axis of symmetry Its dipole moment…
A: A molecule cannot be polar if it has completely symmetric distribution of charge. A symmetric…
Q: The configuration of the chiral centers in the following molecule are: но O 1 (R) & 2 (S) O 1 (S) &…
A: A carbon atom that is bonded to four different groups is called a chiral carbon atom. Due to the…
Q: H H H. е. CH3 CH3 но g. HS H NH2 f.
A:
Q: Identify e absolute configurations of all chiral centers in the following compounds. H CH3 CI OH CH3…
A: The absolute configurations of all chiral compound has to be provided.
Q: Ibuprofen is a chiral compound used as a nonsteroidal anti-inflammatory. Determine the number of…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: butane has three energy minima with respect to rotation around the C2-C3 dihedral angle. Two minima…
A: The molecular structure of butane is as follows: In the analysis of…
Q: How many diastereomers would a molecule with three chiral centers have? A. 2 B. 3 C. 6 D. 8
A:
Q: Ectocarpene is a volatile, sperm cell-attracting material released by the eggs of the seaweed…
A:
Q: Which of the following compounds does not have a plane of symmetry? II II IV Multiple Choice O Only…
A: The plane of symmetry works as an identification for the optical activity of the molecule. If a…
Q: OH NHCH3
A: Determine the absolute configuration of the chiral center from the given structure---
Q: A well known non steroidal anti inflammation drug NSAID exists in two sterochemical forms. Only one…
A: We have to predict the absolute configuration at chiral center.
Q: Which of the following molecules are chiral? I II III CI CH2OH ICH3 H3C CH3 H3C° H3C H. IV V CH3…
A: Given molecules,
Q: Which of the following statements are true? Explain. I. If a molecule has a plane of symmetry it is…
A: Given statements are : I. If a molecule has a plane of symmetry it is achiral.II. If a molecule has…
Q: What are the designations for the chiral centers for molecules A and B? CO,H Br H,N-"CH2OH A B
A: Stereochemistry is a branch of chemistry that deals with the study of relative 3D-arrangement of…
Q: 39. Which of the following compounds has a plane of symmetry? g. 8. 8 & „CI "CI II II IV A) I and II…
A: "Since you have asked multiple question, we will solve the first question for you. If youwant any…
Q: Which of the following is the staggered conformation for rotation about the C1 – C2 bond in the…
A: There are different types of conformation in alkanes and cycloalkanes. They are eclipsed and…
Q: Chair conformations must be considered before determining overall chirality of a molecule. a. FALSE…
A: Chirality is a property of a molecule which tells us that the compound is optically active, and is…
Q: What is the configuration of the two chiral centers in the following molecule? CH H3C H H3c OH а.…
A: We have to find out the R,S configuration for the given molecule at chiral carbon which is at number…
Q: Potential Energy ▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬ 0 60 3 H₂C 120 180 Degree of Rotation II 240 300 A) 1 B) 2 C) 3 D)…
A: Conformational analysis.
Q: 7. In a chair conformation, 1,3-diaxial interactions between atoms lead to: a) angle strain b)…
A: In chair conformation, steric repulsion are there due to electronic repulsion.
Q: What is the configuration of the two chiral centers on the following Fischer projection? A) 2R, 4R…
A: According to CIP rule the priority was taken, and now if we go 1 to 3 priority group if it is clock…
Q: Sketch the chair conformers for the following compound. Then identify which conformer is more and…
A: The question is based on the concept of stereochemistry. we have to identify the most stable…
Q: 1. Classify each of the following common objects as being either chiral or achiral. Assume in each…
A: A question based on chirality, which is to be accomplished.
Q: Assign R/S configurations to the two chiral centers indicated below (Hint: When the 4thpriority…
A:
Q: 8. For the following molecule draw both possible chair conformers and predict the direction of the…
A: It contains two possible chair conformers and the stability depends on the structure. The axial…
Q: CH3 CH3 CH3 H3C CH3 CH3 H H CH3 Br I Br II III
A:
Q: CH3 c=c H H Br ČH3 I II III CH2CH3 CI CI H H IV V VI
A: Plane of symmetry
Q: Clearly draw both chair conformers of the following molecule. Indicate which conformer is more…
A: To draw the chair conformation of the below molecule.
Q: The configurations of chiral centers in following molecule are: H. NH2 H3C HO- CH2-CH3 O R, S S, R…
A:
Q: In a staggered conformation of the Newman projection, the dihedral angle, which is an angle between…
A:
Q: Why don’t molecules that contain a plane of symmetry have a chirality center?
A: Since we know that when four different atoms or groups are attached with a carbon atom then it is…
Q: Is the following molecule chiral? If yes, draw the enantiomers and determine their absolute…
A: The spiro compounds can also exhibit chirality due to the presence of central chirality. When both,…
Q: Determine the absolute configuration at the chiral center in each of the following molecules. CH3 H.…
A: Give priorities to all four groups on chiral centre according to atomic number .Highest atomic…
Q: What is the hybridization of P in CH3PO4?
A: Hybridization is defined as mixing of orbitals having different energy so as to rehybridize to form…
Q: Which of the following compounds is most polarizable? (A) (B) (C) (D) Compound A Compound B Compound…
A:
Q: Which of the following chair conformation is lowest in energy? I II II IV III O V O I O IV I
A: Conformational isomers: The conformational isomers are formed by the rotation of a carbon-carbon…
Q: IDENTIFY WHICH OF THE FOLLOWING EXAMPLES HAS AT LEAST ONE PLANE OF SYMMETRY C H;C OH HO H. N-H co…
A: In this question, we will see that which is chiral and which are not chiral. You can see details…
Q: 7) The following compound has how many chiral centers? CH3 CH3 A) 0 В) 1 C) 2 D) 3 E) 4
A: The compound given is,
Q: Identify the configuration of each of the chiral centres: (R) or (S). OCH3 ii i Oi = S and ii = R Oi…
A: Rules to assign R and S nomenclature The four atoms or group attached to chiral centre are given…
Q: H. ОН F H. H. F. ISI
A: Which of these do not contain a plane of symmetry?
Q: Ectocarpene is a volatile, sperm cell-attracting material released by the eggs of the seaweed…
A:
Q: Which compound does not have a plane of symmetry?
A: Plane of symmetry is the plane which will pass through the molecule in such a way that the molecule…
Q: The configuration in following molecule is: он No chiral center S 50% (R) & 50% (S) R
A: Introduction: We have to calculate the configuration .
Q: Which other factor along with torsion decide the stability of conformer? ionic bonding…
A: Conformers : Conformers or conformational isomerism is present when a compound shows…
Q: Predict the R.S configurations for the labelled chiral centers: Br va)l-S, 2-S, 3-S, 4-R (b) 1-S,…
A: We can draw the line diagram and then according to the CIP rule, we can give the R/S configuration.…
Q: CH-ST9 Which structures are chiral? I H3C NH2 II OH H3C CH3 он III IV. O A. I, III, IV O B. II, III,…
A: To find: The chiral structures
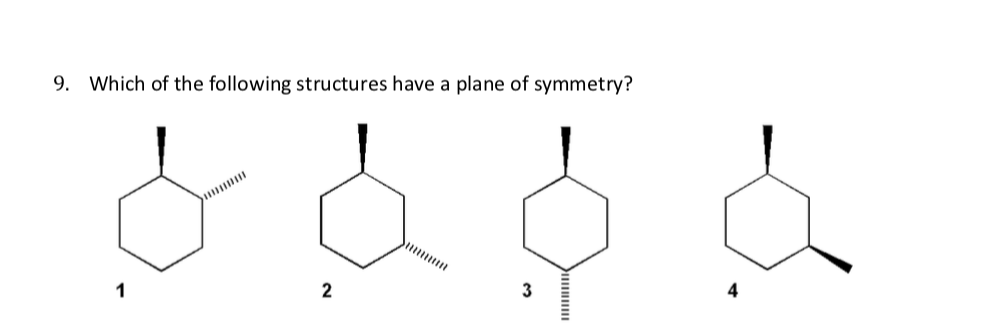
Can i get help with this problem
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 2 images

- Label the following pairs as A) distereoisomers, B) enantiomers, C) conformers, or D) the same. b1 c1 d1 e1 a2 b2 c2 The structures above are labeled a1, a2, b1, b2 ...etc. Using these labels, indicate which compounds above would have an optical rotation ofa) draw the newman projectjon between C3-C4 bond for all of the structures over 360 degrees of rotation. start with highest energy of conformation b) starting at 0 degrees draw a qualitative torsional energy plot as a function of degree of rotation1) Are the molecules A and B... conformational isomers? Diastereomers? Enantiomers? Position isomers? Non-related? 2) What about the molecules B and C? 3) What about the molecules C and D?

