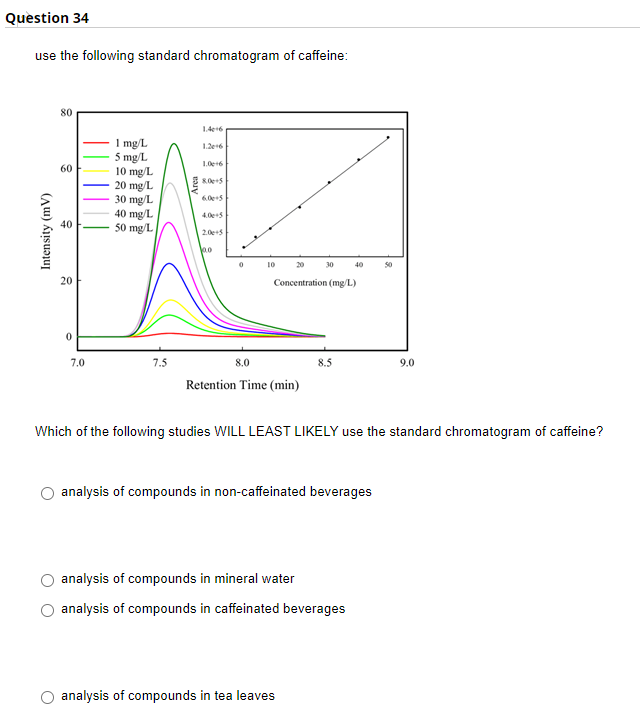
Which of the following studies WILL LEAST LIKELY use the standard chromatogram of caffeine? analysis of compounds in non-caffeinated beverages
Q: Consider the following balanced equation: Cl 2(g) + 2 Br (aq) → Br 2(aq) + 2 CI (aq) [CI 2] [Br ]]…
A: The rate law for the reaction can be written as: rate=kCl2xBr-y......(1) Here, x is the order with…
Q: 1. PBr, 2. NaNg, THF HO 3. Pd/C, H2 (Excess) 4. Mel (Excess) 5. Ag,0, H20 `NH2 NH2 A B D
A: For the given sequences of reaction of the given alcohol, we can predict alkene as the major…
Q: Part A The radioactive isotope 133 Xe is used in pulmonary respiratory studies to image the blood…
A: I don't know how much detail you want for this, but the simplestequation to use iswhere τ1/2 is the…
Q: • Draw the Lewis Structure dot for Butanoic acid, CH3CH2CH2COOH and answer the following questions…
A: Lewis dot structure showing the arrangement of electrons in the molecule. The electrons are shown by…
Q: Calculate AG ° of the following reaction: 2 C (s) + H2 (g) → C2H2 (g) AG2 + Use Hess' Law, AG°= AG¡…
A:
Q: Calculate ΔH° for the reaction using the given bond dissociation energies.…
A: This is a combustion reaction of methane.
Q: 1. Me H2N OEt MeOH, H* Et 2. xylenes, heat 1. H3O*, heat 2. NH O. Meo OMe p-TSOH, PhH, reflux 3.…
A: 1. Fischer Indole synthesis. 2. Ketal to ketone. Ketone to imine. Imine reduction. 3. O-Bromination,…
Q: 4. The following 'H NMR spectrum of a CroH12O2 compound was obtained on a 90 MHz spectrometer. TMS…
A:
Q: The index of Hydrogen deficiency indicates the degree of unsatuiation of the molecule which help in…
A: Degree of unsaturation for hydrocarbons is calculated using formula : U = ( Hydrogen in alkane -…
Q: Ksp for Fe(OH)3 (Fe(OH)3(s) ⇌ Fe3+(aq) + 3OH-(aq)) is 4.0 × 10-38. In the presence of 0.01 M NaOH,…
A:
Q: What is the concentration of co3²- required (molarity) in a solution that is 0.0014 M in Sr2* in…
A: First write dissociation equation of SrCO3 and use given Ksp and concentration of Sr2+ .
Q: Give a clear handwritten answer and explain
A: Balanced the given redox reaction under basic condition and answer the following question---
Q: At what temperature or range of temperatures will a reaction be spontaneous if the value of H° =…
A: H° = +42.8 kJ = 42800 J S° = +331.5 J/K?
Q: reaction will be product-favored or reactant favored when temperature changes occur. If reaction…
A: If reaction favour to the product side, it means that the reaction is spontaneous in the forward…
Q: Would you expect the rate of hydrolysis of methyl-p-fluorobenzoate (X = F) to be substantially…
A: To determine whether the rate of hydrolysis of methyl-p-fluorobenzoate (X=F) to be a) Substantially…
Q: Which has the highest entropy? Explain your answer in a separate sheet. a. H2O(s) at −50°C…
A: Which has the highest entropy ? Explain. a) H2O(s) at -50 °C b) H2O(g) at 150 °C c) H2O(l) at 100…
Q: As the concentration of reactants decrease, the rate of the reaction generally O increases. O cannot…
A:
Q: Enter electrons as e". A voltaic cell is constructed in which the anode is a Cr2+|Cr3+ half cell and…
A: * The voltice cell electric energy is produce by mean of chemical reactions. * In voltice cell…
Q: What reagents are needed in reaction 3? ... Br Br Reaction 1 Reaction 2 Reaction 3 CH3B AIBr3 (A)…
A:
Q: Which of the following factors DOES NOT affect retention time of a sample undergoing normal phase…
A:
Q: 0.0524 moles of h2o equals how many molecules of water?
A: Number of moles of H2O (water) = 0.0524 moles Number of molecules of water = ?
Q: (a) Standardization of iodine solution ( finding its concentration ). 5.00 g vitamin C are dissolved…
A: #(a): Standardization of iodine(I2) solution: Volume of the diluted vitamic C solution used for…
Q: What would be the concentration of X 2- if a 10 mL of a 1 M solution of KOH was added to 50 mL of a…
A: The chemical reaction for the neutralization of a diprotic acid H2X with KOH is needed to be…
Q: Nitration reaction takes away electrons from the ring deactivating the ring and lessening the…
A: Nitration is a process in which there is the replacement of a hydrogen atom (organic compound) with…
Q: The product for reaction 2 is a vicinal diol, which of the following reaction also produces a…
A: a. mild oxidation ( Alkenes react with cold dilute alkaline KMnO4 produces the syn diol). This is a…
Q: The membrane shown is permeable only to solute molecules. In which direction will solutes flow? 0.4…
A: Here, Diffusion will takes place which is a physical process in which net movement of molecules from…
Q: In a normal phase chromatography setup, which would be the predicted characteristics of the eluates…
A: Ans : Eluates will be in decreasing polarity ( Benzoic acid is more polar than benzaldehyde.…
Q: Which molecule given below has only London intermolecular forces?Explain your reasoning (include…
A: Intermolecular interaction depends upon nature of bonded atom and bonds between molecules A) HF…
Q: 1. A gas occupies a volume of 31.0 L at 18.0"C. If the gas temperature rises to 36.0°C at constant…
A: The ideal gas equation is - PV = nRT = wRT/MM Where - P = pressure V = volume n = number of…
Q: The concentration of CO- in a saturated solution of CuCO, is 1.60x10- mol/L at a certain…
A: * Solubility product is the product of molar concentrations of ion present in solution, when ion…
Q: The concentration of Sr2* in a saturated solution of SrCrO4 is 6.29x10-3 mol/L at a certain…
A:
Q: In the laboratory a student finds that it takes 21.6 Joules to increase the temperature of 13.4…
A:
Q: The reaction N204(s) 2NO2(3) has a K, - 0.140 at 25 °C. In a reaction vessel containing the gases in…
A: Given: Kp = 0.140 Partial pressure of N2O4 = 0.138 atm
Q: Use the half-reaction method to determine the net-ionic redox reaction between the permanganate ion…
A: Test tube #5: The redox reaction involves the conversion of MnO4– ion (purple) to Mn2+ (pink) ion…
Q: 3. Ferulic acid is a phenolic compound present in popcorn, bamboo shoots, and cereals. Provide…
A: The ferulic acid, a phenolic compound from the class of hydroxycinnamic acid, has a number of…
Q: Calculate the pH of a solution after 47.0 mL of 0.500 M NaOH is titrated with 53.0 mL of 0.250 M of…
A:
Q: Which amomg the fragments below will be dectected by mass Spectrophotometer? [CH3CH3]+ CH3CH3…
A: Which among the fragments below will be detected by mass Spectrophotometer?
Q: Which solution has the greatest buffer capacity? Which solution can react with the largest amount…
A: Buffer solution: The solution that resists the change in pH is known as a buffer solution. The…
Q: What is the least favorable cation to reduce on the Table of Std. Reduction Potentials? Ca2+ Li+…
A:
Q: delta S° for a reaction can be calculated from the following equation. a. S° = (moles…
A: Entropy is a measure of the randomness of the system. The entropy change in a chemical reaction can…
Q: radius. Explain your reasoning in the given space here in this question. Only use qualitative…
A: As we know, In periodic table if we move from left to right, atomic radius decrease while top to…
Q: Which of the following aromatic compound has a hydroxy substitution? There can only be 1 answer.…
A: Aromatic compounds are those which has 4n+2 pie electrons. Aromatic compounds obey Huckel's rule.…
Q: Chemistry Calculate [Cu2+]eq using Nernst Equation E cell: 0.48 Temperature: 25.0 C n=2
A: EoCu²+/Cu : 0.337 V Given, Ecell : 0.48 V Temperature : 25 oC = 298K n = 2
Q: Solve the word problem and show your detailed solution. The final answer should have a unit and the…
A:
Q: CHEMWORK Choose the statements that are correct. (Select all that apply.) In a galvanic cell the…
A:
Q: Draw the correct Lewis structure for each of the following using the Octet Rule. (Show the total…
A:
Q: Electrochemical cell uses salt bride to complete the circuit. What is true from the following…
A:
Q: In the laboratory a student finds that it takes 49.2 Joules to increase the temperature of 14.8…
A:
Q: 3. The Statue of Liberty consists of copper plates on an iron skeleton. The statue was recently…
A: EoFe3+/Fe = -0.036 VEoCu2+/Cu =0.34 V
Q: "In our bodies, sugar is broken down with oxygen to produce water and carbon dioxide." According to…
A:
Step by step
Solved in 2 steps with 1 images

- In a thin layer chromatography (TLC) experiment, solute A traveled 20 mm while solute B traveled 30 mm. The solvent traveled 40 mm. Which has the greater affinity for the solvent? a. not enough data b. solute B c. solute A d. both A & B have equal affinityHydrocarbons in the cab of an automobile were measured during trips on the New Jersey Turnpike and trips through the Lincoln Tunnel connecting New York and New Jersey. The total concentrations ( ± standard deviation) of m- and p-xylene wereTurnpike: 31.4 ± 27.0 μg/m3 (32 measurements)Tunnel: 54.9 ± 29.8 μg/m3 (32 measurements)Do these results different at the 95% confidence level?You run the chromatography system with paper as the stationary phase and 70% isopropanol as the mobile phase. You determine the Rf of a polar analyte to be 0.40. If you use 100% isopropanol (a less polar solvent) instead of isopropanol as the mobile phase what would you expect to happen to the Rf value? No value effect on Rf higher lower could be higher or lower
- A 50.00 (±0.03) mL portion of an HCl solution required 29.71(±0.03) mL of 0.01963(±0.0030) M Ba(OH)2 to reach an end point with bromocresol green indicator. The molar concentration of the HCl is calculated using the equation below (attached image): a.) Calculate the uncertainty of the result (absolute error). M=0.02333(±?????) M b.) Calculate the coefficient of variation for the result. CV= (Sy/y) x 100%Choose correct letter of the answer. 1. Column/s used for normal phase chromatography- a.ODS b. C18, C8 c. silica, alumina d. ODS silica gel 2. The following statements are true for any chromatographic analysis except:a. The analyte component interacting most strongly with the stationary phase elute faster than those with weaker interactions. b. On the other hand, those with least interaction with the mobile phase elute the slowest. c. Interactions between mobile phase – analyte – stationary phase can be both chemical and physical in nature. d. Chromatography is a separation technique.Trial 1 Trial 2 Trial 3 Mass of solution 29.824 g 29.855 g 29.816 g Volume of solution 25.00 mL 25.00 mL 25.00 mL Density of salt solution 1.193 g/mL 1.194 g/mL 1.193 g/mL Average density of salt solution --> 1.193 g/mL <-- What is the relative average deviation, %?
- In thin-layer chromatography, what is the Rf value of a compound if the solute travels 2.9 cm and the solvent moves 4.5 cm after 5 minutes? Note: Express answers up to TWO decimal places.Figure 1:Experimental results Mass of empty weighing dish,g 2.2522g Mass of weighing dish + CaCl2 · 2H2O,g 6.2540g Mass of CaCl2 · 2H2O, g 4.0018g [Na₂CO₃], mol⋅L−1 0.3330 mol⋅L−1 Volume of the CaCl2 solution used, mL 10.00mL Volume of the Na₂CO₃ solution used, mL 10.00mL Mass of the filter paper + watch glass, g 51.3999g Mass of the filter paper + watch glass + dry product (final), g Weight 1=51.7235g Weight 2=51.7058g Figure 2:Calculated data Mass of dry product, g 0.3059g Moles of CaCl2 used, mol 0.02721mol Moles of Na₂CO₃ used, mol 0.02721mol [CaCl2], mol⋅L−1 0.3330 mol⋅L−1 Limiting reagent Calculated mass of excess reagent remaining in the mixture after reaction, g Theoretical yield, g % yield I need help in my lab. The number of mols of cacl2 and na2co3 gives me the same number. If I need to calculate the limiting reagent, how would I be able to do that if they have…Table of caffeine standards concentration . Sample Conc, ppm Std1 16 Std2 32 Std3 48 Std4 64 Std5 80 If the volume used to make 100 mL of std 1 is 2 uL what is the concentration in M used to make std calibration curve ? The standards are going to be used to build calibration curve to analyze caffeine in an energy drink. if 500 mL of the energy drink has target of 400 mg , how will you prepare the sample if you need 10 mL for the analysis ? Caffeine MM=194.19 g/mol.
- The gravimetric factor used to express CoCBr6·H20 in a sample that is finally weighed as PbClBr is choose below: FW PbClBr / 6 x FW CoCBr6·H20 FW CoCBr6·H20 / FW PbClBr FW CoCBr6·H20 / 6 x FW PbClBr 6 x FW PbClBr / FW CoCBr6·H20Chromatography 1. External Standard Method A. Calculate the regression equation and the R-squared value. B. Calculate the concentration of cocaine found in the blood samples of the presidentiables. Which presidential candidate “reliably” tested positive for cocaine? 2. Internal Standard Method >Calculate the concentration of party drug X in the blood samples of the presidential candidates. >Suppose the LOD for party drug X is 0.200 ppm, which of the presidential candidates “reliably” tested for party drug X?Calculate the following. i) The slope (m) and the standard deviation of the slope. ii) The concentration of Na ( CA) in the unknown.



