Q: 2. Provide a balanced equation for the hydration equation of each of the following: a. Ethanoic…
A: The hydration of carboxylic acid results the formation of a salt of carboxylic acid.
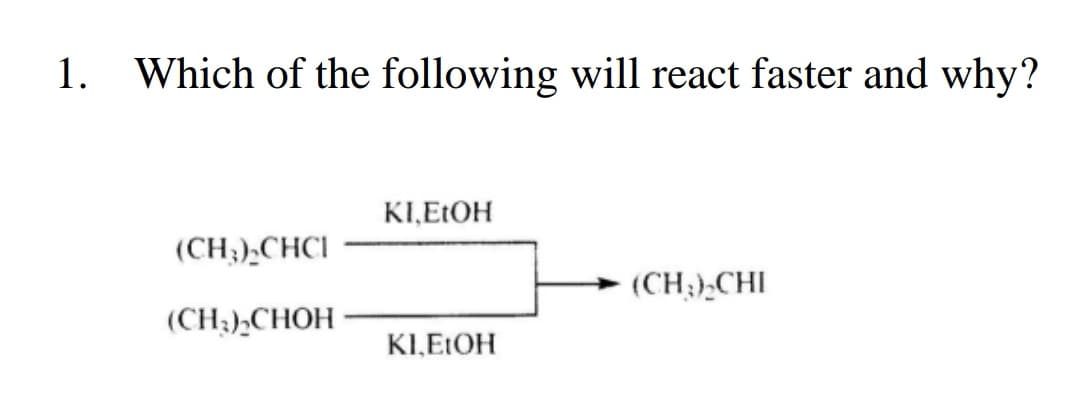
Q: Trans-1-bromo-2-methylcyclohexane will yield a non-Zaitsev elimination product (3-methylcyclohexene)…
A: Alkyl halides give an E2 elimination reaction with KOH. In the E2 elimination reaction, the removal…
Q: provide the structure of the intermediate and product for the following reaction : (c) H CH₂OH/H (C)
A:
Q: How many grams of sodium lactate are needed to prepare 7.50 liters Ringer's lactate? 6.O g
A: Question detail
Q: How many grams of carbon dioxide can be produced from the combustion of 48.74 g of pentane?…
A: To answer this question, we need to use the balanced chemical equation given: C5H12(l) + 8 O2(g) → 5…
Q: 0-10. a The acid dissociation constant K of hydrocyanic acid (HCN) is 6.2 x 107 Calculate the pH of…
A:
Q: :) pgs. 413-416 book 오 1. 2. H2, Ni -NH2 , H+
A: Imines are produced from acid catalyzed condensation between aldehydes or ketones with amines.
Q: Determine the final pH for the following two solutions a) solution A after mixing 500ml 0.2M HCl…
A: Answer: pH tells us about the power of hydrogen ion concentration in its solution and its value is…
Q: Consider the following equilibrium: N₂ (g)+3H, (g) 2NH, (g) 1 AG-34. KJ Now suppose a reaction…
A: Answer: Given reaction is the formation reaction of Ammonia and we have to find out the direction of…
Q: Use the answers to the questions above and the data attached to help you answer the question below.…
A: At the maximum volume of the airbag in the simulation, how many moles of gas are contained? Use all…
Q: The oxidation of iodide ion by arsenic acid, H3AsO4H3AsO4, is described by the balanced equation…
A:
Q: 10. Which of the following reactions would be capable of doing work under standard conditions and at…
A: The question is based on the concept of chemical thermodynamics . we need to identify the reactions…
Q: 2) Write the formula for the conjugate base of each acid. a) HC b) H₂SO, - HCHO 3) Write the formula…
A:
Q: a. b. C. he the following: O e. CH 0 N d. CH3CH₂ CI CH₂CH3 CH CH₂COOH O || C. CH₂C=N CH CHỊ…
A: The given compounds are examples of carboxylic acid and their derivatives with variable carbon…
Q: What would be the density (in g/L) of a sample of C₂H₂Cl₂ gas at 70.0 °C and 2.50 atm of pressure?
A:
Q: The following compound would be classified as a {blank). CH3-CH-CH₂-CH₂-CH3 1 OH O primary alcohol O…
A: primary alcohol : if hydroxyl carbon is attached to only one R group secondary alcohol : if…
Q: In the laboratory a "coffee cup" calorimeter, or constant pressure calorimeter, is frequently used…
A:
Q: How much of the solution can I make?
A: To provide how much of the (9 ppm Atrazine, DMSO) solution could be made out by using 0.04 g of the…
Q: Draw the Lewis structure for the molecule CH3CH2CCH. How many sigma and pi bonds does it contain? A)…
A:
Q: Determine the number of moles of gas particles produced when the following amounts of each chemical…
A: Stoichiometry is a crucial idea in chemistry that enables us to compute reactant and product amounts…
Q: Is this reaction overall more acidic or basic? I am a little confused because it produces an OH- but…
A: Answer: When a salt of weak base and strong acid is added into the water, it gets hydrolyzed to…
Q: What is the pH (to two decimal places) of a saturated aqueous solution of LiF (at 25 °C)? Ksp = 3.8…
A: For the saturated aqueous solution of LiF Ksp = 3.8 × 10-3 Ka(HF) = 6.67 × 10-4
Q: Draw the product(s) of the following reaction, including all stereoisomers: Br₂ hv
A: 3 methyl hexane react with Br2 in presence of light to form bromo compound with seven stereoisomer
Q: For the following conversion, identify if the starting material has been oxidized, reduced, a…
A:
Q: 5.00 L of an ideal gas at 268 K and 1.50 atm are heated to 353 K with a new pressure of 6.60 atm.…
A:
Q: The normal melting point of benzoic acid is 122.4°C. Predict the signs of AH, AS, and AG for the…
A:
Q: OH₂CI
A: For the first structure, hydrogen will make the bond which is known as bifurcated but for the second…
Q: a) Draw the complete mechanism for this reaction: CH₂0 1) NaOCH, 2) H₂0 b) What drives the…
A: This is a type of condensation reaction
Q: Predict the major organic product for each of the following reactions. (Minor products inorganic…
A:
Q: f) ( culi or ? HO
A:
Q: H3PO4 is a polyprotic acid with three acidic protons. Be sure to include the proper phases for all…
A: Since, When polyprotic acids dissociates in water, their protons dissociates one by one following…
Q: The FDA recommends that women who are pregnant avoid eating more than three 6.00 oz servings of…
A:
Q: A 50.00-mL aliquot of 0.0700 M HF is titrated with 0.140 M NaOH. The reaction is: HF + NaOH --> NaF…
A:
Q: 3. From the following reactions couples Which of them will go faster? Explain 1) 2) 3) Br + MeOH Br…
A: Since you have asked multiple question, we will solve the first three questions for you. If youwant…
Q: Question: At the maximum volume of the airbag in the simulation, how many moles of gas are…
A: At the maximum volume of the airbag in the simulation, how many moles of gas are contained? Use all…
Q: 4. Draw a reaction diagram to create an amine from a) benzene and from an b) alkyl halide.
A: (a) Benzene and (b)Alkyl halide We need to create ammine from (a) Benzene and (b)Alkyl halide .
Q: What is the structure of diethyl phthalate? What is its (diethyl phthalate) purpose in the…
A: We need to explain the purpose of chemiluminescence reaction and draw the structure of diethyl…
Q: 10. A recent report describes the reaction of C₂B4H8 with Fe(CO)s at 240°C. Two products were…
A: “Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: From the attached procedures, make a table consisting of all the compounds and their respective…
A: To find the name of the compounds given in the procedure and find their molecular weight, density…
Q: Gaseous ethane CH3CH3 will react with gaseous oxygen 02 to produce gaseous carbon dioxide CO2 and…
A: Answer: Limiting reagent is the reactant that exhaust first in the chemical reaction and decides the…
Q: D-Fructose can be reduced with NaBH4. Modify the molecule to show a product of the reaction. You…
A:
Q: H3C HO =0* CO H3C H3C CH3 CH3 CH3 NaOH, EtOH heat KOH, EtOH heat NaOH, EtOH heat
A: Aldol Condensation can be defined as an organic reaction in which enolate ion reacts with a carbonyl…
Q: At 35.0 °C, a 12.0 L vessel is filled with 6.25 moles of Gas A and 6.55 moles of Gas B. What is the…
A: o calculate the total pressure of the mixture of Gas A and Gas B in the given conditions, we need to…
Q: List the following alkyl halides in order of decreasing reactivity toward SN2 reactions (from 1:…
A: Answer:- This question is answered by using the simple concept of chemical reactions of alkyl halide…
Q: 4. What is the major organic product obtained from the following reaction? 1. NaNH, 2. CH₂CH₂Br + 0…
A: Product of the reactions
Q: From the table of available reagents select the one(s) you would use to convert 2-phenylethanol to…
A: In 1st reaction using pocl in presence of base i.e pyridine we remove water molecule h2o after that…
Q: potassium tert-butoxide in tert-butyl alcohol
A: If primary alkyl halides react with potassium tetra-butoxide then eliminate the halogen group…
Q: 1. Find Moles OH in 50 ml sample 0.050M cacla 100 mL + 0.100 Na OH 100 mL
A: To solve this problem, we need to use the balanced chemical equation for the reaction between CaCl2…
Q: diene diels - Aider →눈 product dienophile 1.0 2. NOHCD3.CHJOH 강 CH₂ OH AG₂0 Eton 1. DIBAL, Toluene,…
A: The Baeyer villiger oxidation is an organic reaction in which a ketone is treated with peroxides in…
Q: Data points at 1 mL increments. (a) What is the pH before any titrant has been added? (b) What is…
A: The curve represents pH Vs volume of titrant (in ml). a) The pH before any titrant has been added =…
Step by step
Solved in 3 steps with 1 images

- Which of the following reactions will have the faster rate and give a better yield? Use drawings of the transition state (show orbitals!) to explain why.Arrange the following according to INCREASING reactivity towards E2: 1st ( least reactive)? 2nd? 3rd? 4th (most reactive)?In which compound is the halogen substituted most rapidly by aq hydroxide ions? a. CH3CH2CH2CH2Cl b. (CH3)3CI c. (CH3)3CCl d. CH3CH2CH2CH2I
- The substitution reaction below forms two ehters (which are consitutional isomers). Please draw the structures of the 2 ethers formed. Are the ethers single enantiomers or racemic mixtures?Which group in following pair is assigned the higher priority? −I, −Br1. which one is the starting material 2. what is the product/reaction 3. is it sn1 or sn2? (what is the reaction rate equation and mechanism?)

