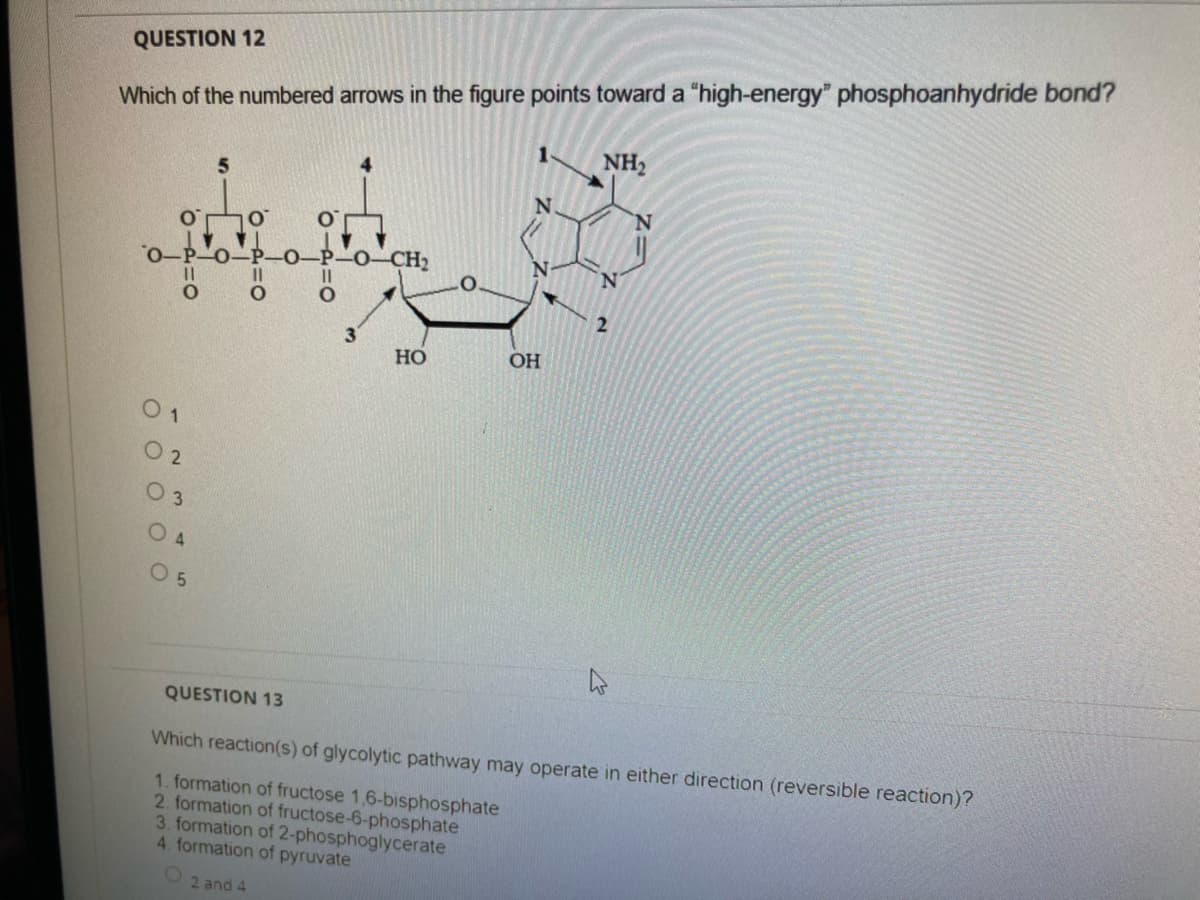
Which of the numbered arrows in the figure points toward a "high-energy" phosphoanhydride bond? NH2 -P-0-CH2 0-P-0-P 3 Но OH 0 1 0 2 0 3 O 4 O 5
Q: Sucrose, C 12 H 22 O 11 , is table sugar or cane sugar. It can be decomposed by heat into carbon (a…
A: Biological molecules such as sugars, lipids, proteins are all organic molecules that are composed of…
Q: Н—С—ӧ—н 5-H I-
A: Bond angle : The angle between bonding orbitals formed around central atom . Bond angle can be…
Q: un an electron-dot symbol of an element, the dots are O only the electrons that will participate in…
A: all atoms have electrons , electrons take part in bond formation . the lewis dot struture is a…
Q: What type of bonds form between oxygen & Hydrogen atom
A: The hydrogen an atom and oxygen atom together forms water molecule.
Q: Water is a in the DEpolymerization of a disaccharide into two monosaccharides reactant catalyst…
A: generally, the water molecule is added to the disaccharide to break it down into two…
Q: You want to make a solution that is 1x TE AND 10% glycerol in the same solution. You have the…
A: A percentage solution can be defined as the amount of volume of a particular chemical compound that…
Q: Fill in the blank, the parentheses are the choices for the blank: When a sodium atom loses a single…
A: Atom is neutral because it contains an equal number of protons and electrons. When an atom accepts…
Q: Which of these atoms has an oxidation number of 2-? O Sulfur O Chlorine O Aluminum O Neon O Silicon…
A: Oxidation State/number And Valency Valency is defined as the number of electrons present in…
Q: The most unstable conformer in cyclohexane? Boat half chair Chair Twist boat
A: Cyclohexane is a non-polar molecule and it does not have any ring strain, which is the abnormality…
Q: Give the number of valance and core electrons for each. Valence electrons are from the group number.…
A: Valence electrons are the one that occupies the outermost shell or highest energy level of an atom…
Q: Organic compounds are present in all living cells. They allshare the following characteristic:(a)…
A: Bio-biological macromolecules are the big molecules needed to live in from smaller organic…
Q: ZI A IZ НО. B OH IZ N H НО, E ZI .N.
A:
Q: Which of the following sub
A: To assign " R " and "S " configurations to chiral carbons in optically acive compound , Cohn…
Q: What coefficient must be placed in the following blanks so that all atoms are accounted for in the…
A: As per the law of conservation of mass, mass can neither be created nor destroyed. This also applies…
Q: Which arrow is pointing at a peptide bond in this molecule? H н4 о H H2N 11 CH H CH2 H OH SH O A) 1…
A: A peptide bond is a covalent chemical bond that is formed between the nitrogen atom of amino group…
Q: ) Complete the following information about charge & are located charge & are located charge & are…
A: Electrons have a charge of -1 and are situated on the atom's perimeter. They are a fraction of the…
Q: You are a teaching assistant in a general chemistry lab. The next experiment is to be an oxidation–…
A: Iodometry also known as Iodometric titration is a method of volumetric analysis which is a redox…
Q: Which structure below shows the placement of the hydroxyl groups of the other carbons that form part…
A: The ring structure is called Haworth projection where as straight chain model is Fischer projection.…
Q: HH нно н но HHO H-N-CC-OH H-N-C C-N-C-C-OH H-NC C-OH CH2 CH2 HC-CH3 CH3 CH2 CH2 HC-CH3 CH3 CH3 ČH3…
A:
Q: Sodium (Na) atoms often lose the single electron that is in their outermost shell. How does losing…
A:
Q: What type of chemical reaction is shown below? HO Monomer 1 Monomer 2 + он но Monomers linked by…
A: Conjunction/combination reaction: Its is combining reaction where two or more molecules combine or…
Q: What is the other reactant required in this reaction? (Box 2) * H H H-C-0-H -Ra но- 2 H-C-0-H но-…
A: The lipids are hydrophobic molecules and one of the most important biomolecules that is present…
Q: For numbers 21-22, refer to the image given below. CH3CH2-0-H + H* 21. What type of bond will be…
A: Alcohols, just like water, are both weak bases and weak acids. The acid ionization constant of…
Q: H What is this molecule? N-C-C OH R Name this molecule Explain
A: Protein is made up of a monomeric unit called amino acid and there are 20 amino acids that code for…
Q: Each carbon atom can bond with as many as-_________ other atom(s). a. one c. three b. two d. four
A: Bonds play a vital role in various biochemical interactions. There are 4 major types of bonds.…
Q: Which of the following statement is false? * Hydrocarbons contains hydrogen and carbon atoms…
A: Hydrocarbons are organic compounds and mainly used as combustible fuel source.
Q: How many carbon atoms are there in the longest chain? Br seven O eight nine none of the above
A: The naming of an organic compound is primarily done on the basis of identification of its longest C…
Q: (a) A homogeneous mixture which contains water as a solvent is called (b) Ni(CIO4)2-6H2O is hydrated…
A: Mixture is the physical combination of two or more substances, where the identities are retained and…
Q: What happens in a dehydration reaction Select one: a. Double bond is converted to single bond b.…
A: Chemical reactions are irreversible reactions that lead to the conversion of one or more reactants…
Q: Carbon atoms are capable of forming single, double, and triple bonds because carbon is looking for…
A: Valence shell The valence shell is the set of orbitals that can accept electrons to form chemical…
Q: HYDROGENATION: I. Involves hydrogen addition across carbon-carbon double bonds II. The removal of…
A: Hydrogenation: Unsaturated fats reacts with gaseous hydrogen to yield the saturated fatty…
Q: At room temperature, all of the elements in group 18 (the noble gases) Are very reactive. Are not…
A: In the periodic table, group 18 is occupied by the noble gases. These are six in number and are…
Q: Which molecules are very dependent on amide bonds? O Proteins RNA O Amylose O DNA
A: Amide bonds is also known as peptide bonds are the covalent chemical bonds that are involved in…
Q: Nitrogen (N) normally forms three covalent bonds with a valence of five. However, ammonium has four…
A: Covalent bonding is a type of chemical linkage between two atoms that involves electron sharing. An…
Q: 4. Letter 'D' corresponds to A. reactants. B. free energy change. C. activation energy. D. products.…
A: This graph represents the relationship between Reaction progress and Potential energy. It is used to…
Q: What is the Electron Geometry of a AB2C molecule? tetrahedral trigonal planar trigonal pyramidal…
A: As no compound is mentioned specifically so Electron geometry and molecular geometry is same , which…
Q: Which one of the following statements about a molecule of acetonitrile (CH,CN) is TRUE? Select one:…
A: Chemical bonds are simply electrostatic attractive forces between atoms. There are mainly 3 types of…
Q: Which bond should be the strongest? Group of answer choices Sodium (Na) - Fluorine (F) Lithium (Li)…
A: There are mainly 3 types of bonds present between the molecules to keep themselves intact and these…
Q: For the following reaction at equilibrium NH3 + H20 e NH4+1 + OH-1 NH3 is the It is not a hydroxide…
A: In the reaction, it involves the formation of ammonium ions from ammonia by accepting the proton…
Q: How to know what the reaction is ?
A: Ionic solids are composed of cations and anions. They are held together by electrostatic forces.
Q: Which of the following is not a functional group that can bond with carbon? a. sodium b. hydroxyl c.…
A: BASIC INFORMATION CHEMICAL REACTION In this one substance gets converted into another substance…
Q: the oxidation state of the metal centres in the [Pt(SCN)2)(NH3)2]3×[Fe(C2O4)3]2 complex
A: The correct answer is option d.
Q: 23. Letter 'C' corresponds to A. reactants. B. free energy change. C. activation energy. D.…
A: The catalysis of reaction involves the reaction of enzyme and substrate molecule, which helps to…
Q: an atom (to form cations) or adding electrons (to form anions). Ions are never formed by 1. It is…
A: electrically neutral: A net charge of zero, which occurs when an atom or molecule/compound has no…
Q: Which of the following is incorrect if its anomeric C is not linked to a group or another molecule?…
A: Sugars are carbohydrates . The monomeric unit of sugars are called monosaccharides. Every…
Q: 11
A: Ans- B
Q: Another fossil-fuel gas used in cooking appliances is "propane", used commonly in backyard grills…
A: Introduction A Fossil Fuel Is A Hydrocarbon-containing Substance Generated Underground From The…
Q: Reactants A-B and B-C react according to the equation below. In what orientation must the reactant…
A: Collision is generally the theory used to predict the rates of chemical reactions, particularly for…
Q: Which of the following would be expected to form hydrogen bonds with water? Choose all that apply. H…
A: Hydrogen bond is a type of intermolecular force. It exists in molecules in which highly polar bonds…
Q: The molecule shown below is NH2 HO N. Но но
A: Deoxyribonucleic acid (DNA) is a molecule comprised of two polynucleotide chains that wrap around…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- Figure 27.3 illustrates the response of R (ATP-regenerating) and U (ATP-utilizing) enzymes to energy charge. a. Would hexokinase be an R enzyme or a U enzyme? Would glutamine: PRPP amidotransferase, the second enzyme in purine biosynthesis, be an R enzyme or a U enzyme? b. If energy charge = 0.5: Is the activity of hexokinase high or low? Is ribose-5-P pyrophosphokinase activity high or low? c. If energy charge = 0.95: Is the activity of hexokinase high or low? Is ribose-5-P pyrophosphokinase activity high or low?The Reactions and Meehanisms of the Leloir Pathway Write the reactions that permit galactose to be utilized in glycolysis. Write a suitable mechanism, tor one of these reactions.The oxidation of 1 mol of glucose supplies enough metabolic energy to form 36 mol of ATP. Oxidation of 1 mol of a typical dietary fat like tristearin (C57H116O6) yields enough energy toform 458 mol of ATP. How many molecules of ATP can form per gram of (a) glucose; (b) tristearin?
- All are single-electron carriers EXCEPT: a. UQH2. b. Cyt bL. c. Cyt bH. d. Rieske protein Fe-S clusters. e. Cyt c1. 2.The energy input for the synthesis of glycogen occurs during which of the following enzyme catalysed steps? a. phosphoglucomutase b. UDP-glucose pyrophTosphorylase c. glycogen synthase d. amylo-(1,4®1,6)-transglycosylase e. none of the above require energy input ______________________________________________________________________________(Using your diagram from #2 as the reactant, draw the full equation for this triacylglycerol undergoing saponification, using KOH.The free energy of hydrolysis of an α(1→4) glycosidic bond is −15.5 kJ ⋅mol−1, whereas that of an α(1→6) glycosidic bond is −7.1 kJ ⋅ mol−1. Use these data to explain why glycogen debranching includes three reactions [breaking and re-forming α(1→4) bonds and hydrolyzing α(1→6) bonds], whereas glycogen branching requires only two reactions [breaking α(1→4) bonds and forming α(1→6) bonds].
- What is the ∆G naught' for a phosphoryl transfer from ATP to glycerol? The hydrolysis of α-glycerophosphate to glycerol and inorganic phosphate has a ∆G naught' of -8.37 kj/mol. ATP + H2O <--> ADP + Pi ∆G naught' = -30.5 kj/mol What is the efficiency of this reaction (i.e. what % of the available energy remains in the system after the reaction)? Express your answer as a % of the total amount of energy available in the system. Where does the rest of the energy go?Part A. If you were to expose cells that are undergoing anaerobic respiration to a radioactive carbon isotope in the form of C6H12O6C6H12O6, which of the following molecules would you not expect to be radiolabeled? a)pyruvate b)succinyl CoA c)fructose-1,6-bisphosphate d)lactate Part b.If you were to expose cells that are undergoing aerobic respiration to a radioactive carbon isotope in the form of acetyl-CoA, which of the following molecules would you not expect to be radiolabeled? a)oxaloacetate b)succinyl CoA c)pyruvate d) CO2When glucose is reduced, only one alditol is produced.When fructose undergoes the same reaction, however,two diasteriomeric sugars are produced. Draw theirstructures.
- Acetocholinesterase is an enzyme possessing a single active site that metabolizesacetylcholine with a turn over number of 1.4 x 10^4s-1. How many grams of acetylcholine(molecular formula C7NO2H16+) will 2.16 x 10^-6 g acetocholinesterase metabolize in 60minutes? (The enzyme’s molecular mass is 4.2 x 10^4 g/mol).An oligosaccharide is a repeating unit of a-D-galactopyranosyl-(a-1 >3)-allopyranoside. Each disaccharide unit is linked via B-1 --->4 glycosidic bond. The oligosaccharide has 10 monosaccharide residues. Required: Is this oligosaccharide a good substrate for glycolysis? Why or why not? Provide two reasons and discuss corn prehensively.These enzymes form covalent intermediates A. Papain B. Alkaline phosphatase C. Elastase D. All of the above  This statement/s describe/s the function of TPP A. decarboxylation of alpha- keto acids B. oxidative decarboxylation of alpha- keto acids C. transketolation reactions D. All of the above True statement/s about intrinsic binding energy: A. More stable ES, slows the reaction B. Increasing the energy of ES is by either loss of entropy or destabilization of ES C. Smaller Ea between ES and ES* means faster the reaction D. All of the above

