General, Organic, and Biological Chemistry
7th Edition
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:H. Stephen Stoker
Chapter10: Acids, Bases, And Salts
Section: Chapter Questions
Problem 10.71EP
Related questions
Question
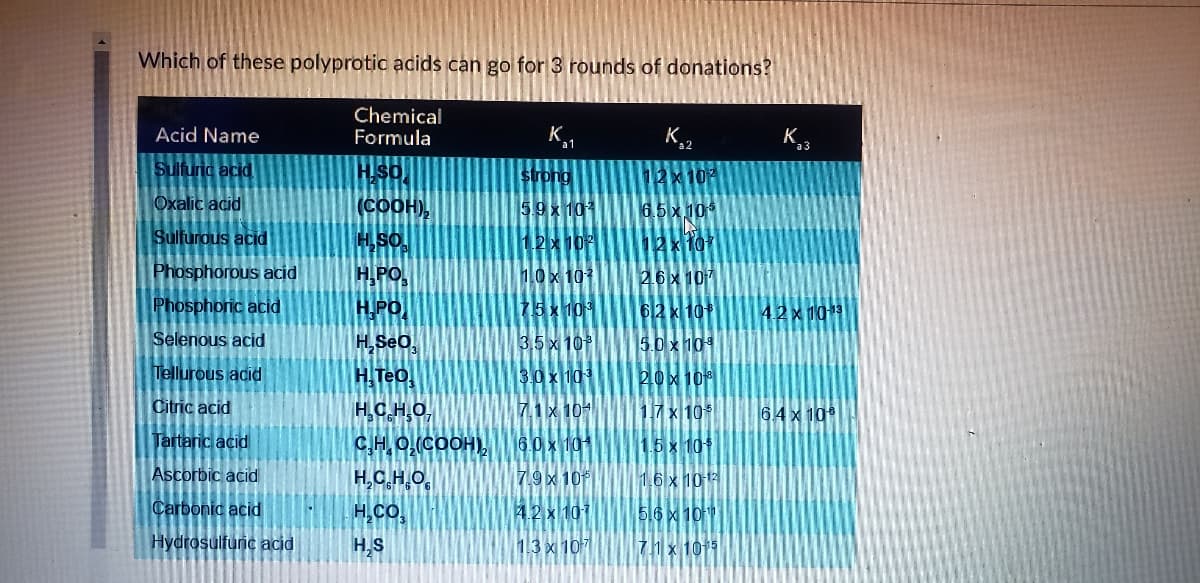
Transcribed Image Text:Which of these polyprotic adids can go for 3 rounds of donations?
Chemical
Formula
Acid Name
K
а1
K.2
Sulfuric acid
strong
12x10
Oxalic acid
(COOH),
5.9x 10
6.5x 10
Sulfurous acid
12x 10
Phosphorous acid
H,PO,
10x 101
26x 107
Phosphoric acid
H.PO
75x 10
62 x 10
4.2x1013
Selenous acid
H,Seo,
3,5 x 10
5.0 x 104
Tellurous adid
H. Teo.
3.0x 10
20x 10
Citric acid
HC.H,O,
C,H O(COOH)
H,C,H.O,
7.1x 101
17 x 10
6.4 x 10
Tartaric acid
6.0 x 101
1.6 x 101
Ascorbic acid
7.9 x 10
1.6 x 1013
Carbonic acid
H,CO.
4.2 x 101
5.6x 10
Hydrosulfuric acid
H,S
13 x 107
7h x 1015
Transcribed Image Text:Sulfuric Acid
Phosphorous Acid
None of these
Phosphoric Acid
O Carbonic Acid
Tellurous Acid
Previous Page
Next Page
Page 3 of 5
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax