Q: Starting with 0.450 g of potassium chlorate (molar mass 122.55 g/mol), a student determined that the…
A:
Q: What type of nitrogen functional group is shown in this picture? NH₂ A) primary amine B) secondary…
A: Formula of ammonia is ----- NH3 When one hydrogen atom of ammonia (NH3) is replaced by one alkyl…
Q: How much heat (in kJ) is required to vaporize a 3.86-g ice cube at -20 oC? The heat of fusion of ice…
A:
Q: An aqueous solution has a hydroxide ion concentration 1.0 x 10-8 of M. What is the hydronium ion…
A: If pH of solution is greater than seven than solution will be basic in nature.if pH of solution is…
Q: (3) Q5.18 Homework Unanswered If electricity costs $0.12 per kWh, then what is the total cost for a…
A:
Q: Match the major product in each reaction with the correct initial alkyl halide. Each alkyl halide…
A: The given strong base in methanol acts as an eliminating agent and gives the respective alkenes upon…
Q: An excited hydrogen atom undergoes a transition, in which an electron moves from the n= 3 orbital to…
A: Which one of the following is correct answer
Q: Balance the chemical equation below using the smallest possible whole number stoichiometric…
A: Given, the unbalanced equation: Ca3(PO4)2 (s) + SiO2(s) + C(s) → CaSiO3(s)+ P4(s) + CO(g) We have to…
Q: What is the concentration of 1.00 cm (path length) sample that has an absorbance of 0.60?…
A: Here we are required to find the concentration of 1.00 cm (path length) sample that has an…
Q: To understand several methods of measuring gas pressure. Gases exert a measurable pressure (P) on…
A: Given, Pressure in container = 385 kPa
Q: Determine the [H+] value for solutions with the following characteristics: a pH=9.90 [H+] = bpH =…
A: (a) pH = 9.90 Using the formula we have pH = - log10[H+] [H+] = 10-pH [H+] = 10-9.90 [H+] = 1.26…
Q: ermine the pH of water solutions with the following characteristics. Classify h solution as acidic,…
A: If pH of solution is greater than seven than solution will be basic in nature.…
Q: A solution is made by dissolving 24 g of sodium chloride, NaCl, in enough water to make exactly 100…
A:
Q: In order to make 0.500 L of a 0.131 M solution, you will need to weigh out how many grams of sucrose…
A:
Q: Which of these is/are a tertiary alcohol? нс он о A A в OH ▲ A and D В CH 3 CH3 ОН
A: Tertiary alcohol is a compound in which. -OH group is attached to a saturated carbon atom which…
Q: Can cadmium (II), and mercury (II) be separated quantitatively by precipitation as sulfides from a…
A: The solution to be separated has [Cd(NO3)2] = [Cd2+(aq)] = 0.15 M [Hg(NO3)2] = [Hg2+(aq)] = 0.10 M…
Q: TAAX O CHEMICAL REACTIONS Predicting precipitation HH CHLI…
A:
Q: Balance the following redox reaction in basic solution. MnO2(s)+Br2(l)→ MnO−4(aq)+Br−(aq)
A: Balancing of redox reaction means the number of individual atoms in the product side must be equal…
Q: Use the following balanced equation to answer the question: 3CuSo4(aq) + 2Fe(s)→ 2Fe2(SO4)3(aq)+…
A:
Q: Consider the balanced thermochemical equation given below: 4 NH3(g) + 5 02(g) -* 4 NO(g) + 6 H20(g)…
A:
Q: Draw a resonance structure that places a pi bond in a different position. Include all lone pairs in…
A: Resonance structure: If the Lewis structure of a molecule or ion cannot explain by a single…
Q: Pressure and temperature affect the amount of space between gas molecules, which affects the volume…
A:
Q: 1. Light bouncing off a lake a.) Reflection b.) Refraction c.) Absorption S
A: While traveling, when light rays pass from one medium to another medium of different refractive…
Q: What is the hydronium ion concentration of a 0.010 M solution of acetic acid? Ka for acetic acid is…
A: Answer is given below
Q: Predict the solubility of the following chemicals: Is it more soluble in water or hexane? a.CH3NH2…
A: a. CH3NH2 b. CH3CH(OH)CH3 c.CH3CH3 d. CH3(CH2)5CH3 e. NaCl
Q: 8. Determine the relative acid strength (RAS) for the anions produced from the dissociation of the…
A: we know that greater the electronegativity оf Central atom Of acid greater will be the acidic…
Q: EXTRA #1 CHALLENGE! Draw 4 Bohr Models If you chose Li for #1, then draw N or O. If you chose Na,…
A: As we go from left to right in the periodic table, the atomic radius of the atom decreases due to…
Q: why cant i see the answer? My computer cant pan to view it entirely.
A: In this question, we have to clearly solve the preceding question.
Q: What is the temperature of 0.64 mol of gas at a pressure of 1.4 atmi and a volume of 11.4 Express…
A: The ideal gas equation is formulated as: PV = nRT. In this equation, P refers to the pressure of the…
Q: The first-order reaction A-8 has k-0.210 min 1. If [Alo -0.500 M, how long will it take [A] to equal…
A:
Q: Which atmosphere represents the highest partial pressure of oxygen in the lungs? Select an answer…
A:
Q: write balanced molecular equations to illustrate the following characteristic reactions of acids,…
A: a.) Reaction of acid with a metal results in evolution of hydrogen gas. b.), c ) Reaction of acid…
Q: The common name of this compound is: H -H
A: The common name is mostly given for compounds based on atoms , substituents or functional groups…
Q: Is disaccharide “4” a reducing sugar
A: Reducing and nonreducing sugar: Carbohydrates that contain free aldehyde or ketone groups can act as…
Q: How do I apply this in the answer chart that was given? in other words how do I write out the entire…
A: Here, we have to find the mass of FeCl3 in 155 mL of 0.762 M FeCl3 solution.
Q: When a 24.7 mL sample of a 0.389 M aqueous hydrofluoric acid solution is titrated with a 0.390 M…
A: Here we are required to find the pH of the solution.
Q: The following reaction can be used to convert carbon dioxide to oxygen gas. 4 KO₂ (s) + 2 CO₂(g) → 2…
A: Given -> 4KO2(s) + 2CO2(g) -----> 2K2CO3(s) + 3O2(g) Weight of KO3 = 0.838 gm Percent yield=…
Q: How much heat will be released when 6.44 g of sulfur reacts with excess O2 according to the…
A: ♦ The given problems are based on the application of Hess's law .Some of the applications are -1.…
Q: Use the reaction quotient to predict whether a precipitate will form. If 12.0 mL of 4.69x104 M…
A:
Q: ± Changes in Temperature The ideal gas law (PV=nRT) describes the relationship among pressure P.…
A:
Q: A 4.90 L sample of neon at 7.24 atm is added to a 13.0 L cylinder that contains argon. If the…
A: Answer is given below
Q: 2. Determine the molecular formulas to which the following empirical formulas and molar masses…
A: 2) Empirical formula and molecular formula: Empirical formula: it can be defined as the simplest…
Q: 4. Calculate the pH of a solution having [H3O+] = 2.5 × 10-³. 5. If the pH of a solution is 9.8 then…
A:
Q: C. Br H₂C-CH₂ I The name of compound C is: O Search CH3 CH3
A: The naming of an organic compound can be done with help of rules of international union of pure and…
Q: mixture of gaseous reactants is put into a cylinder, where a chemical reaction turns them into…
A:
Q: If the molar mass of potassium is 39.10 g/mol and the molar mass of K2SO4 is 174,3 g/mol, then the…
A:
Q: Classify the following reaction: Pb(NO3)2 + KCl → 2PbCl2 + KNO3
A:
Q: Given the following data for the reaction AB, determine the frequency factor, A. the reaction to…
A:
Q: What is the pH of a solution that is 0.080 M in NaClO at 25 °C? The Ka for HClO is 4.0×10^-8
A: NaClO is salt made by strong base sodium hydroxide and weak acid HClO
Q: Which of these would react with CrO3? A OD О в O A OC and D Ос O A, C and D B ОН с D ОН -CH3
A: In this question, we have to select the correct option which Would react with CrO3.
Step by step
Solved in 2 steps with 2 images

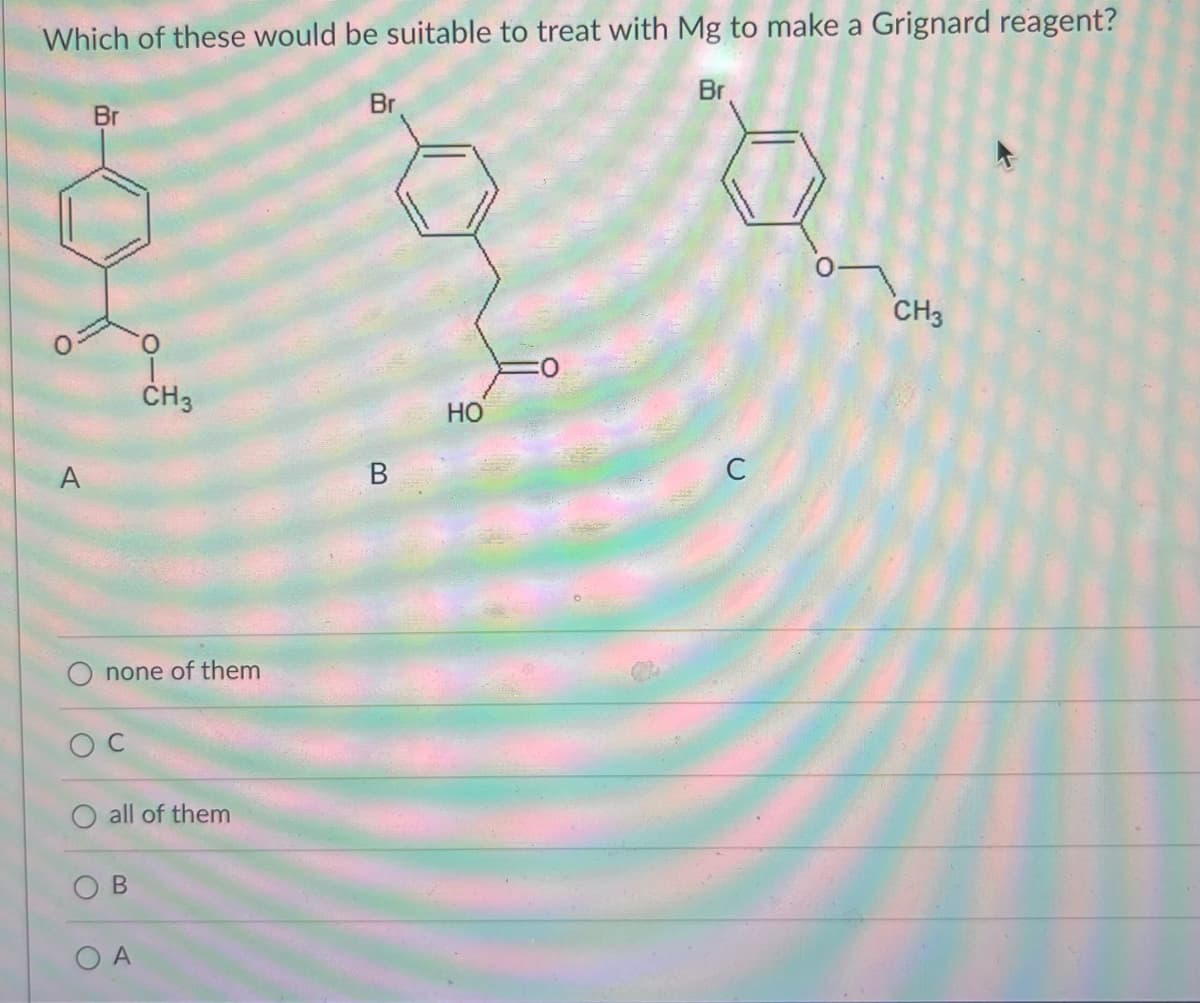
- Define the Reaction of Organometallic Reagents with Other Compounds ?(a)Which compound/s will produce an orange precipitate upon reaction with 2,4-DNPH? (b)Which compound/s will test positive towards Baeyer’s test? (c)Which compound/s will produce a brick-red precipitate upon reaction with Fehling’s reagent (CuSO4, tartrate, NaOH)? (d)Which compound/s will test positive towards the Iodoform test?Which of the following would be a suitable Grignard reagent?
- What product is made when the chemical below is heated with NBS in CCl4. NBS is N-bromosuccinimide?Supply the synthetic routes to the compounds show using starting material with six or fewer carbon atoms. Usage of triphenylphosphine as a reagent may be needed.Many brominated compounds are lachrymators. What is a lachrymator? What is the bestsafety precaution to take when working with a compound that is known to be a stronglachrymator?
- What products would form from a reaction between Potassium tert-butoxide in THF and the following compound?Briefly explain why OtBu- sometimes favored over hydroxide as an elimination reagent?What functional group cannot be present on the substrate when treating it with either an alkyllithium or Grignard reagent?
- Give the reagents and results/observation (ex. color) Test for aldehydesa. Schiff’s testb. Formalinc. Acetoned. Benzaldehydee. AcetophenoneWhich pairs below could be separated by chemically active extraction? What reagent would you use in each case?Classify following solvent as protic or aprotic CH3NO2

