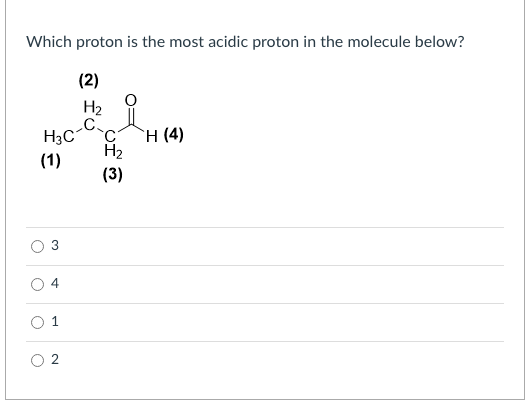
Q: Please circle most acidic hydrogen(s) below.
A: When hydrogen leave the compound then carbanion is formed , so more stable is the carbanion stronger…
Q: Which molecule is more acidic? H F F F F 2,2-difluoro isomer 3,3-difluoro isomer エ-Z エ-Z
A: This is multiple questions type So only first question is solved. F is electronegative is group and…
Q: (i) Identify the acidic proton(s) for each of the following carbonyl compounds: O O O 1 CHỊCH CÁCH,…
A: Acid is substance which release hydrogen ions and base is substance which release hydroxyl ions when…
Q: Which carbon has the most acidic proton in the molecule below? 0000 CH₂ Ofe B P₂ D
A: -> Most acidic hydrogen is that after removel which can generate most stable carbanion .
Q: Rank the compounds listed below from the weakest to the strongest acid. O 1. CGHSCO2H < CGHSCH2NH2 <…
A: Acidity of the compound is defined as the capacity of donation of hydrogen ,the compound which is…
Q: Which of the following hydrogens on the structure below is most acidic? H3- H1 Hs HA O H3 O H1 O H4…
A:
Q: .C. H2C CH2 H2C. CH2 S
A:
Q: Draw curved arrows for the following acid base reaction. Using the pKa values, predict the direction…
A: Acidity can be best explained using pKa values which is inversely proportionate to the acidity.…
Q: а) н ннн b) нн нн c) H3C CH3 H. Н. нн н ннн
A:
Q: 1. Rank the following compounds from most to least acidic at the indicated hydrogen, and explain…
A:
Q: Even though it is usually difficult to remove a proton from a neutral nitrogen atom, the proton on…
A: We are provided with a molecule. We need to find its 1. conjugate base 2. two resonating structures…
Q: 2. Which is the most acidic hydrogen in the following compound? Provide a brief explanation. H H- H.…
A: Most acidic hydrogen is that form most stable conjugate base. Conjugate base is stabilized by -R…
Q: Rank the compounds in each group in order of increasing acidity.
A: The more the electronegative substituent, the stronger the acid. This is because it weakens the bond…
Q: 40. Which of the following is the most acidic compound? H. II IV
A: In this question we have to tell that which compound is most acid. The general rule of acidity is…
Q: 4. The explicitly indicated proton in the following compound is much more acidic than a typical…
A: According to Bronsted-Lowry concept, acid is a substance which can lose proton and forms its…
Q: (4) Answer both sections () and (1). O Arrange the following compounds in order of increasing boling…
A: The given questions are detailly answered below,
Q: Which of the following compounds is more acidic? Select one O a. H3C-C EC-CH3 H3C Ob. H3C CH3 Oc.…
A: Correct answer is (c) H3CH2CC≡CH . 1-butyne (H3CH2CC≡CH) is terminal alkyne and it is most acidic…
Q: The C – H bond in acetone, (CH3)2C = O, has a pKa of 19.2. Draw two resonance structures for its…
A: The pair of acid and base differing from each other by just one proton is called conjugate acid-base…
Q: Which molecule is the most basic? X Selected Answer: Answers: [None Given] OH 'OH NH₂ "NH₂
A: According to Lewis acid-base theory A Lewis acid is a chemical species that contains an empty…
Q: 13. Even if you do not know the exact pk, values, circle the side favored by equilibrium in the…
A:
Q: ch letter is near the most acidic protons in the molecule below. A В с OD А H3C B
A: Hydrogen which bonded with more electronegative atom is most acidic Hydrogen.
Q: Q4. The following compound can become protonated on any of the three nitrogen atoms. Which nitrogen…
A: The most basic nitrogen among 1, 2, 3 has to be identified
Q: но но NO2 `CH3 1 2 3 HO но но N= ČH3 6
A: The acid and base are defined on the basis of hydrogen ion acceptor or donor. If a compound accepts…
Q: r each pair of compounds below, identify the most acidic compound: b) `NH2 HO,
A: Acidity of compound depends upon the stability of conjugate base formed. More stable is the…
Q: Rank the labeled protons in each compound in order of increasing acidity.
A: The labeled protons are in the following order of increasing acidity because- 1)acidity of proton…
Q: Rank the compounds in each group in order of increasing acidity.
A: a. When we see the first set of compound given we compare the electronegativity of the halogens ,…
Q: 6.) Rank the following in order of decreasing acidity of the most acidic proton (start with the most…
A:
Q: Which of the indicated protons is the most acidic? На Ho Hb Hc O a O All of a-d are equally acidic
A: Acidity is defined as the ability of a molecule to release protons. if a compound easily donate…
Q: Which of the following is the conjugate base of the compound shown below? H H c=C H H H A) H-c-co D)…
A: Answer 3: Introduction: The acid or base form after the losing or gaining a proton is called…
Q: For the list of hydrocarbons, rank the hydrocarbons in strength from weakest acid to strongest acid.…
A:
Q: Q.14 Arrange the following compounds in order of increasing basicity. Aay CH;NH, CH, NH , CH,NH- b…
A:
Q: CH3 -N 'H O,N- -NH2 NH2 CI - NH2 CH30- NH2
A: Basicties of aniline are because of donating property of lone pair of electrons of nitrogen
Q: CH,C-H H3C CH * CH3 II III a. Underline the most acidic hydrogens in each of the molecules. b. Rank…
A: The relative acidity of various hydrogen atoms can be determined on the basis of many factors such…
Q: Hello, I hope you are doing well. I need to know how to figure out the conjugate acid for NH2Cl. I'm…
A: Conjugate acid of a base is formed by the addition of H+ in the base.Addition of H+ means one…
Q: 5. Circle the most acidic proton in each molecule. OH OH H2N, .OH NH3
A: Acid is a substance which have tendency to donate proton . Stronger is the acid greater is the…
Q: 11) For each set, identify the strongest acid a) 1. К, %3D 6.2х 104 2. K = 4.4 X 10-5 3) K = 3.2 X…
A:
Q: 8. (8) Circle the most acidic compound from each set below. Then, list the structural influence(s)…
A: A)…
Q: 8. (8) Circle the most acidic compound from each set below. Then, list the structural influence(s)…
A:
Q: What indicated hydrogen atom is the most acidic? B A H₂ C. C H₂ C CH3 -D
A: Acidity of Hydrogen atom of a molecule depends upon stability of it's conjugate base. More is the…
Q: Which letter is near the most acidic protons in the molecule below. 0000 В с D А H3C UI A of o CH3 D
A:
Q: Which among the compounds below is the strongest base? NH
A: Basicity of base depends upon availability of lone pair. If lone pair is easily available for…
Q: 1. Identify which of the labeled hydrogens in the following compounds is the most acidic. H-…
A: Acids are those substance which gives H+ ion on dissociation. The compound which gives H+ ion either…
Q: Ketones typically have a pka of approximately 20. While this is certainly not a strong acid it is…
A: The C-H bonds in ketones (for example acetones) has pKa value of ~20. However, the C-H bonds in…
Q: Which of the circled hydrogen atoms is the most acidic? H (H) H-¢O H,C-¢O H,C-¢ H,C=¢ H,C-C=c<H II…
A: When hydrogen atom leave the compound then compound convert to anion and more stable is the anion…
Q: 27. Rank the following conjugate bases in order of decreasing basicity, putting the most basic…
A: Since you have posted multiple questions, as per guidelines we can answer 1 per session . If you…
Q: 4. Rank the following compounds in order of increasing basicity. Label the least basic compound "1"…
A: Basicity of a molecule is a measure of the ability to accept proton(H+) to form a stable compound.…
Please help me i am very confused
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

- Tenormin, a member of the group of drugs known as beta-blockers, is used to treat high blood pressure and improve survival after a heart attack. It worksby slowing down the heart to reduce its workload. Which atom in Tenormin is the most basic?(a) Which proton in A is most acidic? (b) Decide whether A is more orless acidic than B, and explain your choice.An acid has a Ka of 4.53 * 10-6 in water. What is its Keq for reaction with water in a dilute solution? ([H2O] = 55.5 M)
- Rank acidity (1-4)The pKa of ascorbic acid (vitamin C, page 55) is 4.17, showing that it is slightly more acidic than acetic acid (CH3COOH, pKa 4.74). Compare the most stable conjugate base of ascorbic acid with the conjugate base of acetic acid, and suggest why these two compounds have similar acidities, even though ascorbic acid lacks the carboxylic acid (COOH) group.Identify which of the following compounds is expected to be a stronger base. Justify your choice
- The strongest acid below is water. CHOICES: A) TRUE B) FALSEWhich among the given compounds are less acidic than compound I? II and IV II and III III and IV III and VThe pKa of three C – H bonds is given below. a. For each compound, draw the conjugate base, including all possible resonance structures. b. Explain the observed trend in pKa.
- Consider two acids: HCO2H (formic acid, pKa = 3.8) and pivalic acid [(CH3)3CCO2H, pKa = 5.0]. (a) Which acid has the larger Ka? (b) Which acid is the stronger acid? (c) Which acid forms the stronger conjugate base? (d) When each acid is dissolved in water, for which acid does the equilibrium lie further to the right?Which compound from this unbalanced chemical reaction below is the acid? H2SO4(aq) + NaOH(aq) ⟶ H2O(l) + Na2SO4(aq) Choices: H2SO4 Na2SO4 NaOH H2OIf the pKa of a weak acid HA is 3.84, what is the pkb of its conjugate base A-

