Q: Calculate the mass of water produced when 8.50 g of butane reacts with excess oxygen. 2C4H10(g) +…
A:
Q: Consider the insoluble compound silver hydroxide, AgOH. The silver ion also forms a complex with…
A:
Q: Which reaction requires the least energy? O F(g) → F*(g) + e¯ O Be(g) Be*(g) + e- O O(g) → 02+(g) +…
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: What volume of 0.445 M Na3PO4 solution, in milliliters, is necessary to completley react with 46.2…
A:
Q: How many moles of H2O2 are needed to produce 3.90 mol of H2O?
A: H2O2 decomposes to produce H2O and O2 Number of moles of H2O = 3.90 mol Number of moles of H2O2 = ?…
Q: 3-15. (a) The pH of a solution is 5.42± 0.05. Find [H+] and its uncertainty. (b) What is the…
A: Ans a. Given : pH = 5.42±0.05 hence pH = 5.42 and d(pH) = 0.05 [H+]=10−pH [H+]=10−5.42 [H+]=…
Q: 7. The formula for plumbic sulfate is a. PbSO4 b. Pb₂SO4 c. Pb(SO4)2 d. Pb(SO4)4 e. Pb4SO4 the…
A:
Q: Which of the following stresses to a system at equilibrium cause an increase in the production of…
A: Choose the correct option based on le-chatelier's principle.
Q: MISSED THIS? Read Section 16.6 (Pages 696-699): Watch [WE 16.5. For the reaction 2A (g)=B (g) + 2C…
A:
Q: Vitamin K is involved in normal blood clotting. When 0.641 g of vitamin K is dissolved in 25.0 g of…
A: Mass of the solvent (camphor) is 25.0 g×1 kg1000 g=0.025 kg The lowering of freezing point is…
Q: 2880 J 3. How much heat is required to raise the temperature of 54.5g of PC13 from 18.6°C to Cp PCl3…
A: Since you have asked for question 3 only, we are providing you a solution for the same. If you need…
Q: a. b. Predict the product(s) of the following reactions: + H₂C H H HCN + P(Ph)3 THF solvent Tollens'…
A: Aldehyde and ketones shows the nucleophilic addition reaction due to presence of polar C=O bond.…
Q: A buffer solution is made that is 0.495 Min HCIO and 0.495 M in NACIO. If K for HCIO is 3.50 x 10,…
A:
Q: 60.0 ml of a 0.0500 M solution of HNO3 is being titrated with a 0.200 M solution of NaOH. What is…
A: Limiting reactant : Limiting reactants are those reactant that are consumed completely in a…
Q: Based on the structure of these molecules, what molecular orbital electronic transitions would occur…
A: The most likely electronic transition in benzoic acid is a π→π* transition, which involves the…
Q: Calculate the pH of a solution that is 0.210 M nitrous acid (HNO₂) and 0.290 M potassium nitrite…
A:
Q: +F a plausible 2-step reaction mechanism was determined to be For the theoretical reaction: 2 B 1.…
A: Rule for determining rate law:- •Rate law is always determined using rate determining step or slow…
Q: Draw this electrochemical cell in your notes and answer the following questions. Assume the cells…
A:
Q: ▼ Part A Calculate the concentration of all species in a 0.145 M solution of H₂CO3. H₂CO3(aq)…
A:
Q: Using standard reduction potentials from the ALEKS Data tab, calculate the standard reaction free…
A: The relationship between standard free energy ∆Go and standard electrode potential Eo is: ∆Go=-nFEo…
Q: Give detailed Solution with explanation needed
A: Ketals and acetals on hydrolysis gives ketone as the main product and alcohols as byproducts.
Q: 24.98 Draw the condensed structural formula for and give the name of the amino acid formed when the…
A:
Q: The fraction of molecules with sufficient KE is given by the equation: fkE=A-ERT This fraction…
A: Since, For every reaction an input of energy to activate the reaction, this is called activation…
Q: g. h. i. H₂C H O ОН 1. KMnO4 2. H3O+ ОН CrO3 H3O+ 1. (CH3)₂CuLi, ether 2. H3O+
A: The question is based on the concept of organic reactions. we need to identify and explain the…
Q: The half-life of a reaction is the time in which the amount of reactant has reached half of the…
A: The half-life of a reaction is the amount of time required for a reactant concentration to reduce by…
Q: Listed below are the ionization energies for the first six ionizations of an imaginary element: 1st…
A: Size of the cation is smaller than the neutral atom. The attractive force of the nucleus in case of…
Q: b) Synthesize the following product starting with phenol (hydroxybenzene). NO₂ OH HO. phenol
A: The question is based on organic synthesis. we need to synthesize the product based on Reactant…
Q: The following spectrum of H-RMN corresponds to: El siguiente espectro de 'H-RMN corresponde a:…
A: The question is based on the concept of organic spectroscopy. we need to analyse the spectral data…
Q: 2) Which of these molecules is most prone to decarboxylation? a) F3C مشر د م ش *OH A *OH
A: Decarboxylation means the loss of CO2. The compound having beta keto acid will undergo…
Q: H Ph 1) NEt3, DMF, 40°C 2) mild H3O* workup e.s Ph OH minor product OH Ph major product
A:
Q: 3. Predict the products. a. b. C. H3C/II. AMA H H CH₂CH3 HI H₂O H3C/ AMAY H H HC1 CN H₂O
A:
Q: Classify each transformation as substition, elimination, or addition
A:
Q: Does a reaction occur when aqueous solutions of ammonium carbonate and silver(I) acetate are…
A:
Q: 3 Zn(s)3 Zn(�) +2 Cr3+(aq)+2 Cr3+(��) →3 Zn2+(aq)→3 Zn2+(��) +2 Cr(s) If the [Zn2+][Zn2+] is 4…
A: Answer: Galvanic cell is a type of cell that converts chemical energy into electrical energy via…
Q: Write the IUPAC name of the following saturated hydrocarbons
A: We have to name the given structures
Q: 1. You dissolve 5.00 g sodium hydrogen sulfite (NaHSO,) in sufficient water to produce a final…
A: Given, Mass of NaHSO3 = 5.00 g. Volume of the solution = 100.0 mL The pH of the solution is: Note:…
Q: 4. Think back to this image. -140 ml 15% -120 100 Na+ NO3 AgCl The ions that don't react are…
A: Spectator ions : The ions which do not participate in chemical reactions are known as Spectator…
Q: a) R =) Achiral Identify the following molecule as R, S, or achiral. 3) R c) Achiral b) S d) Cannot…
A: The branch of chemistry which deals with the properties of molecules due to different spatial…
Q: The energy E of the electron in a hydrogen atom can be calculated from the Bohr formula: R₁₂ E=- n…
A:
Q: A buffer solution contains 0.385 M ammonium bromide and 0.448 M ammonia. If 0.0245 moles of…
A:
Q: Give the organic family the compound shown here belongs to. NH₂ C-C-C-C-C-C-C The organic family the…
A: The organic compound given contains –NH2 group.
Q: Calculate the pOH of a solution of 0.1905 M CH3CH2NH3F if Kb = 6.400e-4 for CH3CH2NH2? Key Concept:…
A: Given,Concentration of CH3CH2NH3F = 0.1905 MThe Kb of CH3CH2NH2 = 6.400e-4Required,…
Q: The maximum amount of silver phosphate that will dissolve in a 0.252 M potassium phosphate solution…
A:
Q: 4. Give the structural formula of the compound Q (note the alkene isomerism). J Cat. C PCY3 Ru CI…
A:
Q: what volume of 5.00 M NaOH is used to make 4.00 L of 0.625 M NaOH?
A: Given M1 = 5 M M2 = 0.625 M V2 = 4 L
Q: Tartaric acid is the white, powdery substance that coats tart candies such as Sour Patch Kids.…
A:
Q: The following data were collected for the reaction of cyclobutane, C₂H₂(g), to form ethylene,…
A: Given: Part 1: To plot the graph of concentration versus time for the formation of C2H4 and the…
Q: Draw Lewis structures, naming the following compound, name shapes (molecular geometry example…
A: As per the answering guidelines, since you have posted a question with multiple sub-parts, we will…
Q: A mixture of 87.3 mole % methanol and 12.7 mole % isopropyl alcohol is vaporized at 101.325 kPa…
A:
Q: HQ16.59 Homework Unanswered Due Today, 11:59 PM Find the equilibrium concentration of the…
A: The question is based on the concept of chemical equilibrium. we need to calculate equilibrium…
Please provide only typed answer solution no handwritten solution needed allowed
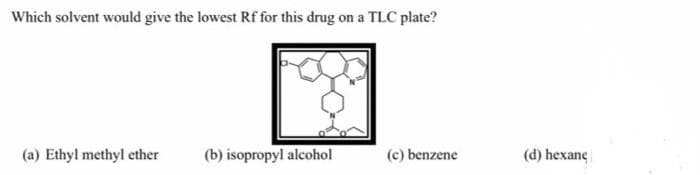
Step by step
Solved in 3 steps

- A fresh TLC plate is spotted with compound A,B and C, but with methylene chloride as a solvent. How does the change in solvent system affect the Rf value of compound A?A thin layer chromatography experiment is planned to determine the Rf values of four compounds. List the following compounds in order of increasing Rf values from shortest to longest. (If B is shortest, then A, C, and D is longest, enter BACD )For Fluorenonce and benzoic acid 1. Which compound would you expect to be the top spot on the TLC and why? 2. Which compound would you expect to be the first to come off the column and why? 3. What data supports the identification of the compound in a yellow fraction?
- Compound A had an Rf value of 0.35 in pentane and 0.45 in diethyl ether when generated on a TLC plate using pentane. Compound B exhibits Rf 0.42 in pentane and 0.60 in diethyl ether. What solvent should be used for the TLC separation of an A/B mixture? Explain.An unknown mixture was analyzed with TLC on a silica gel plate. Development gave two spots with Rf values of 1.0 and 0.0 respectively.a) Can you say for sure how many compounds are present in the mixture? If so, how many are there? Explain. b) If not, what additional TLC experiment(s) might be performed to determine how many components are present? Explain.When naphthalene, butyric acid, and phenyl acetate are separated using dichloromethane as the developing solvent on a silica gel TLC plate, which compound would have the highest Rf value? Which would have the lowest?
- Why mixture of solvents such as ethyl acetate with 5% acetic acid is used as a solvent/mobile phase for developing TLC? Please explain in detail.I had having trouble understanding the TLC labs. Could someone help explain how to tell which compound has a higher Rf value? benzoic acid or trans-stilbene?1. a compound has an Rf value of 0.97 diethyl ether but an Rf value of 0.045 in cyclohexane. Suggest a couple (at least three) solvent (solvents), or solvent system (systems), that might be better suited for this particular compound. 2. suppose your TLC plate shows only one spot at an Rf value of 0.015 (essentially it does not move). Does this mean that your sample is 100% pure? Explain your answer. 3. suppose your TLC plate shows only one spot at an Rf value of 0.98 (essentially with the solvent front). Does this mean that your sample is 100% pure? Explain your answer.
- In a gas chromatogram, the retention time of compounds A and B are 1.25 min and 1.45 min, respectively. If both of these compounds have similar structures, which of the compounds has a higher boiling point?How would you discuss method of separation and identification of compounds in TLC?Identify the statement that correctly interprets this TLC analysis of a recrystallized sample of acetaminophen produced following the procedure outlined in Experiment 5: The TLC of an individuals purified acetaminophen showed two spots. One spot had an Rf identical to that of pure acetaminophen. This spot was also larger and darker than the second spot. The Rf of the second spot was very close to the Rf of pure 4-aminophenol. a) The two spots evident in my TLC analysis indicate my sample is a mixture. Since each spot has an Rf value that is close to the Rf of one of the authentic materials tested, I can conclude my sample is a mixture of acetaminophen and 4-aminophenol. The large size of the spot with an Rf nearly identical to authentic acetaminophen indicates my sample is mostly 4-aminophenol. b)I can conclude from this evidence that my sample is a mixture of acetaminophen and 4-aminophenol because the Rf of each of the spots is nearly identical to one of the authentic…



