Q: Which of the following is/are correct resonance structure(s) for compound A? O A OI and II O II and…
A: Resonance structure are basically group of two or more lewis structure that describe the…
Q: O REPRESENTATIONS OF ORGANIC MOLECULES Identifying isomers and resonance structures Determine the…
A: The compounds with the same molecular formula but different structures are known as isomers. If the…
Q: 1.88/ The curved arrow notation introduced in Section 1.6B is a powerful method used by organic…
A:
Q: Considering bond dissociaton energy, which of the following bonds is the least reactive? O a) H-H O…
A:
Q: Which of the following has resonance? O a. SCI6 Ob. H20 Ос. Оз od. CH4 e. At least two of the…
A:
Q: Assign formal charges to each carbon atom in the given species. All lone pairs have been drawn in.…
A:
Q: Consider compounds A-D, which contain both a heteroatom and a double bond. N. H. A B a. For which…
A: Resonance structure:If the Lewis structure of a molecule or ion cannot explain by a single structure…
Q: Bonds Bond Energy (kJ/mol) C-H 411 C-Br 285 H-Br 362 Br-Br 190 Determine ΔH for the reaction:…
A:
Q: Identify the pairs that are not resonance structures. and H3C-O-N=o: H3C-O=N-O: H3C-0-N=O: and H3C-…
A:
Q: 23. Write another resonance structure for ethyl acetate. Include formal charges. CH ÖCH,CH,
A: Resonance is the phenomenon in which the lone pairs and/or the double bonds delocalize within the…
Q: How are the molecules or ions in each pair related? Classify them as resonance structures, isomers,…
A: a.
Q: What is resonance contributor in which one or more atoms bears a formal change and the most stable…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 66. This structure is an example of bond. OC-H o*c-cio C-CI pi O sigma
A: Pi (π) bond formation takes place by parallel orientation of the two p orbitals and by side by…
Q: which bond is the most polar? O O-F O N-F O O-CI O S-F O S-CI
A: For polarity of bond check the electronegativities of the atoms involved in each bond the greatest…
Q: Which bond is most polar? O A. O–H O B. F-H O C. N-H O D. CI-H
A: Which bond is more polar ? A. O-H B. F-H C. N-H D. Cl-H
Q: 3a. A neutral nitrogen covalent bonds and has 3b) A negatively charged carbon atom typically forms…
A:
Q: Q3/ A- Give the chemical structure form A-F. 0-CH, OH HNO, HCI (B) less than 5 °C HNO, Fe/HCI (A)…
A: The steps involved in this organic transformation are nitration, reduction of nitro group,…
Q: Which of the given resonance structures (A, B,or C) contributes most to the resonance hybrid? Which…
A: Most contributing structure as be seen on the basis of stability and stability can be seen as:…
Q: 1. Which of the following pair does not represent resonance structures? O.. C=N-O: and 20 O :C=N=O…
A: We have to choose option that is not a resonance structures.
Q: What are resonance descriptions for the following molecules? a. NO2- [] O=N=O
A: The correct resonating of the following given species is as follows:
Q: Which of these is not a valid resonance structure of A? :0:0 :0: :0:0 ;0: A B A) D (B) they are all…
A:
Q: Which pair(s) of structures below are resonance contributors to the same resonance hybrid? II II I…
A: Solution Resonance structures are sets of Lewis structures that describe the delocalization of…
Q: Single Bonds Multiple Bonds H-H 432 N-H 391 149 c=C 614 H-F 565 N-N 160 |-CI 208 C=C 839 H-CI 175…
A:
Q: Rank the resonance structures below from most to least important. :0: :0: H. H. H. 3) 2) 1)
A: Resonance structure: When more than one Lewis structure can be drawn, then the molecule or ion is…
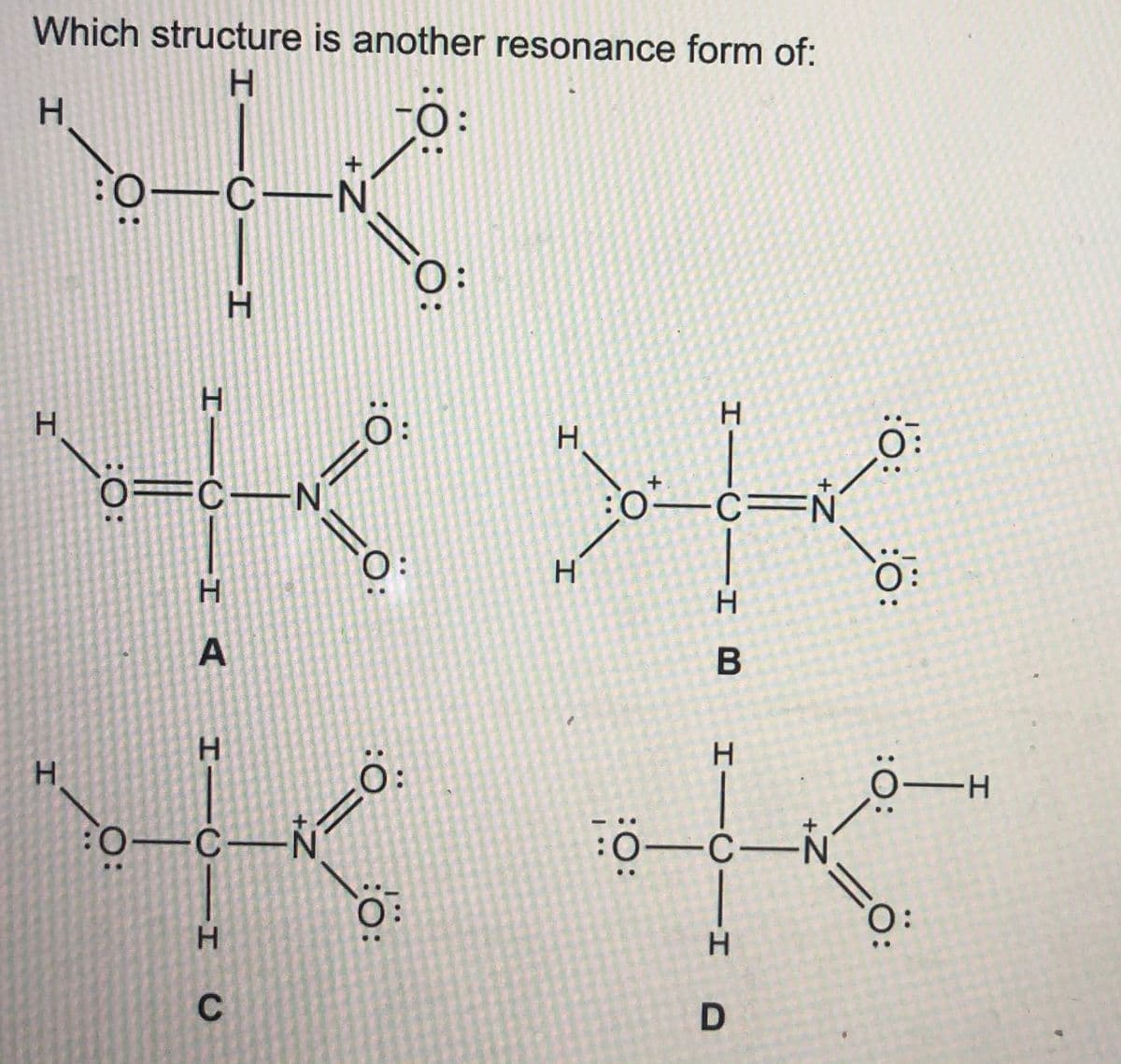
Q: Vhich structure is another resonance form of: H :0: H-C-S- :0: H. :0 H. H C-S H CS= H. :0: :0: A :0:…
A: Resonance is the process in which lone pair of electron from one atom is delocalised to adjacent…
Q: Decide wnether each row is a set of resonance structures for a single molecule and select "yes" or…
A:
Q: Sort the following as valid or invalid resonance structures for HCOO (formal charges are not shown).…
A:
Q: proposed Lewis structure Is this a reasonable structure? :o: O Yes H-C-H O No H, H. či: O Yes O No H…
A:
Q: Label each pair of compounds as resonance structures or not resonance structures.
A: The movement of pi electrons within the molecule to give rise different contributing structure is…
Q: Which of the following bonds is the most polar? O N--H O P---H O C-H O B--H
A: Given Bond N----H P----H C -----H B -----H Most polar bond = To be determined
Q: For each full Lewis structure below, draw a bond-line structure. H. ннн b) : Br-C-с-с-н н нно-н н…
A: Change the given lewis structure into bond line structure-
Q: 50. Calculate the formal charge of oxygen in the compound H H H Mark only one oval. -1 1 none of the…
A: Formal charge The formal charge over an atom of a polyatomic molecule or ion is the difference…
Q: Calculate the formal charge on each indicated atom (a and b) in the given molecules. All lone pairs…
A: The formal charge on each indicates atom has to be given,
Q: Part 2: Combustion of Ethanol Equation for Enthalpy AH = Haonds broken- Hoonds formed The following…
A: Option B. 5974KJ/mol is correct Given combustion reaction of ethanol; C2H5OH + 3O2 ------>…
Q: Consider the resonance structures below. Which resonance structure is the MAJOR contributor to the…
A: Given structures are : Which resonance structure is the MAJOR contributor of the true structure ?…
Q: Consider the resonance structures below. Which resonance structure is the MINOR contributor to the…
A: we have to identify the minor contributor from the resonance structures
Q: Which bond is most polar? C-F C-O C-I C-Cl C-Br
A: In chemistry, a polar bond is a type of covalent bond between two or more dissimilar atoms, in which…
Q: 5. Do the following structures represent resonance structures? If yes, identify them by stating the…
A:
Q: 4. Which of the following structures have a formal charge on at least one atom? H. HN: H--0-H H-C-F:…
A: Formal charges establish the comparison between electrons around a neutral atom and electrons around…
Q: Which of the structures shown below has two sigma (o) bonds? H-Ċ-H H-Ñ=ö: :Ci-C-Ċi: :CEO: O CCI20 O…
A: Single bond has only one sigma bond Double bond have one sigma and one pi-bond. Triple bond have…
Q: Part 1-Combustion of Methane Equation for Enthalpy AH = Honds broken- Hoonds formed The following…
A: The enthalpy of the combustion reaction is calculated by measuring the enthalpy of the reaction. The…
Q: Which of the structures shown below has three sigma (o) bonds? H-C-H H-N=ö: Ci-C-Ci: :CEO: O CH4 O…
A:
Q: Consider the resonance structures below. Which resonance structure is the MAJOR contributor to the…
A: In this question, we will check that which resonance structure is the major contributor. In the…
Q: 2. Which of the following pairs represent resonance structures? H2N & *HN & NH NH2 & H3C CH3 H3C CH2…
A: Of the given pairs of compound, some are pair of resonance structures and some are isomers.…
Q: Average Bond Energies (kJ/mol) Single Bonds Multiple Bonds H-H H-F H-CI 432 N-H 391 149 C=C 614 565…
A:
Q: A forms.office.com/Pages/R 14 Which of the following pairs are NOT * ?resonance structures (ähäi 2)…
A: Ans
Q: which of the following is the weakest bond ? A. C-C B. C=C C. C ≡ C
A: The answer is given as follows C-C has weakest bond
Q: Label the resonance structures in each pair as major, minor, or equal contributors to the hybrid.…
A: The lone pair on atoms can delocalized to form different resonating structures. These resonating…
please help with the following!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images

- Consider Lewis formulas A, B, and C:(a) Are A, B, and C constitutional isomers, or are they resonance contributors? (b) Which have a negatively charged carbon? (c) Which have a positively charged carbon? (d) Which have a positively charged nitrogen? (e) Which have a negatively charged nitrogen? (f) What is the net charge on each? (g) Which is a more stable structure, A or B? Why? (h) Which is a more stable structure, B or C? Why? (i) What is the CNN geometry in each according to VSEPR?The C=O double bond is called a “carbonyl bond.” Acetone and othercarbonyl compounds are introduced in some texts along with this structure a. Is this an important resonance structure of acetone? Explain. b. Does this structure convey any useful information about acetone? If so, what?Which is the most contributing resonance structure from this: Explainnnnn
- A. Find the moleular formula of diazomethane. b. Draw iits lewis structure and resonance forms as neccessary c. Explain why diazomethane is not very stable25.Which of the following does have H- bond? electronegativities: ) = 3.5, H = 2.1, C = 2.5, Cl = 3.0, S = 2.8 O || I. H- C- O-H II. SCl2 III . H | H - C - S -H | H IV. H H - C - O - H | H a.IIIb.IIc.I, II, IVd.I and IVArrange the following X---H bonds from smallest electronegativity difference to largest electronegativity difference. Si---H O---H N---H F---H C---H
- Which has the higher electronegativity? C or O? Br or Cl? Pb or H? O or H? C or H?How do you do number 112? I think the greater the stengtg of the h bond, the more energy relaesed , the more stable, thus greater expthermic. Thst makes f + fhf the strongest h bond as f is most electronegLabel each non-hydrogen atom in the structure below with its formal charge
- #1: What is the formal charge of bromine in the structure of bromate, BrO3-? a) 0 b) +1 c) +2 d) -1 #2: Carbonate (CO32-) has three resonance structures. Choose the stayement that best expains the meaning of those 3 resonance structures. a) Carbonate consists of two single CO bonds and one double CO bond. b) Carbonate consists of one single structure which is an average of the three resonance structures. c) Carbonate quickly flips back and forth between three structures. d) Three different structures for carbonate coexist. #3: When compaing a carbon-carbon double bond (C==C) and a carbon-carbon single bond (C-C), the double bond is a) longer and shorter b) longer and weaker c) shorter and stronger d) shorter and weakerWhat would be the formal charge on Se and S and both Os for the most stable resonance structures of SeSO22- (considering it's stabilized by minimizing formal charge) ? Group of answer choices Se(+1) , S(-), O(-), O(-) Se(-) , S(+1), O(-1), O(-) Se(-) , S(0), O(+), O(-) Se(0) , S(-), O(-), O(0) Se(0) , S(0), O(-), O(-)Chemistry (a) Write three more resonance structures for each of compounds 1 and 2. (b) In each of compounds 1 and 2, determine which resonance structure contributes the most and explain your answer. (c) Are the 3/4 structures resonance structures or different compounds? Same question for 5/6 structures. Explain your answers.

