Which uncharged amino acids (at pH 7) have the tendency to become positively charged? O Asparagine and Glutamine O Serine and Tyrosine O Arginine and Glutamic Acid O Glutamine and Threonine QUESTION 5 The product of the glyxoylate cycle is O Glucose O Malate O Oxaloacetate O Succinate
Which uncharged amino acids (at pH 7) have the tendency to become positively charged? O Asparagine and Glutamine O Serine and Tyrosine O Arginine and Glutamic Acid O Glutamine and Threonine QUESTION 5 The product of the glyxoylate cycle is O Glucose O Malate O Oxaloacetate O Succinate
Biology 2e
2nd Edition
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:Matthew Douglas, Jung Choi, Mary Ann Clark
Chapter7: Cellular Respiration
Section: Chapter Questions
Problem 9RQ: GTP or ATP is produced during the conversion of isocitrate into ketoglutarate succinyl CoA into...
Related questions
Question
Chemistry
please answer me this question as soon as possible
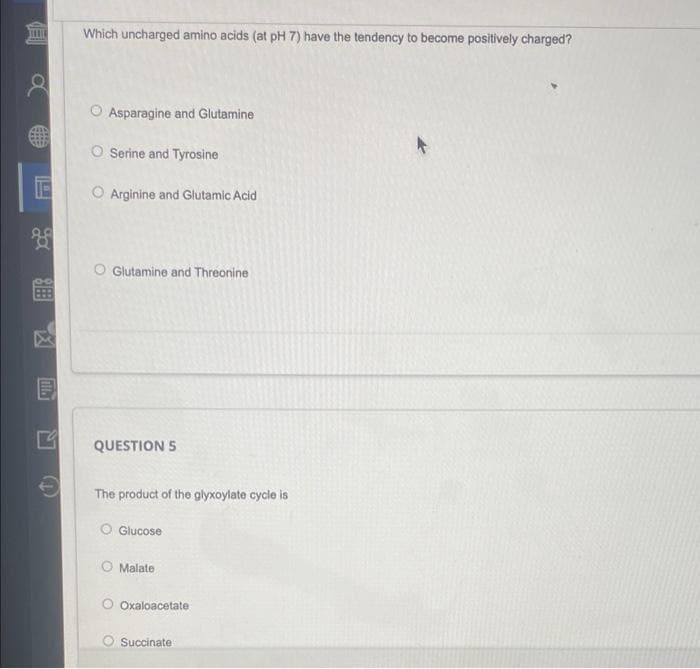
Transcribed Image Text:ED
Which uncharged amino acids (at pH 7) have the tendency to become positively charged?
O Asparagine and Glutamine
+
O Serine and Tyrosine
15 O Arginine and Glutamic Acid
O Glutamine and Threonine
QUESTION 5
The product of the glyxoylate cycle is
O Glucose
O Malate
aff
ch
SIEBI
E
Oxaloacetate
Succinate
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 6 steps

Recommended textbooks for you

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning