Q: A student takes a sample of KOH solution and dilutes it to 100.00 mL of water. The student…
A: Dilution of solution can be done by adding water in it and the concentration of original solution…
Q: An analytical chemist weighs out 0.029 g of an unknown monoprotic acid into a 250 mL volumetric…
A: Assuming the monoprotic acid is HA. Hence the reaction between the acid and base is HA + NaOH…
Q: A trainee in a medical lab will be released to work on her own when her results agree with the…
A: Below are the data obtained by the trainee: Mean concentration = 14.57 mg/dL standard deviation, s =…
Q: When you walked in to lab today, you noticed that your partner group had a 0.05432 ml mixture sample…
A: The mixture which is immisible can be separated easily by various techniques. For example: Water can…
Q: What type of glassware is used for quantitative work? A. Volumetric flask B. Beaker C. Erlenmeyer…
A: Given : Different type of glassware used for quantitative work. To find : which type of glassware…
Q: Which piece of laboratory equipment can make the most precise measurements? Top-loading balance (in…
A: In chemistry laboratory we always prefer an instrument which is having more no. Of measuring units .
Q: 2. The densities of Methane at °C were measured at several pressures with the following results:…
A:
Q: In a gravimetric determination of lead content in brass, there is a constant loss by solubility of…
A: Given: The mass of lead lost by solubility is 1.55 mg. The mass percentage of lead present in 0.3514…
Q: An analytical chemist weighs out 0.120 g of an unknown diprotic acid into a 250 mL volumetric flask…
A: Unknown Diprotic acid = 0.120 gm, its formula will be H2A The reaction of NaOH with H2A
Q: I am allowed to work with someone I just don’t understand this Using the Volumetric Pipet Data from…
A: 10 mL graduated cylinder is most accurate and 100 mL beaker is least accurate.In the 10 mL graduated…
Q: Imagine that you accidentally used 30 mL of water instead of ~20 mL to wash down your flask in the…
A: Acid-base titration of vinegar is done in laboratories to find out the concentration of acetic acid.…
Q: Part B Before investigating the scene, the technician must dilute the luminol solution to a…
A: Luminol Concentration = 6.00 × 10-2 M = 6.00 × 10-2 mol/L Volume of Diluted luminol Solution = 2.00…
Q: What volume of titanium isopropoxide is required to form 10 ml of a solution in which it is 0.2 M?…
A:
Q: A. Types of Error. A group of students is doing an analysis of copper in a 1-peso coin. They were…
A: 1) Makisig forgot to calibrate the analytical balance before weighing. Solution : Determinate error…
Q: Use the lab introduction to answer the following question. When answering this problem, report the…
A: Volume of beverage powder sample mixture = 3.78 L Volume of sample taken for dilution (V1) = 10 mL…
Q: The following image depicts a centrifuge rotor with six sample holders. The blue sample holders…
A: To use a centrifuge rotor, we need to balance it. To balance it - All sample tubes should be evenly…
Q: Ethylene glycol, commonly used as automobile antifreeze, has a specific gravity of 1.1088 at room…
A: Given values, Specific gravity is defined as the density of substance divided by density of water…
Q: coffee cup is found to have a volume of 263 mL. Using unit analysis, show what the volume of this…
A: By unit analysis we can conclude that, 1 L = 1000 ml OR 1000 ml = 1L
Q: Determine the amount of potassium chloride in each solution. 21.7 g of a solution containing 1.04%…
A: Mass percent of the solution is equal to the ratio of mass of solute and mass of solution multiplied…
Q: Stockroom Workbench 1 Information Name: AgNO3 Volume: 54.646 ml Solution 3 ARNO, 100.74 mL@ 25.0°C…
A: Given: The chemical is AgNO3 i.e. Silver nitrate. The volume of AgNO3 =100 mL The mass of AgNO3…
Q: Conversion of 23.12 mm2/s to kg/ms
A:
Q: The Daily Value for phosphorus is 900 mgmg. How many grams of phosphorus are recommended? Express…
A:
Q: Wine goes bad soon after opening because the ethanol (CH,CH,OH) dissolved in it reacts with oxygen…
A: The balanced reaction of ethanol with oxygen to form water and acetic acid is as follows:
Q: A student sets up the following equation to convert a measurement. (The ? stands for a number the…
A:
Q: During the experiment, you recover 4.7 mg of Fe by testing 100 mg of powder. Assume a full pill…
A:
Q: A blood concentration of 0.065M ethyl alcohol (EtOH) is sufficient to induce a coma. At this…
A: Since concentration = moles of solute / volume of solution in L Solute here is ethanol And volume of…
Q: The density of methanol at 20C is 0.791g/mL. What is the mass in grams of 280mL sample.
A: Given : Density of methanol = 0.791 g/mL And volume of sample = 280 mL
Q: Show all work using the dimensional analysis. You must show all work including each unit for full…
A: The given reaction is, 2 Ni(s) + 6 HCl(aq) → 2 NiCl3(aq) + 3 H2(g) Given, moles of…
Q: Answer Bank Iron(s) NaCl(s) Sand(s) Naphthalene(s) iron (s) naphthalene (s) sublimation dissolve in…
A: The compounds can be separated from the mixture on the basis of their chemical and physical…
Q: Part A Water and rubbing alcohol have distinct properties and will react differently with the same…
A: Rubbing alcohol is isopropyl alcohol (CH3-CH2-OH). It has a dielectric constant of 19.92 whereas…
Q: 3. Convert 5.0 Tbytes to Mbytes. 5tutesx 0.000001 mby- ts - 5.00 0no mbytes 1 चि je %3D 1 fera…
A: Since you have posted a question with multiple questions, we will solve first question for you. To…
Q: What is the uncertainty of 125.00 ml using a 25.00 (+/- 0.04) mL volumetric pipet? Type your numeric…
A: Uncertainty: In chemistry it is used to indicate the true value that lies between two limits.
Q: Mass of solvent in kilograms. Hint: Mass solvent in grams was 151.5 Use the metric definition 1…
A: Given: Mass of solvent in g = 151.5 g. Since 1 Kg = 1000 g.
Q: You plan to prepare 500 mL of 0.01 M NaCl solution, and you have 5.00 L of a 1.00 M stock solution…
A: Dilution of the solution:When we dilute a concentrated solution with pure water, the mole of solute…
Q: Convert 0.73 pound-foot/second square to dyne complete solution using dimensional analysis
A: A numerical problem based on dimensional analysis, which is to be accomplished.
Q: -> Ô https://app.101edu.co Question 25 of 41 A patient is required to take an IV drug for cancer…
A:
Q: f an industrial worker knows that his chemical process is 87.3% efficient and he needs to collect…
A: Percent yield=actual yieldtheoretical yield×100It is given that chemical process is 87.3% and 100.0…
Q: Que: I would like you to submit a family recipe along with all instructions for preparing the food…
A: Most common family recipe is Chilli Chicken that can be made at home.
Q: Answer Bank Iron(s) NaCl(s) Sand(s) Naphthalene(s) iron (s) naphthalene (s) sublimation dissolve in…
A: The mixture given consist of iron, NaCl, Naphthalene and sand. Since the iron has magnetic…
Q: www-awn.aleks.com O MEASUREMENT Deducing the unit missing from the solution to a basic qu.. A…
A: The given equation is
Q: 1. Identify the metal used in the experiment by computing the specific heat and solve for the…
A: Given - Mass of the metal = 6.25 g Initial temperature of the metal = 98°C…
Q: Below is a data table from an experiment in which an effervescent tablet is added to a test tube of…
A: The data given is,
Q: A student takes a sample of KOH solution and dilutes it to 100.00 mL of water. The student…
A:
Q: Gold occurs in the ocean in a range of concentrations of 0.1 to 2 mg gold per ton of seawater. Near…
A: Given recovery = 64% 1 troy = 31.1g Step 2 Recovery us 64% So amount of gold that can be…
Q: Given: wt NaOH = 10.0g Density of NaOH= 2.16 g/ml Wt. H2O = 200g Density of H2O=…
A: Molarity of a solution may be calculated by dividing the number of moles of solute by the volume of…
Q: The concentration of cholesterol (C27H460) in blood is approximately 5.0mM. How many grams of…
A: Given: The concentration of cholesterol in blood = 5.0 mM We have to find the mass of cholesterol in…
Q: Part B Vinegar is a solution of acetic acid in water. If a 225 mL bottle of distilled vinegar…
A:
Q: fter performing precipitation gravimetry, Asta placed the weighing form inside the crucible, heated…
A: To determine the value of mass that should be used for the calculations after performing…
Q: Gold occurs in the ocean in a range of concentrations of 0.1 to 2 mg gold per ton of seawater. Near…
A: Given recovery = 64℅ 1 troy = 31.1g
Q: An analytical chemist weighs out 0.073 g of an unknown triprotic acid into a 250 mL volumetric flask…
A: Given : Mass of triprotic acid = 0.073 g Concentration of NaOH = 0.1500 M And volume of NaOH…
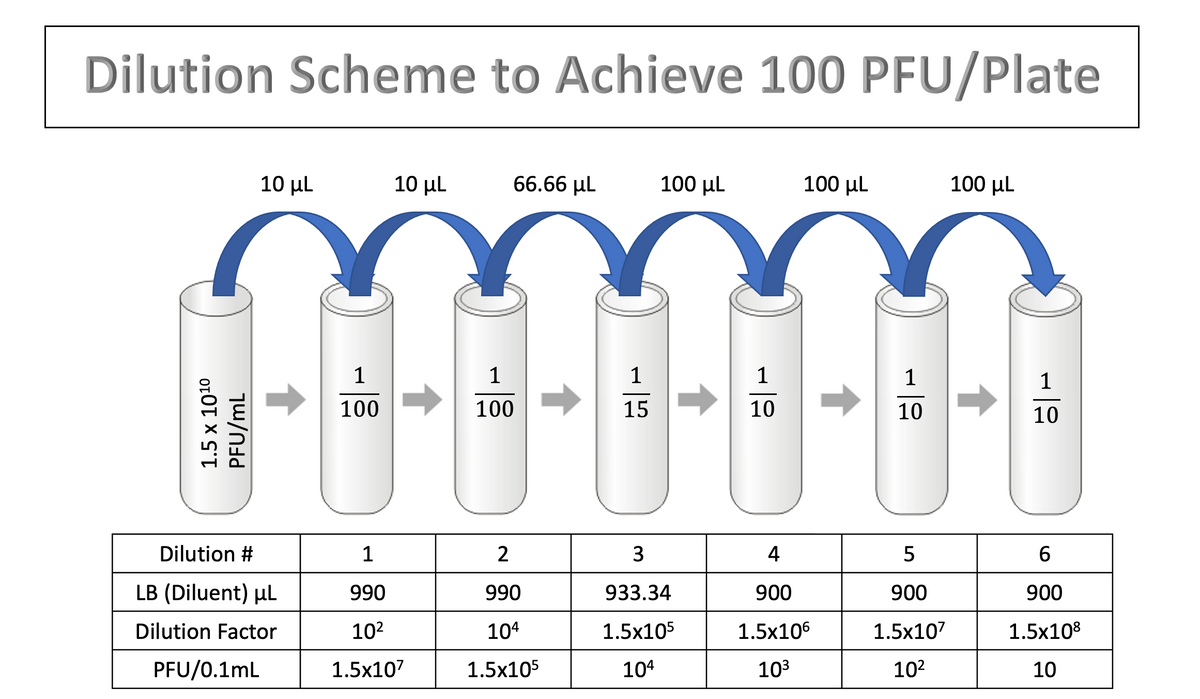
The technician then plates 0.1mL of the last three dilutions in duplicate. Why are the last three dilutions plated, as opposed to only one dilution? Why are the dilutions plated in duplicate?
Step by step
Solved in 2 steps with 1 images

- Standardization of Sodium Thiosulfate Solution Primary Standard used: Potassium Dichromate Formula mass of 1o standard: __________________ % Purity of 1o standard: 99.80% Trials 1 2 3 Weight of K2Cr2O7 (g)Weight of K2Cr2O7 (g) 0.0315 0.0331 0.0380 Final Volume Reading Na2S2O3 (ml) 27.50 27.50 34.90 Initial Volume Reading Na2S2O3 (ml) 1.10 0.00 1.00 Net Volume Na2S2O3 used (ml) Molarity of Na2S2O3The gravimetric factor used to express CoCBr6·H20 in a sample that is finally weighed as PbClBr is choose below: FW PbClBr / 6 x FW CoCBr6·H20 FW CoCBr6·H20 / FW PbClBr FW CoCBr6·H20 / 6 x FW PbClBr 6 x FW PbClBr / FW CoCBr6·H20Standard deviation- 0.0371 mL, 0.0392 mL, 0.0392 mL, 0.03887 mL, 0.0398 mL Mean- 0.194 mL
- What weight of mercury bichloride should be taken as sample, assuming 100% purity, so that 0.5 g og HgS will be obtained in a gravimetric analysis? Gravimetric factor = 1.1669I would need help with these questions. The method referenced above was followed by a student and she got the following results: Caffeine Std. Conc. (ppm) Absorbance 100 1.806 50 0.899 40 0.724 30 0.545 20 0.365 10 0.183 Further, she analyzed an unknown sample and she got the following results: Sample # Absorbance 1 0.398 1. Graphically find the concentration in ppm and then calculate in mol/L of Caffeine in Sample #1? 2. Which solvent was used to extract Caffeine and why? Why is it necessary to do extraction three times? 3. Cuvettes used in this experiment were made from which material and why? 4. At what wavelength the Absorbance was measured?To determine the organic material in a dried lake bed, the percent carbon by mass is measured at two different locations. To compare the means of the two different locations, it must first be determined whether the standard deviations of the two locations are different. Location 1 standard deviation: 0.050 Location 2 standard deviation: 0.34 What is the calculated ? value for comparing the standard deviations?
- y= mx+ b Slope () = -4E-09 y-intercept () = 0.4021 and OD= 0.540 , Calculate the concentration (x)Concentration of AR Stock Solution (ppmppm) 21.22 Unrounded Rounded Concentration of AR Stock Solution (μMμM) 42.7461 42.7Standard deviation 0.6318 mL, 0.1216 mL, 0.1056 mL, 0.0681 mL, 0.0056 mL Mean-0.18654 mL
- To determine the organic material in a dried lake bed, the percent carbon by mass is measured at two different locations. To compare the means of the two different locations, it must first be determined whether the standard deviations of the two locations are different. Location 1 Standard deviation = 0.050 Location 2 standard deviation = 0.34 a) What is the calculated ? value for comparing the standard deviations? Fcalc = __________________If a patient added a 17-g measured dose of polyethylene glycol 3350 (miralax) to 180 mL of water to use as a laxative. If the given volume of the resultant mixture was 195.6 mL. Now, determine the apparent density of polyethylene glycol 3350 and the specific gravity of the mixture.Measurements of boiling temperatures of samples of a liquid (°C) are given below: 78.4 79.0 78.8 79.2 78.6 Calculate the meanCalculate the modeCalculate the standard deviation

