Q: 2. What mass of insulin must be dissolved in 38.0 mL of water to produce a solution with an osmotic…
A: Since you have asked multiple questions, as per the guidelines, we will provide you with a solution…
Q: Three moles of FeCl2 contains THREE atoms True False
A: 1 mole of any substance contains Avagadro number(NA) of atoms ,i.e. 6.022×1023 atoms Number of…
Q: What will be the carboxylate ion produced when 3-methylhexanoic acid ionizes in water? مله O…
A:
Q: Note: If there is a mistake in a row, only one of the three quantities listed is wrong. Also, you…
A:
Q: of 16 Macmillan L It took 835.5 J to raise the temperature of a sample of water from 17.5 °C to 33.5…
A:
Q: Ok what about this one? I will also put the answer I got in there so you can check if I'm correct or…
A: We have to tell whether the given structure is correct or not. Introduction: IR and NMR…
Q: Calculate the molar solubility of Ba3(PO4)2 in a solution of 0.120M BaCl2. Ksp for Ba3(PO4)2 is 3.40…
A:
Q: a weak Dase fal cigarette smoke. Pyridine ionizes in water as follows: C5 H5N(aq) + H₂O(1) C5H5NH+…
A: Pyridine is weak base hence it dissociated feabily in aqueous solution
Q: A chemist makes 0.930 L of zinc oxalate (ZnC₂O) working solution, by adding distilled water to 0.240…
A:
Q: Give a clear handwritten answer with explanation..please give answer sub parts...
A:
Q: Wrong answer
A: Equilibrium constant at constant pressure, Kp is equal to the product of the partial pressure of the…
Q: If 70.0 grams of carbonic acid are sealed in a 2.00 L soda bottle at room temperature (298.15 K) and…
A:
Q: To what pH should a solution that is 0.072 M each in Ca²+ and Cd²+ ions be adjusted in order to best…
A:
Q: ration of 25.0 mL of 0.125 M acetic acid (CH3COOH, K₂ = 1.8x105) with 0.125 M NaOH, what volume of…
A: Molarity is defined as number of mole of solute divide by volume of solution in liter
Q: Metallic iron can be made by the electrolysis of molten Fe2O3. (a) What mass of Fe is formed by…
A:
Q: Draw the appropriate curved arrows and products for each set of reactants undergoing a coordination…
A:
Q: Find the electron and molecular geometries around the central atom. Is the Electron Geometry: bent,…
A: Since, According to the VSEPR theory, steric number (which represent hybridization) is the…
Q: A chemist adds 150.0 mL of a 6.8M silver nitrate (AgNO3) solution to a reaction flask. Calculate the…
A:
Q: Which alkyl halide below reacts the fastest in an E1 reaction ? I Br Select one: A. III B. II C. I…
A: Organic reactions are those in which organic reactant react to form organic products. In the given…
Q: Suppose a drug molecule binds to a protein target. Complete the table which describes possible…
A: The types of non covalent interactions which are responsible for the binding of protein to drug…
Q: @33 °C Cr(H₂O)6³ +SCN ---> Cr(H₂O)5NCS² + H₂O(1) Initial Rate 2.0x10-¹1 2.0x10-10 9.0x10-10 2.4x10-⁹…
A: “Since you have posted a question with multiple sub-parts, we will solve the first three subparts…
Q: What is the molar solubility of Ca(OH)2 in a 0.340 M NaOH solution? The Ksp of Ca(OH)2 is 6.5 x…
A:
Q: 1. If the hydrogen ion concentration is a measure of the strength of an acid, how much stronger is…
A:
Q: Which of the following has the largest F-X-F bond angle? Question 8 options: A) BrF3 B) N2F4 C)…
A:
Q: Predict the major organic product formed when the compound shown below is heated with CH3NH₂.…
A: Aminolysis reaction - ester can convert into amides when it heated with amines.
Q: Calculate the pH of each of the following strong acid solutions. (a) 0.00640 M HIO4 pH = (b)…
A: We will find H+ concentration and then calculate pH.
Q: The enthalpy change for the reaction of hydrogen gas with fluorine gas to produce hydrogen fluoride…
A:
Q: Give the IUPAC name of the compounds below. Note that "acetic" is considered as the IUPAC standard.…
A:
Q: Draw the skeletal line bond structure of (r)-3-fluorobutanoyl chloride
A: Given -> R-3-fluorobutanoyl chloride
Q: Which of the highlighted chemical bonds in the molecules below is longest? Shortest? In between?…
A:
Q: Give the organic product(s) formed in the following reaction: CH3 Br to CH3 I III Select one: O A.…
A:
Q: Standard Reduction Potentials (Volts) at 25 °C Cl2 (g) + 2 e- 2 Cl- (aq) 1.360 O2(g) + 4 H3O+(aq) +…
A: The process of electrolysis is a combination of oxidation and reduction. When any element releases…
Q: How many secondary carbons are in the following hydrocarbon molecule? C-C- с C13 Umu. С с J-U L с -…
A: A carbon atom that is bounded to two other carbon atom is a secondary carbon atom...designated by…
Q: Benzoic acid is a weak acid that has antimicrobial properties. Its sodium salt, sodium benzoate, is…
A:
Q: In practice, we weigh out a value in GRAMS and not moles. So we need to be able to convert from…
A: To solve this problem we have to calculate the mole of Gold .
Q: STARTING AMOUNT X 4 0.98692 ADD FACTOR *( ) 1.05 797 atm 101.325 Pa Convert 797 mm Hg to atm 14.70…
A:
Q: lacmillan Learning Carbohydrates serve as a vital source of chemical energy to the human body.…
A: 1 KJ = 0.239 Kcal So 119 KJ = 119 X 0.239 Kcal = 28.44 Kcal
Q: Suppose that Daniel has a 3.00 L bottle that contains a mixture of O₂, N₂, and CO₂ under a total…
A: Given: The total pressure of the gas mixture = 4.90 atm The volume of the bottle = 3.00 L The number…
Q: Give the IUPAC name of the following compound: CH3-CH-C=C-CH3 CH₂ CH3 (Hint: Notice that the longest…
A:
Q: 1) Write down the conjugate acid/base pairs for the following reactions a) HCIO4+H₂O CIO4 + H3O+ b)…
A:
Q: Complete the table below by deciding whether a precipitate forms when aqueous solutions A and B are…
A: Whether or not a reaction forms a precipitate is dictated by the solubility rules. These rules…
Q: Question) Draw the by the Chemdraw following and write the Molar Mass? 2,4,5-Trimethylaniline…
A: The organic compounds given are 2,4,5-Trimethylaniline 4-Iodoaniline 1,2-Bis(2-aminophenyl)ethane…
Q: What is the energy of a photon of frequency 4.8 × 1014 Hz
A: Energy can be calculated from frequency using the relation : E = hv h = Plank's constant v =…
Q: What is the organic product formed in the following reaction ? :N(CH3)2 I Select one: O A. II OB. I…
A: We have to give the major organic product of the given reaction. Introduction: Substitution…
Q: The simulation (as well as the full-lecture notes) indicated the difference between atomic mass and…
A: We need to use the periodic table to determine the answers. Here atomic mass would be in amu and…
Q: Q5. Draw a slip system in the figure provided below, and determine the appropriate crystal structure…
A: We have find out the crystal structure.
Q: still PDF UNOFFIC TRANSCR What is the IUPAC name of the following compound? CH3-CH=CH-CH2-CH2-CH2…
A:
Q: Give the IUPAC name for the following. OH O 3-methyl-2-pentanol methylpentanol O 3-methyl-4-pentanol…
A: IUPAC Nomenclature rule: 1) Identify the parent change 2) Identifying the substituent and functional…
Q: Calculate the standard enthalpy of formation of solid magnesium sulfide (MgS) using the following…
A: Recall the given thermochemical equations, 2 Mg s + O2 g → 2…
Q: What is the concentration of manganese ions in a saturated solution of manganese sulfide? M…
A:
Step by step
Solved in 2 steps with 1 images

- Linearly polarized light can be assigned a specific irreducible representation of a symmetry point group. If the electronic ground state of benzene has A1g symmetry and y- polarized light has the label E1u, what are the symmetry labels of allowed excited electronic states? Use the D6h character table in Appendix 3.In your own words, explain why an object that has more symmetry elements is said to have higher symmetry than an object with fewer symmetry elements.Linearly polarized light can be assigned a specific irreducible representation of a symmetry point group. If the electronic ground state of methane has A1g symmetry and x- polarized light has the label T2, what are the symmetry labels of allowed excited electronic states? Use the Td character table in Appendix 3.
- a Unlike methane, bromochlorofluoromethane (CHBrClF) is chiral. Determine all symmetry elements that are present in CHBrClF and identify its point group. b If the fluorine in this molecule were substituted with a hydrogen atom, what is the point group for the new molecule? Is it chiral?a In the Td point group, an S41 improper rotation is equivalent to what other improper rotation? b In the D6h point group, the symmetry operation labeled C21 is equivalent to what other symmetry operation?Determine the point groups of the following molecules. a Fe(CO)5, which has a trigonal bipyramidal structure, b CO32, which has three resonance structures that contribute to its overall shape, c The perfectly staggered conformation of ethane, d The perfectly eclipsed conformer of ethane.
- Assume that x- polarized light can be assigned an E symmetry species in a system that has C4v symmetry. Can a transition from E state to a B2 state occur? Why or why not?Determine which single symmetry operation of the following point groups is equivalent to the given combination of multiple symmetry operations. a In C2v, C2v=? b In C2h, iC2=? c In D6h, C6h=? d In D2d, C2C2=? e In Oh, iS4=?Identify the symmetry elements present in the following objects. a A ream of blank paper, no holes. b A ream of blank three-holed paper. c A round pencil, unsharpened, with cylindrical eraser. d A round pencil, sharpened, with cylindrical eraser.
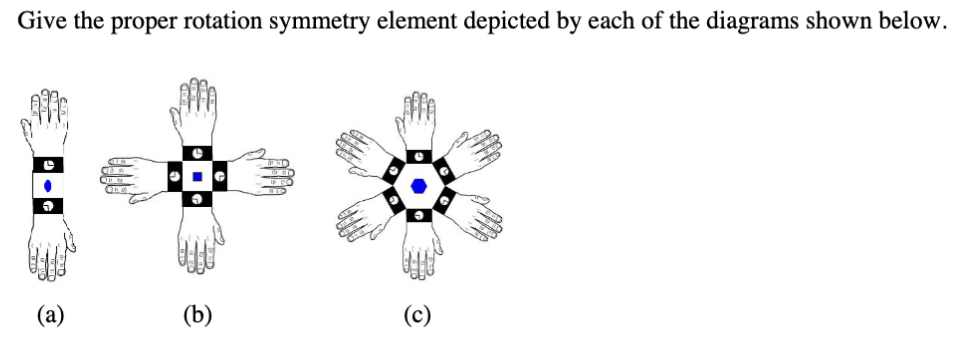
- How to determine symmetry elements, σh and σν and how to differentiate between both. Also , how is σh related to E.How many symmetry elements/ operations can decide symmetry of a molecule? Explain with appropriate examples.Using appropriate diagram(s), show which of the symmetry operation(s) does the [SO4]2- ion have: (i) C4 (ii) σh (iii) S4.

