Q: Select the reagents you would use to synthesize the compound below from benzene. (More than one step…
A:
Q: The H- ion cannot be solved exactly because it includes more than two distinct particles. True False
A: Answer: Usually Hydrogen is a cation. But some reaction Hydrogen is act as an anion. Hydrogen anion…
Q: Transmittance (%) 4000 3000 1500 Wavenumber (cm-¹) 2000 1000 500
A: Given IR spectrum of unknown compound
Q: Consider the following reaction at equilibrium: 2H₂S (g) 2H₂(g) + S₂ (g) What would the direction of…
A: For the first question, we have to find the direction of the reaction when the volume of the…
Q: Give an acceptable name for the major organic product formed by the following reaction:…
A: The question is based on the concept of organic reactions. we need to identify the product formed in…
Q: What is [H+] in a 0.270 M solution of acrylic acid, CH₂CHCOOH (Ka = 3.16 × 10-5)?
A:
Q: 4.35 L He/1 x 1 mole He/22.4 L He=0.19 moles He. Explain why the final answer of 0.19 moles makes…
A:
Q: Why do you assume X is small?
A: Answer: This question is based on the approximations used in performing chemical calculations.
Q: Macmillan Learning You heat a 5.2 gram lead ball (specific heat capacity=0.128 Joules/g-deg) to 183.…
A: Given data is The mass of lead ball heated to 183oC = 5.2 g Specific heat capacity of lead =…
Q: H-X HF HCI HBr HI ΔΗ (kJ mol-¹) -13 -59 -63 -57
A:
Q: 14.27 Label each of the following structures as a cyclic hemiacetal, hemiketal, acetal, ketal, or…
A: Since, Hemiacetal is that structure in which one carbon is attached with two oxygen out of which…
Q: Select all descriptors that are correct for the 3-methylcyclopentene molecule. Group of answer…
A: Answer:- This question is answered by using the simple concept of writing the structure of organic…
Q: 1. Which of the following types of filter are made to operate continuously? a. rotary drum b.…
A: In the given questions, we have to find the correct option for the given question.
Q: Draw the structure of the major organic product(s) of the reaction. 1. LIAIH4, ether 2. H₂O You do…
A: The question is based on the concept of organic reactions. reaction of amide with lithium aluminium…
Q: When the following equation is balanced properly under acidic conditions, what are the coefficients…
A: Since, In redox reaction, Oxidation and reduction reaction both takes place simultaneously.
Q: Hess Law Act. 1 Find the AH for the reaction below: A) HCl (9) + NaNO₂ (58) → HNO₂(g) + Nack(s)…
A:
Q: 20gCuO to particles =
A: We know- 1mole=6.022x1023 particle Number of moles can be calculated as-⇒Mass of compound(g)Molar…
Q: OH KMnO4
A:
Q: Determine the POLARITY of molecules and TYPE/S of INTERMOLECULAR FORCES that exist in each of the…
A: According to Q&A guidelines of Bartleby, we are supposed to answer only the first 3 sub-parts of…
Q: actually added to 1 tablet (L) *Concentration (M) of HCI Report to 4 sig figs Expand M out to a…
A:
Q: Balance the following reaction in basic solution. Te (s) + NO3(aq) → TeO32- (aq) + N₂O4 (g)
A: To: Balance the given redox equation in basic medium, Te(s) + NO3- (aq) → TeO32- (aq) + N2O4 (g)
Q: 13-15. What mass of oxygen gas, O2, from the air is consumed in the combustion of 500 g of octane,…
A: In this question, we will be the mass of oxygen gas in gram unit from the given mass of octane. Mass…
Q: Discuss the reasons for the differences in glass transition temperature for the following pairs of…
A: Discussion of the reasons for the difference in glass transition temperature of the polyethylene and…
Q: Consider the following reaction at equilibrium: 2CO (g) + O₂ (g)2CO₂ (g) What would be the direction…
A: Note: Since you have asked multiple questions, we will solve the first question for you. If you want…
Q: Aluminum reacts with42 g of iron(II) sulfate.How many grams of iron are formed? Aluminum sulfate is…
A: A balanced chemical equation contain equal number of atoms of each elements on both sides of the…
Q: Choose the two alkyl bromides and the hydrogenation method that must be used to synthesize these…
A: This question is answered by using the simple concept of chemical reactions of organic compounds…
Q: When toluene is sulfonated (concentrated H₂SO4) at room temperature, predominantly (about 95% of the…
A: Generally, kinetic product are favoured at low temperature or at room temperature and thermodynamic…
Q: Using the formula M1V1 = M2V2, how many millimeters of 6.0M sodium hydroxide are required to…
A:
Q: The molecular weight of FeCl2 is 151.91 g/mol. In this assignment, you will determine the mass %…
A: In the given question we have to calculate the % mass of FeCl2 in unknown mixture by titrating with…
Q: 2. Examine the two scenarios below. • Scenario A: 100 g of glucose in the presence of unlimited…
A: two condition are give , we have to tell in which condition more product is formed
Q: Classify the following substituents according to whether they are electron donors or electron…
A: Inductive effect: Attracting the electron density towards themselves from the adjacent atoms is…
Q: in the reaction, PrBr with NH3 gives a mixture of products. Why is an excess of NH3 required to give…
A: In the given question, a reaction is given and we have to find the reason that why NH3 is required…
Q: cl CHÍCH 32 THF, A Elimination
A: This is a simple question about the reaction mechanism of organic reaction specially reaction of…
Q: Which covalent bonds would need to be broken for these reactions to occur? A B H-X (covalent bond…
A: Answer: Dissolution of any compound takes place when first of all its gets dissociated into its ions…
Q: 7-9. Fermentation is a complex chemical process of making wine by converting glucose into ethanol…
A: Given: Mass of glucose = 53.0 grams Molar mass of glucose is: Molar mass of glucose=6×Molar mass of…
Q: quar (iv) Why is the universe rega A4. Define Gibb's free energy. Show that the Gibb's free energy…
A: Gibb's free energy (G) is a thermodynamic function that measures the maximum amount of work that can…
Q: Which of the following is true? A. Group of answer choices B. The reaction of an ester with alkoxide…
A: A. False. The reaction of an ester with alkoxide base is reversible. The alkoxide base can…
Q: What if we wanted to calculate the initial concentration of acetylsalicylic acid
A: 1.00 g of acetylsalicylic acid is dissolved in 120 mL of water.We have to calculate the initial…
Q: What is the resulting volume of a 0.452 M solution ammonium nitrate NH4NO3 if it was prepared by…
A:
Q: Click the "draw structure" button to activate the drawing utility. Supply the missing organic and/or…
A: Amides can be produced from the ester by treating with amines. It is nucleophilic…
Q: Draw detailed arrow-pushing mechanisms for the following transformations: of MgBr Hint this one…
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: 4) Bioluminecence is the emission of light by a living organism. Fireflies (lightning bugs) em light…
A:
Q: HF + H₂O → HCI + H₂O → HBr + H₂O → HI + H₂O →
A: The interactions that are consistent across all four reactions are hydrogen bonding between the…
Q: I need the name of the product
A: Answer: In this question we have to write down the IUPAC name of the organic compound forming as…
Q: 5. What is the final ONPG concentration (in mM) of each of the Reaction tubes? ***Hint: Use the…
A: O-nitrophenyl-beta-D-galactopyranoside (ONPG) test id done to study enzyme kinetics of β…
Q: How many moles of hydrogen atom are present in 25.6 g urea [(NH2)2CO], which is used as a…
A:
Q: might dissolve at different times. Consider: • which day the least sugar dissolve in which day the…
A: When solute dissolves in a solution that process is called dissolution. Rate of dissolution depends…
Q: Which of the following reagents might serve as the basis for a simple chemical test that would…
A: Alkene reacts with Br2 in carbon tetrachloride and decolourises the bromine water but this reaction…
Q: A7. (a) Given that for an ideal gas rin(1) AG=nRT In aG aP =V, show that for a change in pressure…
A: Given is the relation of change in Gibb's free energy with change in pressure at constant…
Q: HO H-C-C-N-H H H Following is the structural formula of acetamide, a molecule that contains a…
A:
Step by step
Solved in 4 steps with 3 images

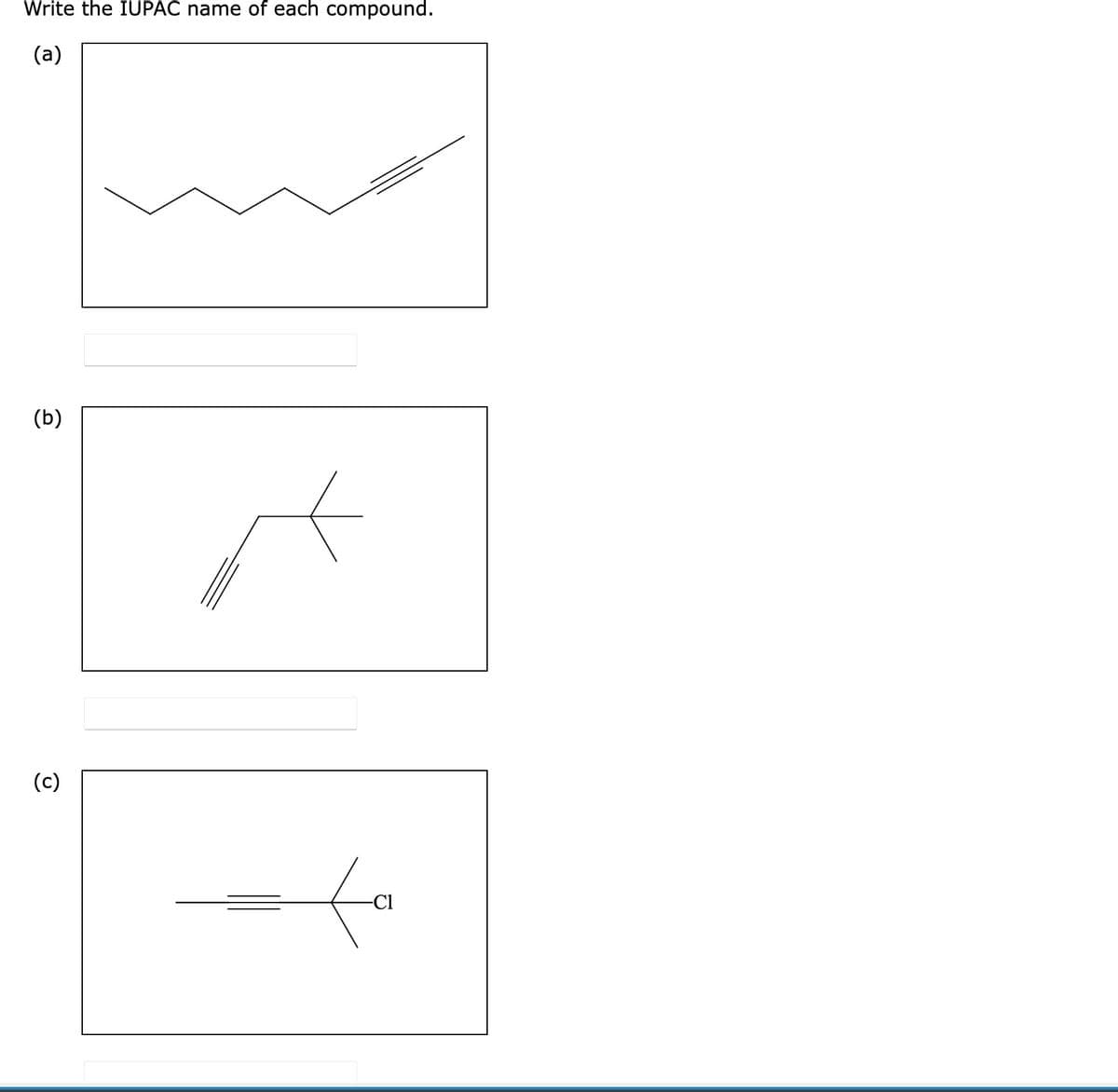
- Write the IUPAC name of each compound, showing stereochemistry where relevant.Two diastereomeric sets of enantiomers, A/B and C/D, exist for 3-bromo-2-butanol. When enantiomer A or B is treated with HBr, only racemic 2,3-dibromobutane is formed; no meso isomer is formed. When enantiomer C or D is treated with HBr, only meso 2,3-dibromobutane is formed; no racemic 2,3-dibromobutane is formed. Account for these observations.Give the IUPAC or common name for each compound. (A - E)



