Q: 5. For the balanced &quation shown below, what would be the limiting reagent if 18.9 grams of HNO3 w...
A:
Q: What are the differences of forward reaction and reverse reaction
A: Below attached file showing the details answer. All the best .
Q: Select all that apply. Select the equilibrium constant expressions (K, and Kp) for the formation of ...
A:
Q: Name Melting point (°C) Unknown 121-122 Mandelic acid 120 Benzoic acid 123 Unknown + Benzoic acid Un...
A: Melting point of a substance is used to determine the purity of a substance. Melting points of mande...
Q: Temperature of hydrobromic acid (*C) 24.0 Volume of potassium hydroxide (mL) 100 mperature of potass...
A: As per battle by guidelines, as an expert I am allowed to solve first three sub parts. Please re upl...
Q: Aqueous hydrochloric acid (HCI) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium ...
A:
Q: Determine if the following salt is neutral, acidic or basic. If acidic or basic, write the appropria...
A: Nature of salt can be identified from the nature of acid and base used to form the salt as:- Stron...
Q: What will be the expected pH of the hydrolyzed aspirin solution after the excess NaOH has been neutr...
A: Aspirin other name is acetylsalicylic acid. Hydrolysis of aspirin means breaking of aspirin by addit...
Q: 1. A solution is prepared by dissolving 12 grams of turmeric powder in 350 grams of water. Calculate...
A: 1) Given, Mass of turmeric powder = 12 g Mass of water = 350 g Mass percent of each component of the...
Q: The pka volue of cacium fluoride is 10.41. Calculate the solubTty in g/ at 25 C in a pure water b. 0...
A: Given: pKspCaF2 = 10.41 To determine solubility in g/L at 25oC in a) pure water b) 0.0200 M Calcium ...
Q: Hce
A: Here we have to write the mechanism of hydrolysis of amide in basic medium as well as in the acidic ...
Q: Enter your answer in the provided box. Carbonyl chloride (COCI), also called phosgene, was used in W...
A:
Q: Q: Indicate the effect caused by the following situations on the value of the weight molecule of a s...
A: Weight of solute is only affected in condition when there is Loss of solute in Process.
Q: 3. Identify if the following is a pyrimidine/purine nucleotide or a pyrimidine/purine nucleoside and...
A: Pyrimidine :- pyrimidine ring is a six membered heterocyclic aromatic organic molecules having 1,3 p...
Q: rate
A:
Q: Identify the compounds that are more soluble in an acidic solution than in a neutral solution. HgI, ...
A: Strong-base-weak-acid salts, such as MgF2 .(soluble in acidic solution) That is because when these s...
Q: Calculate HgCl,2-] in a solution prepared by adding 0.0200 mol of NaCl to 0.250 L of a 0.100– M HgCl...
A: The formation constant, Kf of HgCl42- is 1.1×1016. It can be represented as: Hg2+aq+4Cl-aq→HgCl42-aq...
Q: The value of AH° for the reaction below is -126 kJ. The amount of heat that is released by the react...
A: Given reaction is : 2Na2O2 (s) + 2H2O (l) → 4NaOH (s) + O2 (g) Standard change in enthalpy of the re...
Q: Is point source pollution or nonpoint source pollution easier to address, and why? Nonpoint source p...
A: Point source pollution is the pollution in which location is identified whereas nonpoint source poll...
Q: STATEMENT OF THE PROBLEM Problem 2: Consider a 250.0 mL solution that contains 0.215 mole KNO3. What...
A: Answer: In both the questions we have to find out the molar concentration of the solution where in s...
Q: MULTIPLE CHOICE Question 9 A dam releases water during the summer months when farmers are in need of...
A: B
Q: 12) Explain why compound 2 is a much weaker bane than compound 1 (approximately 1000 tirmes less bas...
A:
Q: А. Alkenes CH3 CH3 CH3 1. CH3-CH=CH-ĊH-CH2CH2-CH3 EC-CH-CH3 ČH3 3. СH-C: CH3 ÇH3 2. H-с— с—сн-ҫн-сH-...
A: Given alkenes are =>
Q: A 1.00 g sample of NaOH which has been exposed to the air for some time is dissolve in water and w...
A:
Q: Arrange the following compounds in the following order. Explain your arrangement. Br,, CO, H,, CH. L...
A: Br2 , CO2, H2 , CH4
Q: ata Endgroup analysis by titration: 8 mols of 1,6-hexanediamine were reacted rith 9.8 mols of adipic...
A:
Q: Which of the following is not a polyprotic acid or base? HCl H3PO4 Ca(OH)2 H2SO3...
A: Given, HCl H3PO4 Ca(OH)2 H2SO3
Q: In an experiment to determine the amount of aspirin in a tablet, one of the bottles of NaOH used was...
A: Here we required to predict the effect of concentration NaOH in determination of the concentration o...
Q: 1. Suppose there is an atom of oxygen, containing eight protons and eight neutrons in the nucleus, a...
A: 1. An oxygen atom has eight protons and eight neutrons in the nucleus. Another four neutrons are add...
Q: 2. Calculate the partial pressures of the components of the reaction I2(s) + Br2(9) ++ 21Br(9) given...
A:
Q: Nuclear Binding Energy II Calculate the binding energy per nucleon for the nucleus. (Atomic masses: ...
A: B.E per nucleon calculated as total B.E /Mass no.
Q: An analytical chemist is titrating 105.0 mL of a 0.5500M solution of ethylamine (C,H,NH,) with a 0.1...
A:
Q: Give a possible equation you could use to prepare 2-methyl-2-phenyl-pentanal using a pinnakol rear...
A: Below attached file showing the details answer. All the best
Q: ed in a variable pathlength cell. The absorbance of the reference and the sa cm. What is the concent...
A:
Q: Indicate whether the following salts will be soluble or insoluble in water: HgCi, BacO3, SrBr2, and ...
A: Given salts : HgCi, BacO3, SrBr2, and ZnSO4.
Q: 8. While studying the adsorption isotherm of acetic acid on charcoal, if 2.5 gm of charcoal be taken...
A: We use the Freundlich equation to plot Freundlich adsorption isotherm curve. Here adsorbate is aceti...
Q: What is the molarity of a solution that was prepared by dissolving 14.2 g of NaNO3 in enough water t...
A: Molarity is defined as the number of moles of solute dissolved per litre of solution.
Q: Experiment 2: Investigating the Properties of Buffer Solutions Test Data: pH of Cola Solution 2...
A: Buffer solution : It is the aqueous solution of of mixture of weak acid and conjugate base or vice v...
Q: Q: Indicate the effect caused by the following situations on the value of the weight molecule of a s...
A: The solute only effected when there is Loss of solute in the Experiment
Q: HNO2 (Ka = 1.4 x10-4) to which you add 10.0 mL of 0.10M NaOH What is the % pH change?
A: Given, Volume = 0.50L Molarity of salt = 0.10M NaNO2 Molality of Acid = 0.10M HNO2 (Ka = 1.4 x10-4) ...
Q: Predict the direction of the reaction; consider the equation: CO(g) + H2O(g) (double arrow) CO2(g) +...
A:
Q: INSTRUCTIONS: Explain how the following images relate to chemical equilibrium.
A:
Q: Calculate the pH of 0.730 M hydrazine
A:
Q: Problem 1: The following equations describe how our circulatory and respiratory systems remove carbo...
A: The equilibrium reaction taking place is given as, => Given: Concentration of HCO3- = 0.028 M An...
Q: (c) What is the solvent effect on the SN1 and SN2 reactions?', (d) Which reactant in each of the fol...
A: In SN1 carbocation intermediate is formed.. And in SN2 transition state is formed..
Q: 5. Explain why metals exhibit the following properties. – page 269 shiny flexible Electrical conduct...
A: Metals are shiny-The positive metal ions are surrounded by pool of electrons. The electrostatic forc...
Q: 2 NOBR (g) = 2 NO (g) + Br2 (g) A) adding NOBr. B) removing NO C) doubling the concentration of NO a...
A: Given- 2NOBr(g) <->2NO + Br2(g)
Q: QUESTION: Explain IN DETAIL at least one instance in which chemical equilibrium is utilized in our d...
A: The chemical equilibrium is the state of a reaction in which the forward reaction rate is equal to t...
Q: Step by step naming of ionic compound - calcium iodide
A: Rules for naming simple ionic compounds. 1. Name the metal by its elemental name. 2. Name the nonmet...
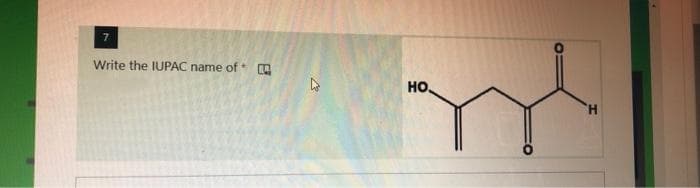
Step by step
Solved in 2 steps with 1 images


