Q: Show how the Gabriel malonic ester synthesis could be used to prepare: (a) valine (b) phenylalanine…
A: the answer is b) Phenylalanine
Q: Define the following terms:a. Maillard reactionb. Schiff basec. Amadori productd. adducte. reactive…
A: In Organic chemistry, There are numerous reactions that are named based upon the scientist who…
Q: OMe NME2
A: In this question, we want to convert starting material to final product by using Reagents. You can…
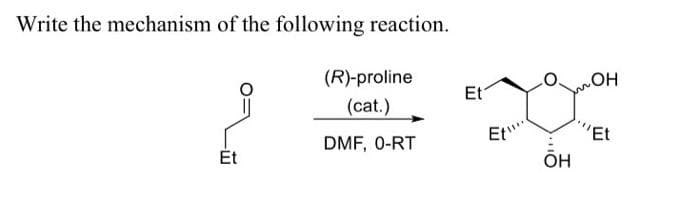
Q: The rate constant for the uncatalyzed reaction of two molecules of glycine ethyl ester to form…
A:
Q: Complete Reaction NBS (N B1omo Succinamide) DCM (CHCI,) dchlore methone
A: Introduction: Addition reaction is a reaction in which substituent get attached to double bond to…
Q: Dehydratase is a PLP-requiring enzyme that catalyzes an a,ß-elimination reaction. Propose a…
A:
Q: Mechanism for the base catalyzed hydrolysis of nitriles ? Explain in detail?
A: The basic hydrolysis of nitriles gives carboxylic acid. Step 1: The hydroxide ion attacks at the…
Q: MCPBA МеMgBr (excess), н- Swern a) b) c) оxidation
A:
Q: Nole, formatión of the enolate is carried out under irreversible conditions. 1. LDA, THF, -78 °C…
A:
Q: Propose a mechanism for the reaction of methyl isocyanate with 1-naphthol to giveSevin® insecticide.
A: Sevin® insecticide is a brand name whose chemical name is 1-naphthyl methylcarbamate or Carbaryl . A…
Q: Draw the mechanism for the reduction of Camphor to Isoborneol and Borneo CH3 CH3 (ехо) exo attack…
A: Endo mechanism is most favourable because of stearic hindrance In exo mechanism H- will bear a…
Q: NaOEt DMSO BoeO. DMAP THE HO POBr POB NOE EOH 70°C NaOE DMSO 70°C Method B Triflacroacetic acid DCM…
A: While performing any organic synthesis, it is very important to look into the compatibility of the…
Q: Co2+ catalyzes the hydrolysis of the lactam shown here. Propose a mechanism for the metal-ion…
A: Given reaction:
Q: (-)-Ipc2BOTf i-Pr2NEt ЗА CH2CI2, -20 oC TBSOTF 2,6-lutidine 4B 50 CH2CI2, -78 °C structure of 3A?…
A: The geometry of enolate depends on the chiral borate reagents. (-)Ipc2BOTf majorly produces…
Q: Draw the mechanism with curly arrows for the following hydrolysis reaction including the structure…
A: Detail mechanistic pathway is given below to find out the missing product
Q: When some sugars dissolve in water they spontaneously undergo changes in optical rotation called…
A:
Q: derivative is studied. a. Indicate the reaction intermediate and product. b. Perform the reaction…
A: Given : We have to make products for the following reaction.
Q: Write down the reaction mechanism for obtaining iodoform from ethyl alcohol u Na2CO3 base.
A: It is strong oxidising agent it will alcoho convert alcohol into aldehyde
Q: The reaction of 1-phenyl ethyl chloride compound with NaOCH3 in DMF find the product by writing the…
A: See major product in pink box. Name of product is Phenyl Ethyl Methyl Ether
Q: What is the effect of a protic solvent in an SN1 reaction?
A: Protic solvent can for hydrogen bond with nucleofuge anion to stabilize it during solvation. eg.…
Q: Provide hrecereet Prud uct Br koH noreathon
A: There are number of functional group associated with organic compounds which impart specific…
Q: Show a detailed mechanism for the coupling of N-acetyl-L-proline to L-ValOBn using (a) DCC/HOBT
A: Amino acid consists an amino group and a carboxylic acid group and both the functional groups are…
Q: React the substrate with excess CH3MgBr in ether, followed by subsequent water work-up. O O
A:
Q: Draw a reaction mechanism to account for the formation of the dipeptide (cy = cyclohexyl)
A:
Q: Find and name the reaction mechanism of the product formed as a result of the reaction of (S)-1-…
A:
Q: Proindetie Product CN CN NacN DMSO horcachion
A: Nucleophilic substitution occurs when the leaving group is good.
Q: Propose a reaction mechanism for isocyanide formation.
A:
Q: 2. Give a mechanism for the acid-catalyzed hydrolysis of lactose.
A: Lactose is made by condensation of two sugar molecules that are glucose and galactose. The…
Q: Complete the following multistep synthesis, depicting the possible stereochemical control in the…
A:
Q: Show a reasonable mechanism for the enzymatic transimination reaction 6. : between the PLP-lysine…
A: The transimination, just like transamination, is a biochemical reaction in which an imino group is…
Q: Propose a mechanism for the formation of Melmac.
A: Melmac is a polymer which is formed by the polymerisation of monomers melamine and CH2O
Q: What kind of a mechanism is it in the last step of lumefantrin-synthesis?
A: Lumefantrine: lumefantrine inhibits the formation of β-hematin by forming a complex with hemin and…
Q: b. Suggest mechanism for synthesis of the following cyclic ester. OH CO, Bu,N Pd(OAc),
A: Ans
Q: MCPBA aqueous HCI A CH2Cl2 Me Me Me Ме
A: mCPBA is used for epoxidation of alkene. After epoxidation tautomerization takes place.
Q: (b) Provide a mechanism for the coupling of Boc-Gly-OH with H-Phe- O'Bu using EDCI, DMAP and Hünig's…
A: Carboxylic acid react with EDCl DMAP react with product of carboxylic acid of EDCl Nucleophilic of…
Q: Synthesis route with the highest yield percentage of solefacin succinate and step by step chemical…
A: Given Synthesis route with the highest yield percentage of solefacin succinate and step by step…
Q: Draw a detailed, arrow-pushing mechanism for the conversion of benzoyl chloride to benzamide using…
A: The structure of benzoyl chloride is- It has an acid chloride functional group. The structure of…
Q: rottamatenCNT quon ianobion HO smeotol oealt on mokest s yligns t Propose a way to carry out the…
A: Introduction: To convert the alcohol group into nitrile group, first form the alkyl halide from…
Q: Illustrate mechanisam for the hydrolysis of an acide chloride ?
A: Hydrolysis of an acid chloride mechanism has to be given.
Q: Explain the mechanism for an acid catalyzed hydrolysis of nitrile ?
A:
Q: 3. The following sequence comes from a synthesis of strychnine. Fill in the reagents to complete the…
A:
Q: Write a mutlistep synthesis for the reaction below CH3 H.
A:
Q: Propose a mechanism for the following reaction Phố PhCI MegSi- -OMe -OMe
A: The reaction proceeds via a nucleophilic attack of the electron rich benzene ring on the carbonyl…
Q: Show experimental conditions by which to carry out the following synthesis. MeOOC Ph CHO + OMe Ph…
A: The experimental conditions by which the given synthesis occurs has to be given.
Q: Define the following terms: a. reaction mechanism b. carbocation c. enediol d. general acid e.…
A: Since we only answer up to 3 sub-parts, we’ll answer the first3. Please resubmit the question and…
Step by step
Solved in 2 steps with 1 images

- Illustrate mechanisam for the hydrolysis of an acide chloride ?Mechanism for the base catalyzed hydrolysis of nitriles ? Explain in detail?Draw the mechanism for the formation of 6-iodovanillin product (Carbon 1 is the aldehyde carbon, 3-methoxy, etc..). Include the reaction of Oxone with KI to afford the active electrophile and resonance structures for the EAS reaction.
- What is the mechanism for this reaction? We get the hint that after the Grignard-reaction there is a intramolekular reaction with the ester (CO2Et-group).All steps of the reaction for obtaining fumaric acid starting from maleic anhydride areshow the mechanisms in detailAccount for the rapid rate of methanolysis of ClCH2OCH2CH3 even though the substrate is a primary halide. Hint: The reaction of the substrate with ethanol proceeds by SN1 mechanism. Draw the Lewisstructures.
- Please write out the mechanism for the acid (WHICH IS 3-BROMOBENZOIC ACID) reacting with anilide. PROVIDE THE MECHANISM AND BRIEFLY EXPLAIN THE STEPS OCCURING PLEASEwrite the mechanism of the reaction when (R)-2-bromo-2-methylpropane is reacted with NaI.For the base-catalysed hydrolysis of 3-bromo-3-methylhexane (i.e. reaction with the nucleophile OH-): State whether the reaction is likely to proceed by an SN1 or SN2 mechanism, and explain why give the likely rate law and explain why
- Give the mechanism for the reaction belowThe reaction of 1-bromoheptane with the propionate anion to give heptyl propionate ester is greatly accelerated by adding catalytic amounts of KI. Explain.The rate constant for the uncatalyzed reaction of two molecules of glycine ethyl ester to form glycylglycine ethyl ester is 0.6 M- 1s - 1. In the presence of Co2 +, the rate constant is 1.5 * 106 M- 1s - 1. What rate enhancement does the catalyst provide?

