You have a 16 mL sample of acetylcholine (a neurotransmitter) with an unknown concentration and a pH of 7.66. You incubate his sample with the enzyme acetylcholinesterase to convert all of the acetylcholine to choline and acetic acid. The acetic acid issociates to yield acetate and hydrogen ions. At the end of the incubation period, you measure the pH again and find that it has ecreased to 5.52. Assuming there was no buffer in the assay mixture, determine the number of nanomoles of acetylcholine in he original 16 mL sample. CH,—C−O−CH,—CH,—*N–CH, Acetylcholine CH₂ acetycholine in original sample: CH₂ H₂O CH₂ I HO–CH,—CH,−*N−CH, + CH,-C−o + H Choline I CH3 Acetate nmol
You have a 16 mL sample of acetylcholine (a neurotransmitter) with an unknown concentration and a pH of 7.66. You incubate his sample with the enzyme acetylcholinesterase to convert all of the acetylcholine to choline and acetic acid. The acetic acid issociates to yield acetate and hydrogen ions. At the end of the incubation period, you measure the pH again and find that it has ecreased to 5.52. Assuming there was no buffer in the assay mixture, determine the number of nanomoles of acetylcholine in he original 16 mL sample. CH,—C−O−CH,—CH,—*N–CH, Acetylcholine CH₂ acetycholine in original sample: CH₂ H₂O CH₂ I HO–CH,—CH,−*N−CH, + CH,-C−o + H Choline I CH3 Acetate nmol
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter14: Acid- Base Equilibria
Section: Chapter Questions
Problem 118CP: Malonic acid (HO2CCH2CO2H) is a diprotic acid. In the titration of malonic acid w ith NaOH,...
Related questions
Question
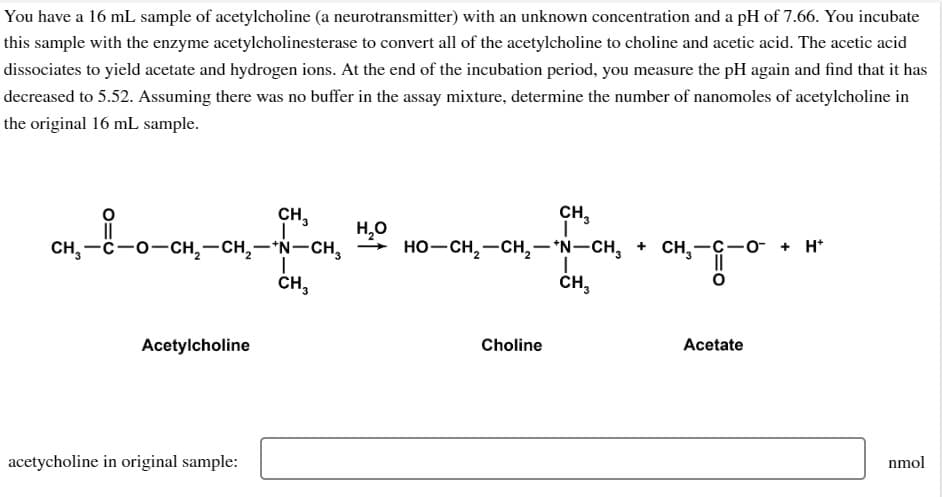
Transcribed Image Text:You have a 16 mL sample of acetylcholine (a neurotransmitter) with an unknown concentration and a pH of 7.66. You incubate
this sample with the enzyme acetylcholinesterase to convert all of the acetylcholine to choline and acetic acid. The acetic acid
dissociates to yield acetate and hydrogen ions. At the end of the incubation period, you measure the pH again and find that it has
decreased to 5.52. Assuming there was no buffer in the assay mixture, determine the number of nanomoles of acetylcholine in
the original 16 mL sample.
Acetylcholine
CH₂
acetycholine in original sample:
CH,—C−O−CH,—CH,AN-CH, HO–CH,—CH,*N–CH, + CHy
H₂O
I
CH3
CH3
Choline
CH₂
CH,-GO + H
Acetate
nmol
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning