Latent heat and phase change
A physical process in which a conversion among the basic states or phases of matter, i.e., solid, liquid, and gas takes place under the effect of a certain temperature and pressure is referred to as a phase change. Generally, the phase change of a substance occurs when heat transfer takes place between the substance and its surroundings. Based on the direction in which heat transfer takes place, different types of phase changes can occur.
Triple Point of Water
The branch of physics in which observer deals with temperature related properties is called thermodynamics.
Boiling Point of Water
Everyday examples of boiling is, boiling milk, heating water. One would have observed that when we heat water it goes through various stages and at one point bubbles show in water, and water keeps splashing with bubbles bursting, we in layman terms say that water is boiling.
Freezing Point of Water
In general, the freezing point of water is 0° Celsius, or 32° Fahrenheit. This is the temperature at which water will ordinarily change from its liquid state to its solid state (ice). However, there are certain conditions that can affect the freezing point of water. For example, a liquid may be supercooled or contain impurities so that it does not freeze at the ordinary freezing point.
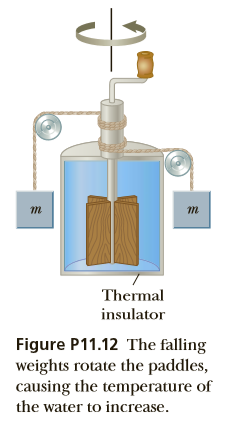
The apparatus shown in Figure P11.12 was used by Joule to measure the mechanical equivalent of heat. Work is done on the water by a rotating paddle wheel, which is driven by two blocks falling at a constant speed. The temperature of the stirred water increases due to the friction between the water and the paddles. If the energy lost in the bearings and through the walls is neglected, then the loss in potential energy associated with the blocks equals the work done by the paddle wheel on the water. If each block has a mass of 1.50 kg and the insulated tank is filled with 0.200 kg of water, what is the increase in temperature of the water after the blocks fall through a distance of 3.00 m?
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 3 images








