Chemistry for Engineering Students
3rd Edition
ISBN:9781285199023
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter5: Gases
Section: Chapter Questions
Problem 5.23PAE
Related questions
Question
Propose a structural formula for compound A, C5H13N, given its IR and calculated 1H-NMR spectra.
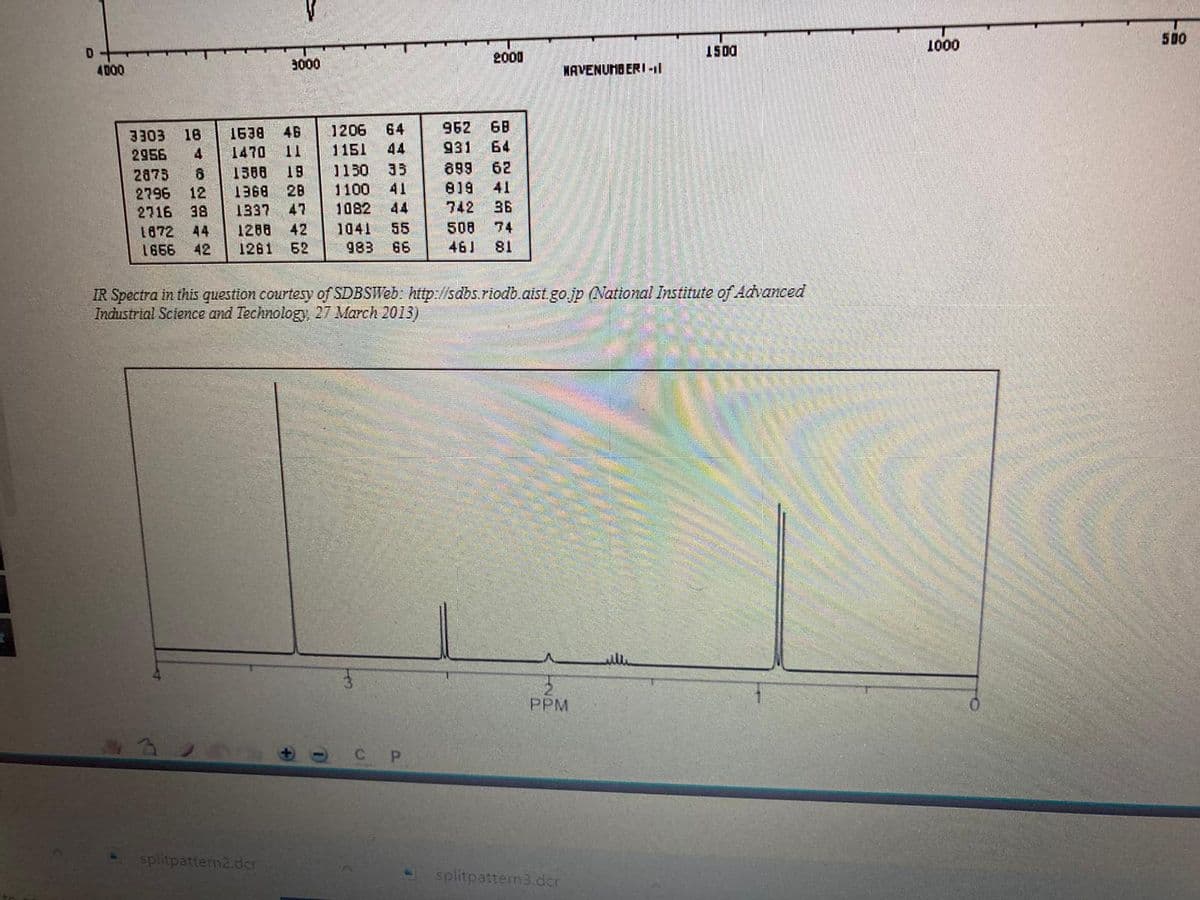
Transcribed Image Text:500
1000
D
2000
4DO0
3000
MAVENUMB ERI-il
18
1638
46
1206
64
962
68
3303
2956
931
64
1151
1130
1470
11
44
62
899
41
19
35
2875
12
1588
1368
1337
28
1100
41
819
2796
2716
38
47
1082
44
742
36
55
74
508
46J
1041
1288
1261
1872 44
42
1666
42
62
983 66
81
IR Spectra in this question courtesy of SDBSWEB: http://sdbs.riodb.aist. go.jp (National Institute of Advanced
Industrial Science and Technology, 27 March 2013)
wlllh
PPM
splitpattern2.dcr
splitpatterm3.der
![VtakeAssignment/takeCovalentActivity.do?locator=Dassignment-take
[References]
D.
4000
3000
2000
1500
1000
500
HAVENUMBERI-1
1206
1151
962
68
64
16
1638
46
64
2956
4.
1470
11
44
931
2873
1388
19
1130
33
899
62
2796
12
1968
2B
1100
41
819
41
2716
38
1337
47
1082
44
742
36
1872
L656
44
1288
42
1041
55
508
74
42
1261
52
983
66
461
81
IR Spectra In this question courtesy of SDBSWeb http: sabs.riodb atst go.jp (National Institute of ahanced
Industrial Science and Technology 27 March 2013)
TRANSMITTANCEI2](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2Fc2b0fd2e-7313-40ae-aeec-a6f3ed3ace1a%2F416a2b17-83ae-49a5-b596-4aefdf7fbb3f%2Frljfbwi_processed.jpeg&w=3840&q=75)
Transcribed Image Text:VtakeAssignment/takeCovalentActivity.do?locator=Dassignment-take
[References]
D.
4000
3000
2000
1500
1000
500
HAVENUMBERI-1
1206
1151
962
68
64
16
1638
46
64
2956
4.
1470
11
44
931
2873
1388
19
1130
33
899
62
2796
12
1968
2B
1100
41
819
41
2716
38
1337
47
1082
44
742
36
1872
L656
44
1288
42
1041
55
508
74
42
1261
52
983
66
461
81
IR Spectra In this question courtesy of SDBSWeb http: sabs.riodb atst go.jp (National Institute of ahanced
Industrial Science and Technology 27 March 2013)
TRANSMITTANCEI2
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning