Q: What is the pH value of a buffer prepared by adding 10 mL of 0.1 M acetic acid to 20 mL of 0.1 M sod...
A: Given: Volume of acetic acid=10mL Molarity of acetic acid=0.1M Volume of sodium acetate=20mL Molarit...
Q: A mixture containing only KCl and NaBr is analyzed by the Mohr method. A 0.5072-g sample is dissolve...
A: %w/w=mass of solutemass of solution×100 this is the one of the method to calculate the concentrati...
Q: What is the pH value of a 1x10^-3 M CH3COOH solution? (Ka:1.75x10^-5)
A: CH3COOH ionizes as follows: CH3COOH (aq) + H2O(l) <-----> CH3COO-(aq) + H3O+(aq)
Q: What cyclic product is formed from each dihalide using the malonic ester synthesis?
A: By using malonic ester synthesis, substituted carboxylic acids can be synthesised. In this reaction,...
Q: c) Ageing is an important aspect of catalyst preparation, briefly describe the ageing process in ter...
A: The external species that are added to a reaction to increasing the rate of the reaction is known as...
Q: Suppose there are three covalent compounds with similar molar mass and relative molecular shape. Exp...
A: If the compounds have similar shape and molar mass but different boiling points, they must have diff...
Q: please help with the following!
A:
Q: Conversation Transcript Boss: Please sit down, Employee X. I understand that you have been prepa...
A: The grams of the substances could be converted to moles of the substances using their molar masses. ...
Q: Identify the hybridization of each carbon atom in the following molecule. (The arrangement of atoms ...
A: Hybridization is a hypothetical concept which involves mixing of orbitals to form new orbitals calle...
Q: Calculate the molar solubility of CdCO3 in a buffer solution containing 0.115 M Na2CO3 and 0.120 M N...
A:
Q: A reaction is of second order with respect to a reactant. How is its rate affected if the concentrat...
A: The rate law is an expression in which the rate of a reaction is the product of the molar concentrat...
Q: A student obtained a solid product in laboratory synthesis. To verify the identity of the solid, she...
A: By merely looking at any compound we cannot identify what its structure is. Melting point is an imp...
Q: Mark each chiral center in the following molecule with an asterisk. How many stereoisomers are poss...
A: A chiral center is the one where all the groups attached to the C are different. If there are n chir...
Q: The mass spectrum of a compound with a molecular formula C5H10O is shown below. Which compound would...
A: The mass spectrum is used to determine the structure of a compound and the molecular mass of the com...
Q: At 8:00 a.m., a patient receives a 1.5-mg dose of I-131 to treatthyroid cancer. If the nuclide has a...
A: introduction: all the nuclear reactions are first order reactions , in which one nuclie decay withi...
Q: Which element, W or Au, has the greater number of electrons in antibondingorbitals? Which one would ...
A: Molecular orbital theory (MOT) is a method which is used to explain the bonding in the molecules. Th...
Q: The enzyme urease catalyzes the hydrolysis of urea to ammonia plus carbon dioxide. At 21 °C the unca...
A: It is given that the energy of activation (Ea) of uncatalyzed reaction is 125 KJ/mol or 125000 J/mol...
Q: Complete the following table: Cation Formula Anion Formula Compound Formula co,²- Mg2+ NO, Ba2+ so,
A: In an ionic compound magnitude of positive and negative charges must be equal. That means charges mu...
Q: Calculate the ΔMG when mixing 2 mols H2 at 25ºC and 4 mols N2 at 25 ºC , being both of them at the s...
A: For an ideal gas mixture,ΔMG = -T ΔMSwhere ΔMS = -nR (x1 lnx1 + x2 lnx2) x1 and x2 are mole fractio...
Q: Question 4: Two students prepare two cyclohexane solutions having the same freezing point. Student 1...
A: Given, that the students use the two solutions of cyclohexane that have the same freezing point. Her...
Q: how much nacl is required to render 60ml of 1% solution of apomorphine HCl isotonic with blood serum
A: The amount of NaCl required can be calculated by using the Sodium chloride equivalent method. 60 ml ...
Q: What mass of methane must be burned to release 185 kj of heat
A: Given reaction : CH4 (g) + 2O2 (g) ------> CO2 (g) + 2H2O (g) ∆H°rxn = - 890 kJ/mol
Q: What is the reason which makes any atom highly reactive?
A: Valence shell can be defined as the outermost shell of an element. Atom of the every element have di...
Q: Predict the uv maximum for the following compound: A) 373 B) 385 C) 320 D) 331 E) 325
A: The given system is an alpha-beta unsaturated cyclohexanone compound. The UV maximum for the given c...
Q: An organic compound X with a relative molecular mass of 88.0 was found to contain 54.54% carbon, 36....
A: To determine the molecular formula, first, the empirical formula of the compound will be determined....
Q: Initiator lj häs hålf the izes 4 times as fast as monomer M2 at 80°C, when initiator I is used for b...
A: Degree of polymerization of the monomer is given by the following formula As it is mentioned in th...
Q: The photochemical formation of molecule Z uses a radiation of 300 nm. The sample is irradiated with ...
A: It is given that a photochemical formation of Z uses radiation of 300 nm and the sample is irradiate...
Q: k) The reactions of Group I and Group II with ammonia and chloride ligands give distinctly different...
A: Group I and group II elements are alkali or alkaline earth metals with valency +1 and+2 respectively...
Q: Complete the following table: Cation Formula Anion Formula Compound Formula K+ Co,- Mg2+ NO, Ba2+ so...
A: To find: Compound formula of the given ions
Q: Draw the correct structure for (2S,3R)-2,3-diiodopentane. Show stereochemistry clearly. To ensure pr...
A: Stereochemistry of a molecule describes the 3-D arrangement of the given molecule. CIP rule is used ...
Q: How long will it take to plate out each of the following with a current of 200.0 Ampere? a. 1.0 kg o...
A: The given current, I = 200.0 Ampere. Known, I = Qt Here, I = The current in Ampere. t = The...
Q: please help with the following!
A: The concept: First, you have to find for how many moles of Al(s), the delta H is provided in the que...
Q: The moment of inertia of CH4 can be ca lcu lated from the expression I= 8/3 MHR2 where R = 109 pm is...
A: First, calculate the moment of inertia for CH4 as follows: I = 83MHR2= 83×1.67×10-27kg×109×10-12m2=...
Q: A hydrogen-filled balloon is ignited and 1.50 g of hydrogen reacts with 12.0 g of oxygen. How many g...
A:
Write the IUPAC name of each compound
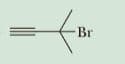
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images


