. A mixture of two liquids with the boiling point of 65 oC and 73 oC has been subjected to separation using a simple distillation technique. Identify ONE (1) reason for the failure of this experiment if the final product still contains the same mixture. State ONE (1) condition for the successful separation using this technique. b. In an experiment, 97.0 % from 1.00 g of solute X was extracted from 50.0 mL of an aqueous phase using two 25.0 mL portions of the organic solvent. Calculate the distribution ratio for solute S.
. A mixture of two liquids with the boiling point of 65 oC and 73 oC has been subjected to separation using a simple distillation technique. Identify ONE (1) reason for the failure of this experiment if the final product still contains the same mixture. State ONE (1) condition for the successful separation using this technique. b. In an experiment, 97.0 % from 1.00 g of solute X was extracted from 50.0 mL of an aqueous phase using two 25.0 mL portions of the organic solvent. Calculate the distribution ratio for solute S.
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter13: Gases
Section: Chapter Questions
Problem 12QAP
Related questions
Question
|
a. |
A mixture of two liquids with the boiling point of 65 oC and 73 oC has been subjected to separation using a simple distillation technique. Identify ONE (1) reason for the failure of this experiment if the final product still contains the same mixture. State ONE (1) condition for the successful separation using this technique. |
|
|
|
|
b. |
In an experiment, 97.0 % from 1.00 g of solute X was extracted from 50.0 mL of an aqueous phase using two 25.0 mL portions of the organic solvent. Calculate the distribution ratio for solute S. |
|
|
|
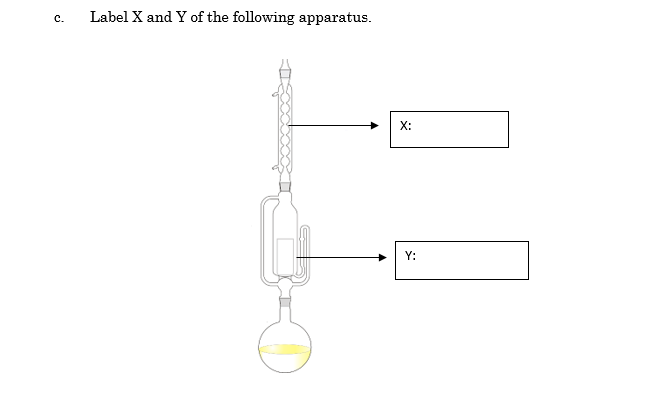
Transcribed Image Text:Label X and Y of the following apparatus.
с.
X:
Y:
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning