Q: The mass of solute per 100 mL of solution is abbreviated as (m/v). Mass is not techically the same…
A:
Q: Can someone help with this Mechanism question? Please make sure to include all curved arrow notation…
A: Acetal are organic compounds that have OR group attached to the same carbon. They are reactive due…
Q: 1.2.4 Interpret the data provided for the following reaction in terms of probable mechanism.…
A: Given are two organic reactions.To find the rate-determining step of the following reactions.
Q: 21. What is the possible product of the reaction shown? (B) H H C=C (A) H (C) Br C=C CH3 D CH3 CH3…
A: Alkyl halide gives an elimination reaction by reacting with a strong base. E2 reaction involves a…
Q: Predict the MAJOR ORGANIC product for the following reaction seque CH3OH, H₂SO4 (cat) 1.2 moles of…
A: Organic reactions are those in which organic reactant react to form organic products. In the given…
Q: Give detailed mechanism Solution with explanation needed..don't give Handwritten answer
A: A question based on the reaction of ketones. The most appropriate product for the given reaction…
Q: Antifreeze solution (a mixture of water and propylene glycol) is used in automobiles as a heat…
A: Answer:Properties that depend on the number of solute particles present in the solution are called…
Q: Draw the product of the reaction shown below. Ignore inorganic byproducts. 1 I 1. PhMgBr 2. H3O+ I I…
A: Answer:-This question is answered by using the simple concept of chemical reactions of aldehydes…
Q: the structures of the amino acids formed at physiological pH when the tripeptide below is hyd НО н…
A: Given is tripeptide molecule.Tripeptide consists of three amino acids.The tripeptide on hydrolysis…
Q: 5.0 g sample
A: When the mass of a substance is given, the number of moles is calculated by dividing the given mass…
Q: 6) The Diels-Alder reaction of anthracene (which is achiral) and maleic anhydride (also achiral)…
A: Anthracene is achiral and fumaric acid is also achiral because it have center of symmetry.
Q: 17) The two chair conformations of trans-1-bromo-2- tert-butylcyclohexane are shown. Identify…
A: A kind of elimination reaction of organic compounds that follows second-order kinetics. In such an…
Q: Ph Q ۱۱۱Ph OH но со
A: Here in the question, we need to find a reagent which can add two hydroxyl groups in an anti…
Q: Curved arrows are used to illustrate the flow of electrons. Use the reaction conditions provided and…
A: The objective of the question is to draw the product of the reaction.
Q: What is the major problem with the equation Ca (s) + 2 H₂O (1) --> Ca(OH)2 (aq) + H₂ (g)? Select…
A: When calcium reacts with water, it results in the production of hydrogen gas and calcium hydroxide.…
Q: Using bond energies, what is the heat of reaction for this chemical change? (units of kJ/mol are…
A:
Q: Draw the missing organic structures in the following multistep synthesis at physiological pH (pH =…
A: Step-1 ---> [NaOCH2CH3/CH3I] : Sodium ethoxide is a strong base, abstracts acidic protons.…
Q: rganic product of this 1. TsC1, Pyridine 2. NaCN 2017 ProtonGuru.com e R-configuration e…
A: The rate of SN2 reaction depends upon the concentration of both alkyl halide as well as…
Q: Name the functional groups and circle any chiral centers in these molecules?
A: The carbon atom in organic molecules is attached to four groups which are completely different atoms…
Q: Part B You have a racemic mixture of d-2-butanol and /-2-butanol. The d isomer rotates polarized…
A: Polarisation can be characterized as a phenomenon generated by wave characteristic nature of…
Q: What is the hybridization around the carbonyl carbon in the following compound: methanoic acid SP…
A: During orbital hybridization, the mixing of atomic orbitals takes place to form new orbitals known…
Q: Draw the two products of the reaction shown below. Assume only 1 equivalent of HBr is consumed in…
A: This is an example ethoxy deprotection
Q: BAT 10E. Flask A contains He, flask B contains O₂ and flask C contains Ne. All three closed flasks…
A: Given,Flask A contains 'He' gas.Flask B contains ' O2 ' gas.Flask C contains 'Ne' gastemperature =…
Q: CH₂CHCCI CH3 -OH
A: Carboxylic acid reacts with alcohol to form an ester in the presence of an acid catalyst, the…
Q: Figure 13-6 C4H8O2 11 10 HSP-03-534 9 8 1 7 1 6 ppm 5 1 2 4 : 3 3:3 2 1 0 A. B. c. 요 요. 애 애
A: Given,The molecules:
Q: Enter your answer in the provided box. Diethyl ether is produced from ethanol according to the…
A: The stoichiometry of the balanced chemical reaction states that two moles of ethanol produce one…
Q: Calculate the K a of a 0.32 M solution of X H that has a p O H of 12.2. Answer to three significant…
A:
Q: IUPAC
A: According to IUPAC nomenclature :Select the longest continuous chain of carbon atoms called as…
Q: In 13C NMR, carbon atoms with the lowest electron density have the largest chemical shift (largest…
A: Given that,In 13C NMR, carbon atoms with the lowest electron density have the largest chemical shift…
Q: Starting Material OH Terpineol KMNO4, KOH H₂O A R-carvone ... O S-carvone ki OH Limonene
A: The objective is to determine the starting reactant that would lead to the given product in the…
Q: Gasoline vaporizes in the carburetor of an automobile engine 2Hg (1) + O₂ (g) --> 2HgO (s)…
A: Given data:Different types of processes are givenMatch each reaction to the change in…
Q: Fill the blanks for the following reaction. CI + HCI + CO + CO₂
A: Acid chlorides are organic compounds that are represented using the following general formula,…
Q: 12. How many atoms decay in 12 hours in a 3.5 × 10-8 Ci source? 4.1 x 10-¹4 atoms 1.6 × 104 atoms…
A: Decay time= 12 hrsActivity or rate of decay = 3.5 x 10-8 Ci We have to find the number of atoms…
Q: What would be the mechanism for the acylation of iodobenzene to form the major product with the data…
A: When Iodo benzene is treated with acetyl chloride in presence of a Lewis acid (AlCl3 ), para…
Q: is fluorite unit cell electrically neutral support answer through calculions for this faced centered…
A: The objective of the question is to check whether fluorite is electrically neutral or not.
Q: What would be the structure of the final product when methanol is reacted with propionic acid…
A: Fisher esterification is a process of converting carboxylic acids and alcohols into esters (which…
Q: Draw the product of this reaction. Ignore inorganic byproducts. G KOH, A CI
A: The given reaction is based on elimination reaction.
Q: For many purposes we can treat ammonia (NH3) as an ideal gas at temperatures above its boiling point…
A: The objective of this question is to find the final pressure of a sample of ammonia gas when the…
Q: + Draw the Lewis structure of NCS (with minimized formal charges) and then choose the appropriate…
A:
Q: For the reaction below, identify the acid, the base, the conjugate acid, and the conjugate base.…
A: According to Bronsted and Lowry concept an Acid is a substance that has the tendency to donate a…
Q: 10: 8 2.04 (m, 2H), 2.91 (t, 4H), 7.17 (s, 4H).
A:
Q: Draw the 2 possible dipeptide products that can form from glycine and leucine. Draw at physiological…
A: The condensation reaction of amino acids to form a peptide bond is a key step in the synthesis of…
Q: Draw the product of the reaction shown below at physiological pH (pH = 7.4). Ignore inorganic…
A: During the synthesis of peptides, sometimes it is necessary to protect the amino group or carboxylic…
Q: Consider the reaction: CH₂COOH(aq) +SO (aq) HSO4(aq) + CH₂COO (aq) The K₂ for CH₂COOH is 1.75 x 105…
A:
Q: ropose a structural formula for the product of this reaction. O Et3N + CH3CH₂COCHCH₂CH₂CH₂Cl CHCH3…
A:
Q: Which of the following combinations of reactants are not likely to result in formation of a…
A: Answer:When solutions of two different substances are mixed and formation of precipitate takes place…
Q: Why do most living organisms have an enzyme to decompose hydrogen peroxide? deal Gas and…
A: Hydrogen peroxide is an inorganic compound with the chemical formula .
Q: What is the more stable radical in each pair? Pair 1 Pair 2 A A C a B A. B. D Pair 1 Pair 2 A C A D…
A: Radical is also known as a free atom. It is an ion, atom, or molecule which contains one unpaired…
Q: 2. Soaps (surfactants) are amphiphilic, meaning they have both polar and nonpolar domains. For…
A: Answer:Hydrophilic means water loving and hydrophobic means water repelling. It means if a specie is…
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product…
A: A Lewis acid is a substance that can accept a pair of electrons. On the other hand, a Lewis base is…
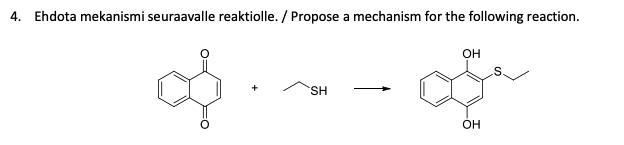
Step by step
Solved in 3 steps with 3 images


