Q: t the esterification mixture
A: This is a multiple questions.According to Guidelines, in multiple questions, we can solve only three...
Q: Question 7 What is the molarity of a solution made by dissolving 8.56 g of potassium acetate (KC2H3O...
A: Given data : Mass of KC2H3O2 = 8.56 g Volume of water = 750.0 mL = 0.750 L
Q: What is/are the proper descriptor(s) of the product of the following reaction? HN and trace acid O h...
A: Ro answer this question , we would first write the product of the reaction. Then we can check the de...
Q: grams óf NICI 2 (MW=129.5994g/mol) with 36.0 mL of 4.00M NaOH. How many grams of solid product (MW =...
A: Given : We have to find out the mass of solid product.
Q: Background Size A A A NaNO, + PbO > Pb(NO3)2 Na,0 +.
A: Since you have asked multiparts, we will solve the first three subparts for you. If you want any spe...
Q: Consider the following balanced reaction: Cu + 4HNO3 → Cu(NO3)2 + 2NO2 + 2 H20 If 0.0424 mol Cuare m...
A:
Q: Calculate the following for each run: the initial molarities of I- and H2O2 (from the solutions tab...
A: Total volume of the reaction mixture = 65.5 mL [KI]stock = 0.04 M [H2O2]stock = 0.04 M [S2O...
Q: What is a greenhouse gas? Give the chemical formulas of two compounds that function as greenhouse ga...
A: Greenhouse gases are the gases that cause greenhouse effect by absorbing infrared radiation which wa...
Q: Iron has a density of 7.87 g/cm^3 and crystallizes in a body-centered cubic structure. What is the a...
A: Body centred cubic unit cell have atoms that occurs at the corners as well as in the centre of unit ...
Q: Lime, CaO, can be produced in two steps as shown in the equations below. If the percentage yield of ...
A:
Q: At Stp conditions, 11g of SO2 have a volume of
A: Given :- mass of SO2 = 11 g To be calculated :- volume of SO2 at STP
Q: Ammonium nitrate fertilizer is sometimes used as an explosive. How many moles of water can be formed...
A:
Q: What's in your Bucket (write the molecule and what type--WB, WA, SA, SB, buffer, or water) before an...
A: Answer: Before the addition of NaOH, in the solution we are having C2H5NHClO4, which is the salt of ...
Q: A student prepares a dilute solution by adding 10.00 mL of a stock solution of a monoprotic weak aci...
A:
Q: L204 J. Cas(PO4)2 5. Acid-Base and Gas-Evolution Reactions Write a balanced molecular, total ionic, ...
A:
Q: Visualize/sketch molecular 4. Model orbital(s), HOMO, HOMO-1 and HOMO-2 and identify if it is o- or ...
A: Solution Given that Model C6H6. Visualize/sketch Find Molecular orbital(...
Q: C2H4: HOMO-1 ole C2H2: HOMO ng ond Sond C2H2: HOMO–1 o pais ng eis C2H2: HOM0-2 regativj momeut ch V...
A: Solution Given that C2H4: HOMO - 1 C2H2: HOMO C2H2: HOMO - 1 C2H2: HOMO...
Q: Compare the experimental bond length data shown below with your Lewis Structure drawing. Describe an...
A: The answer is as follows:
Q: The initial concentrations of \ce{SCN-}SCNX− after dilution were calculated as part of the Prelab Ex...
A: Answer.
Q: What is the IUPAC name for the compound Fe[Fe(CN)6] A. Iron(III) hexacyanoferrate(II) B. Iron(11) he...
A: In IUPAC nomenclature we have to name the cation first then we name the complex. In the compound fou...
Q: What mass of Ba(IO3)2 (487 g/mol) can be dissolved in 1 L of water at 25°C? Ba(IO3)2 ⇌ B...
A:
Q: Describe the similarities and differences between ionizing and nonionizing radiation. FORM OF RADIA...
A: An atom is composed of protons, neutrons, and electrons. It will require some energy to eject electr...
Q: 2Fe(OH)3 + 3(NH4)2SO4. Calculate the mass of (NH4)2SO4 produced when 3.67 g Fe(OH)3 Consider Fe2(SO4...
A:
Q: How do you draw the structure with the name (3S)-4-chloro-2,6-dimethylhepta-1,5-dien-3-ol
A: Given :- IUPAC name = (3S)-4-chloro-2,6-dimethylhepta-1,5-dien-3-ol To draw :- Structure of given...
Q: Which is true of a system at equilibrium Kc - 1? Equilibrium favours neither reactants nor products ...
A: Answer :
Q: 2. Differentiate Boyle's law and Charles' law.
A: Given, Boyle's law & Charles's law Required, Differentiate between ...
Q: hich one of the following conjugate acid-base pairs is correct? Acid Base HCO3 NO3 a. b. NO3 HNO3 HC...
A:
Q: For the following reaction, 17.4 grams of chlorine gas are allowed to react with 3.50 grams of water...
A: The correct answer is given below
Q: Indicate which atoms in the following compound are chiral centers. H3C. HO H3C CH2 (Provide a list o...
A:
Q: Draw the major organic product of the coupling reaction shown. Select Draw Rings More Erase H Pd(OAc...
A:
Q: Hello, I need help in determining the IUPAC name for the following compound. I have been stuck on it...
A: This is bicyclic compound , to name it keep following rules in mind. 1. Numbering should start from...
Q: hydrogen peroxide was added to a piece of potatoes contain the enzyme, catalyze, hydrogen peroxide w...
A: It is an interesting phenomena ,when we add hydrogen peroxide to fresh potatoes it breaks down into ...
Q: b.) How is the rate Of disappearance of S,0J -relnted to the rate of It appeatratice of Ij ? (al.
A:
Q: Explain the meaning of third-spacing.
A: The movement of bodily fluid from the blood, into the spaces between the cells. Or the accumulati...
Q: Please provide a complete, arrow-pushing mechanism for each of the below reactions. The following pr...
A: Since you have asked multiple question, we will solve the first question for you. If you want any sp...
Q: 50.0 g of N2O4 is introduced into anevacuated 2.00 L vessel and allowed to come to equilibrium ...
A: The equilibrium reaction given is, => N2O4 (g) → 2 NO2 (g) Given: Initial mass of N2O4 = 50.0 g. ...
Q: Determine the molar solubility for Cd:(PO.)2 by constructing an ICE table, writing the solubility co...
A: Given: Concentration of Na3PO4 = 0.500 M And Ksp of Cd3(PO4)2 = 2.5 × 10-33 The solubility reaction...
Q: Calculate e* values for the galvanic cells based on the following overall reactions. Assume that all...
A:
Q: 0.60 g of hydrogen is allowed to react with 4.00 g oxygen to form water. 2 H2(g) + O2(g) → 2 H2O(l) ...
A: We have to predict the excess chemical and amount of product formed.
Q: 0.2 0.5×10-4 1x10-4 1.5x10-4 2x10-4 [Fe(SCN2+)]equ, (M) y = 4.26442 × 103 x + 0.0202852 R2 = 0.99990...
A:
Q: You put a pot of water on the stove and the water begins to boil. System: Surrounding: Direction of ...
A: Exothermic reaction are those reaction in which heat is evolved while reaction in which heat is abso...
Q: Which never changes the position of a system at equilibrium? O Changing the concentration of one com...
A:
Q: Mark whether the following compounds are soluble or insoluble in water: A: Hg2l2 B: Barium Sulfate C...
A: To check the solubility of these compounds , we would use solubility rules . Note : As you have ...
Q: 3. E ) Listen What type of system is able to exchange both energy and matter with its surroundings? ...
A: An Open system is a system in which exchange of both matter and energy with the surrounding take pla...
Q: Caliulate the pth of l15 M tydrofluoric acíd (HP) it the k =7.2x10*? YYAN
A: Given, Concentration of hydrofluoric acid, [HF] = 1.15 M Ka for HF = 7.2 × 10-4 pH of the solution =...
Q: A solution contains 0.106 M sodium cyanide and 0.344 M hydrocyanic acid. The pH of this solution is ...
A: A question based on equilibrium concept that is to be accomplished.
Q: Suppose a 500. mL flask is filled with 1.5 mol of O, and 0.60 mol of NO. The following reaction beco...
A: Please note- As per our company guidelines we are supposed to answer only one question. Kindly repos...
Q: What is the molarity of ions in a 0.838 M solution of Ca(OH)₂ assuming the compound dissociates comp...
A:
Q: 1.(b): we assume that they dissociate 100% Complete the ICE table: HNO3(aq) + H2O(l) -> H3O+(aq) + N...
A: Answer: In this question we have to find out the concentration of hydrogen ions in acid solutions to...
Q: Sodium formate reacts with permanganate in neutral solution according to the equation CHO2 + MnO4 +...
A:
Step by step
Solved in 2 steps with 2 images

- Ibufenac, a para-disubstituted arene with the structureHO2CCH2C6H4CH2CH(CH3)2 , is a much more potent analgesic thanaspirin, but it was never sold commercially because it caused livertoxicity in some clinical trials. Devise a synthesis of ibufenac frombenzene and organic halides having fewer than five carbons.Compound 1 is an anticoagulant that is extracted from, among other plants, the sweetclover plant, Melilotus Officinalis. However, the compound is moderately toxic and alsofinds use as a rodenticide. Name this compoundA chiral amine A having the R conguration undergoes Hofmann elimination to form an alkene B as the major product. B is oxidatively cleaved with ozone, followed by CH3SCH3, to form CH2 = O and CH3CH2CH2CHO. What are the structures of A and B?
- A chiral amine A having the R configuration undergoes Hofmann elimination to form an alkene B as the major product. B is oxidatively cleaved with ozone, followed by CH3SCH3, to form CH2 = O and CH3CH2CH2CHO. What are the structures of A and B?4.4 Show how the compound thiodiazine - C21H26N2S2 - decomposes anaerobically and how these end products in turn decompose aerobically to stabilized sulfurand nitrogen compounds.Quinapril (trade name Accupril) is used to treat high blood pressure andcongestive heart failure. One step in the synthesis of quinapril involvesreaction of the racemic alkyl bromide A with a single enantiomer of theamino ester B. Given the structure of quinapril, which one of these two products isneeded to synthesize the drug?
- The shrub ma huang (Section 5.4A) contains two biologically activestereoisomers—ephedrine and pseudoephedrine—with two stereogeniccenters as shown in the given structure. Ephedrine is one component ofa once-popular combination drug used by body builders to increaseenergy and alertness, whereas pseudoephedrine is a nasaldecongestant.a.) Draw the structure of naturally occurring (−)-ephedrine, which has the1R,2S configuration.b.) Draw the structure of naturally occurring (+)-pseudoephedrine, whichhas the 1S,2S configuration.c.) How are ephedrine and pseudoephedrine related?d.) Draw all other stereoisomers of (−)-ephedrine and (+) pseudoephedrine, and give the R,S designation for all stereogeniccenters.e.) How is each compound drawn in part (d) related to (−)-ephedrine?5. Suggest the synthesis pathways for the following:Write the structure of the Maleic Anhydride compound and label each non-equivalent carbon with a letter, A,B,C...
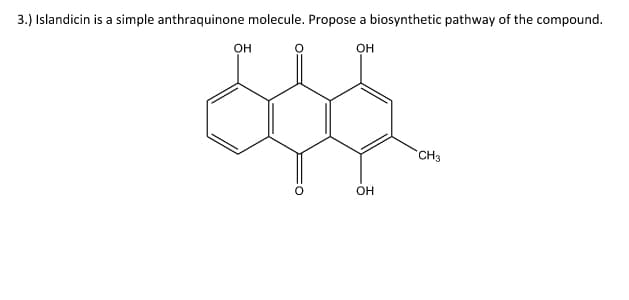
- The following three derivatives of succinimide are anticonvulsants that have found use in the treatment of epilepsy, particularly petit mal seizures. Q. Show how this same synthetic strategy can be used to prepare ethosuximide and methsuximide.Propose a synthesis pathway for the production of the following compounds (d) to (e) from the given starting materials plus necessary organic and inorganic reagents.Give the expected organic product when phenylacetic acid, PhCH2COOH, is treated with reagent Q.)NaHCO3, H2O



