Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter11: Solutions And Colloids
Section: Chapter Questions
Problem 27E: What is the microscopic explanation for the macroscopic behaviour illustrated in Figure 11.15?
Related questions
Question
100%
Give detailed Solution with explanation needed..please explain..give all possible reagents to making following product.
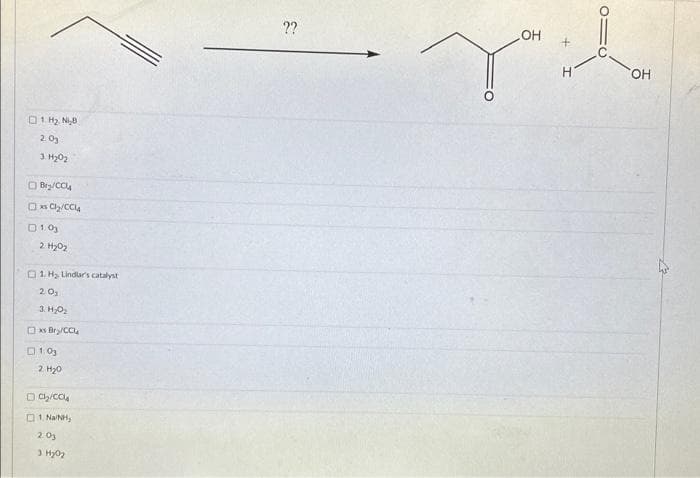
Transcribed Image Text:0 1 H2 N 8
2.03
3 H₂0₂
Bry/CCl4
xsCl₂/CCL4
01.0
2 H2O2
0 1.H2, Lindue's catalyıt
20₂
3. Н.02
xs Bry/CCL
1:03
2 H2O
Ch/COLA
1. Na/NH,
2.0₂
3. H2O
??
OH
+
OH
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax